- Theoretical Neuroscience Research, LLC, Ridgeland, MS, United States.
DOI:10.25259/SNI_351_2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Russell L. Blaylock. Viruses and tumor cell microenvironment: A brief summary. 09-Aug-2019;10:160
How to cite this URL: Russell L. Blaylock. Viruses and tumor cell microenvironment: A brief summary. 09-Aug-2019;10:160. Available from: http://surgicalneurologyint.com/surgicalint-articles/9570/
Abstract
An infectious etiology for a number of cancers has been entertained for over 100 years and modern studies have confirmed that a number of viruses are linked to cancer induction. While a large number of viruses have been demonstrated in a number of types of cancers, most such findings have been dismissed in the past as opportunistic infections, especially with persistent viruses with high rates of infectivity of the world’s populations. More recent studies have clearly shown that while not definitely causing these cancers, these viruses appear capable of affecting the biology of these tumors in such a way as to make them more aggressive and more resistant to conventional treatments. The term oncomodulatory viruses have been used to describe this phenomenon. A number of recent studies have shown a growing number of ways; these oncomodulatory viruses can alter the pathology of these tumors by affecting cell signaling, cell metabolism, apoptosis mechanisms, cell-cell communication, inflammation, antitumor immunity suppression, and angiogenesis. We are also learning that much of the behavior of tumors depends on cancer stem cells and stromal cells within the tumor microenvironment, which participate in extensive, dynamic crosstalk known to affect tumor behavior. Cancer stem cells have been found to be particularly susceptible to infection by human cytomegaloviruses. In a number of studies, it has been shown that while only a select number of cells are actually infected with the virus, numerous viral proteins are released into the cancer and stromal cells in the microenvironment and these viral proteins are known to affect tumor behavior and aggressiveness.
Keywords: Cancer microenvironment, Cancer stem cells, Oncomodulation
ONCOMODULATION BY PERSISTENT VIRUSES
There are some 1400 human pathogens that include 220 types of viruses. A small percentage of these are known to cause cancer by a number of methods and are referred to as oncogenic viruses. A newer concept is one that involves virally induced mechanisms that alter the biology and aggressiveness of a growing number of cancers including gliomas, medulloblastomas, breast adenocarcinomas, prostate adenocarcinomas, osteosarcomas, colon adenocarcinomas, and mucoepidermoid salivary carcinomas.
Oncomodulation is a process, whereby tumor behavior is affected by virally induced mechanisms acting on cancer cells as well as stromal cells within the tumor microenvironment, including suppression of cancer cell apoptosis mechanisms, altering tumor metabolism, suppressing antitumor immunity, triggering tumor microenvironment inflammation, initiating angiogenesis, stimulating tumor cell proliferation, increasing tumor invasion, and enhancing tumor cell migration (metastasis) [
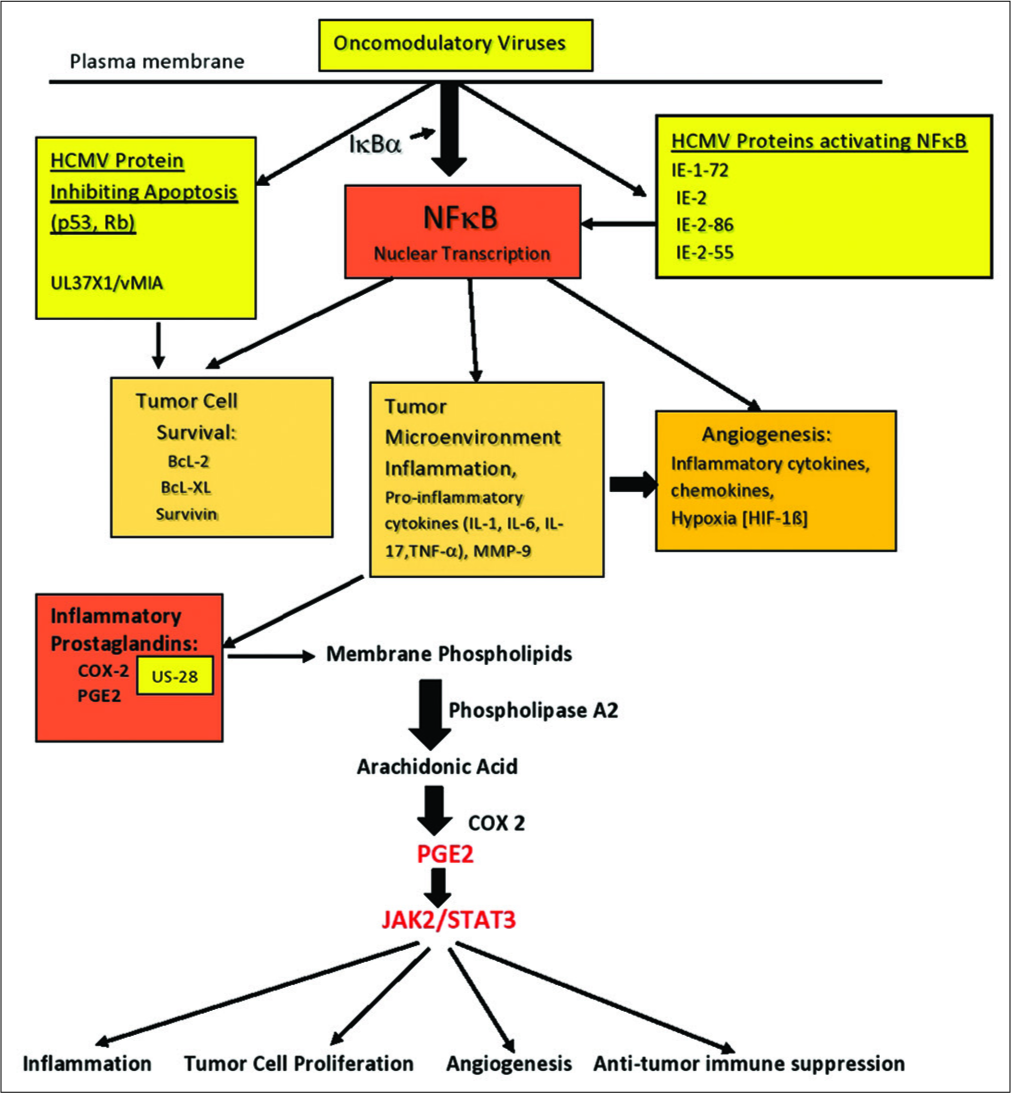
Figure 2:
Activation of cyclooxygenase-2 leading to the generation of prostaglandin E2 (PGE2) and subsequent enhancement of inflammation. Tumor cell proliferation, angiogenesis, and suppression of antitumor immune activity are enhanced in the face of tumor microenvironment inflammation. The central role played by NF-kB, found in all cells, is demonstrated. Activation of this factor occurs in the stromal cells, tumor cells, and invading immune cells within the tumor microenvironment. PGE2 activation of JAK2/STAT3 acting on genes controlling tumor invasion/migration, cell proliferation, angiogenesis, and initiating immune suppression is demonstrated.
In the past, the debate was centered on the idea that for a virus to have oncogenic effects, all of the cells in the tumor must be infected. Until recently, most studies used inadequate technology to detect these viruses. Most importantly, it was determined that it was not necessary for all of the tumor cells to be infected with the virus in question. Rather, only a few cells need to be infected, with the release of viral proteins from the viruses, which occupy most if not all of the tumor cells, including stromal cells. This finding invalidates many of the previously reported negative studies.
CHRONIC INFLAMMATION, VIRUSES, AND THE DEVELOPMENT OF CANCER CELLS
Some concluded that the virus was present in the tumor merely because the tumor supplied a growth environment that was favorable to the virus. Newer studies have elucidated a number of mechanisms, by which these viruses affect tumor biology and aggressiveness.
It is now accepted that inflammation is playing a central role in most, if not all, cancers [
Tumors are composed of not only cancer cells but also over 50% stromal cells, which make up the tumor’s microenvironment. Within this microenvironment, one sees a number of cell types, including T-cells, B-cells, NK cells, fibroblast, adipocytes, vascular endothelial cells, and pericytes, all of which crosstalk and release factors that support tumor survival, growth, invasion, and migration.
VIRUSES INFECTING IMMUNE CELLS AND TUMOR CELLS: CHANGING IMMUNE CELLS TO BECOME IMMUNOSUPPRESSIVE AND PRODUCE FACTORS CHANGING TUMOR CELL METABOLISM
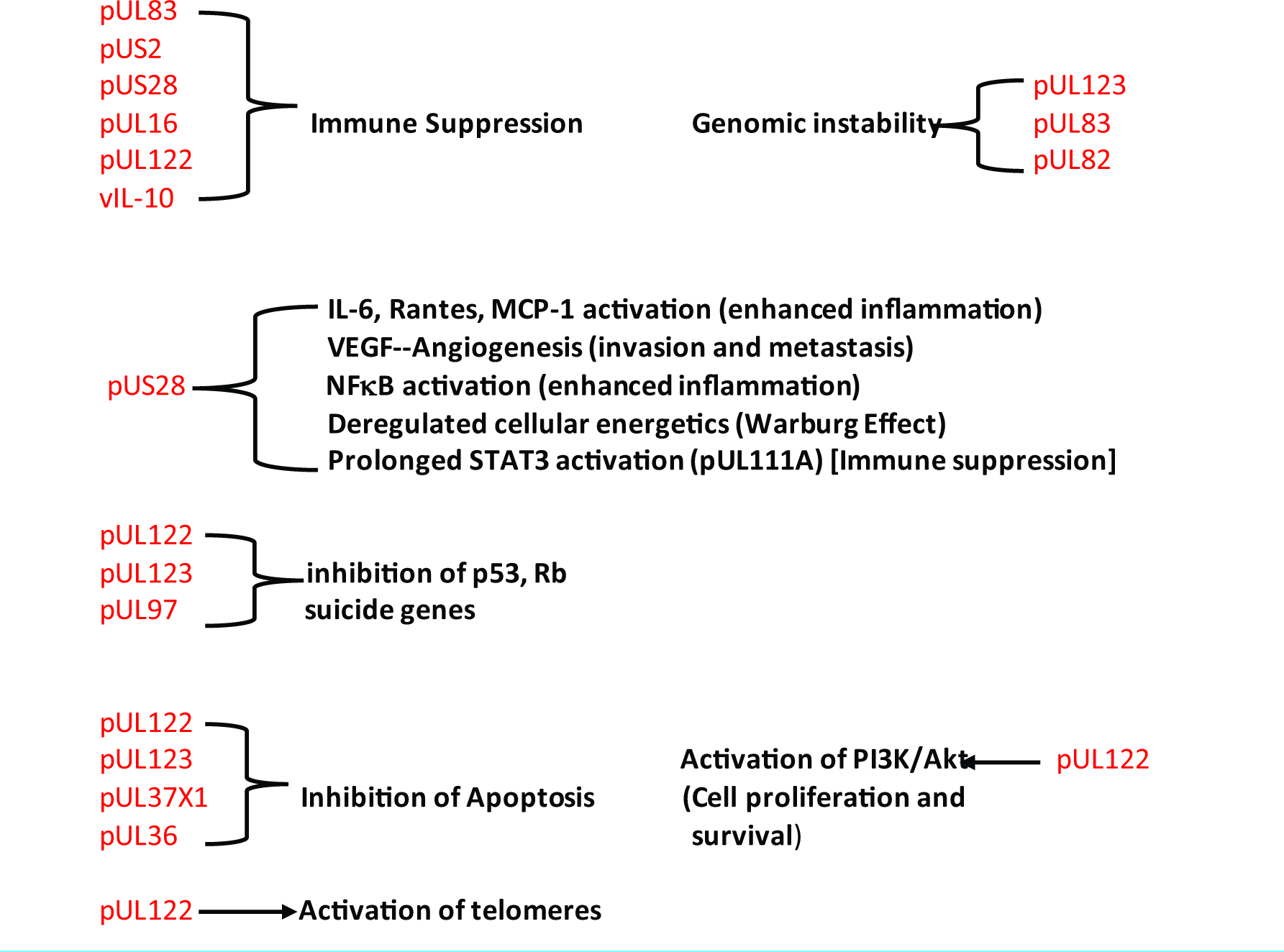
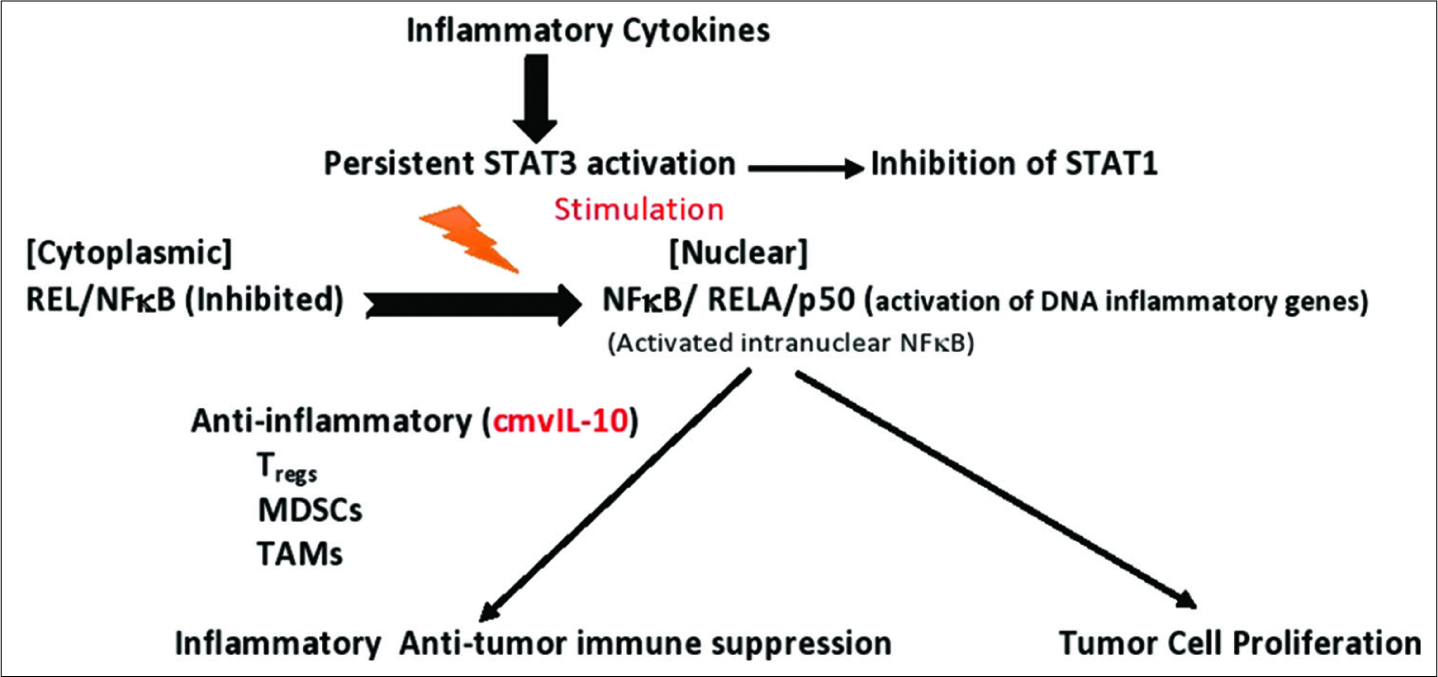
HCMV preferentially infects immune cells and tumor stem cells. The infected immune cells assume an immunosuppressive phenotype and there is evidence that HCMV-infected cells can produce a protein that resembles IL-10 (cmvIL-10), a major immune suppressing protein [
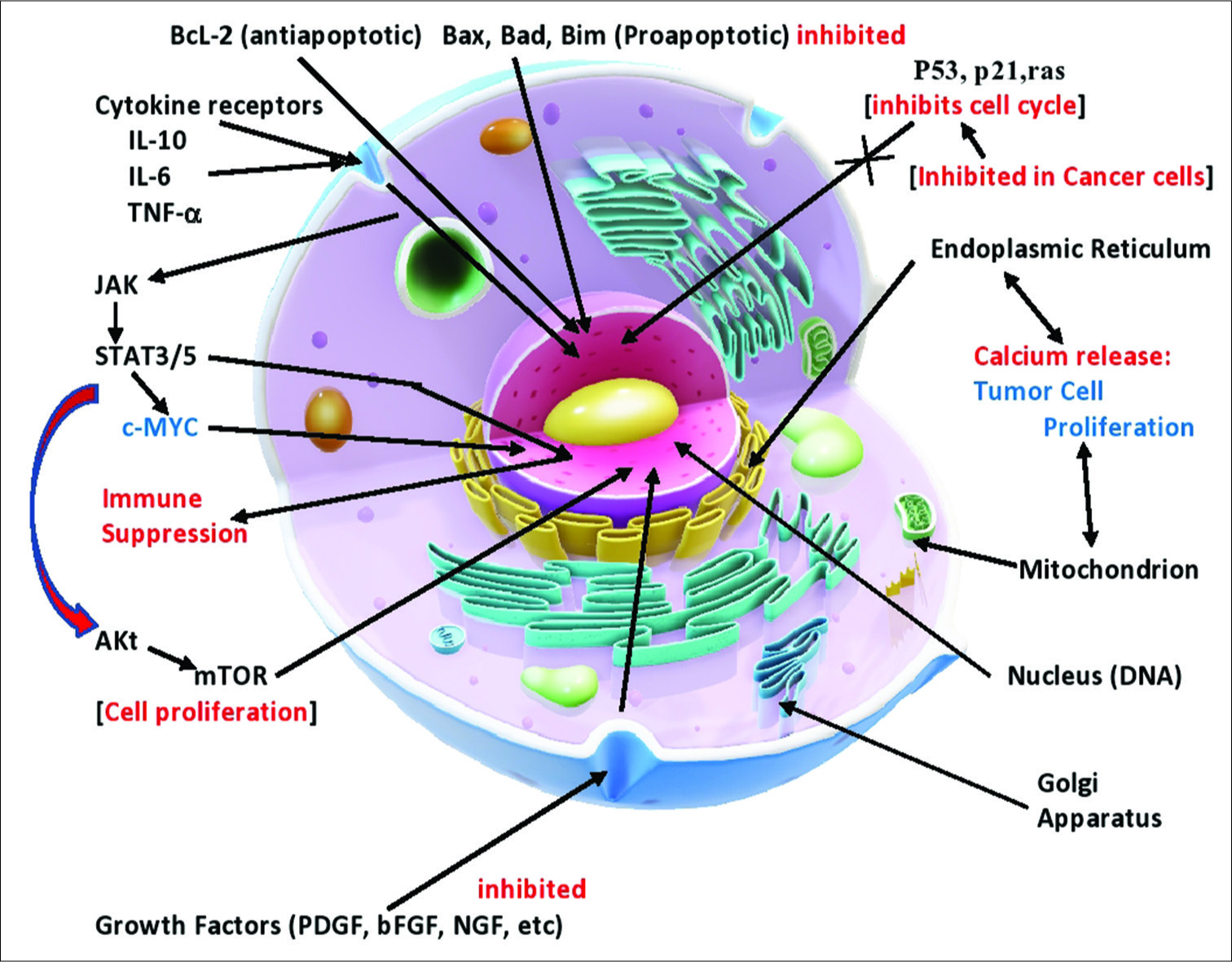
A number of immune cytokines play critical roles in cancer behavior, including tumour necrosis factor-α and IL-6, which are both linked to activation of the transcription factor, NFκB. This transcription factor is intensely activated in most cancers. IL-6 generation by NFκB plays a major role in tumor aggressiveness, and cancers with high levels of this cytokine have a poor prognosis. HCMV activates all of these factors [
VIRUSES INFLUENCING TUMOR CELL SIGNALING PATHWAYS LEADING TO TUMOR CELL GROWTH
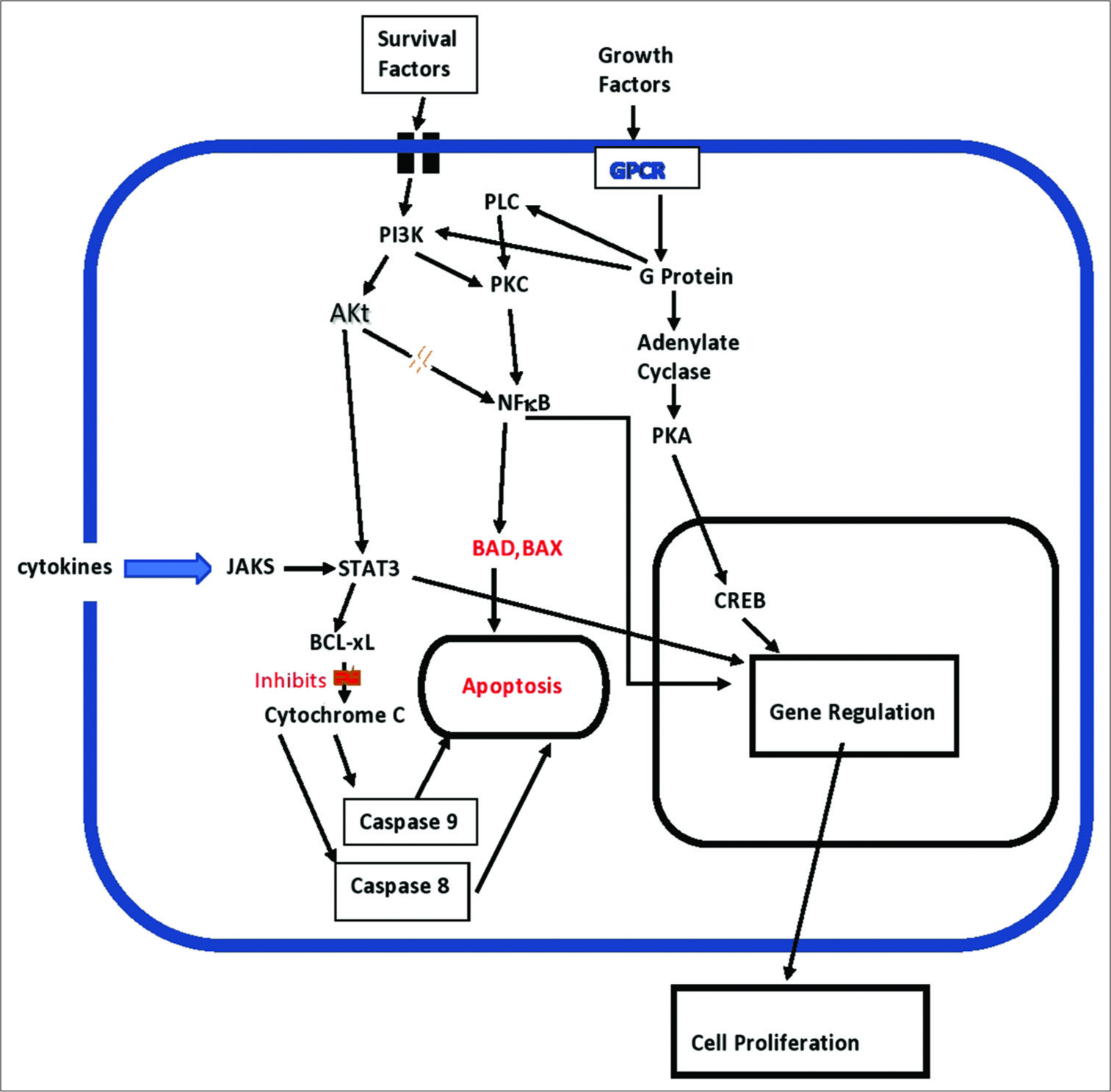
Another way oncomodulatory viruses influence tumor biology is by affecting cell signaling. HCMV and other oncomodulatory viruses have been shown to activate the JAK/Stat pathway toward overproduction of STAT3, which has a number of effects supporting tumor aggressiveness, including stimulating intense inflammation, suppression of antitumor immunity, stimulation of cancer cell proliferation, inhibiting apoptosis mechanisms, stimulating survival mechanisms, promoting angiogenesis, enhancing tumor invasion, and promoting cancer cell migration [
HCMV also stimulates the PI3K/AKt cell-signaling pathway, which enhances cell proliferation and promotes cell survival by reducing apoptotic proteins, such as BAD, and activating cell survival proteins, such as bcl-2 and bcl-XL [
Figure 5:
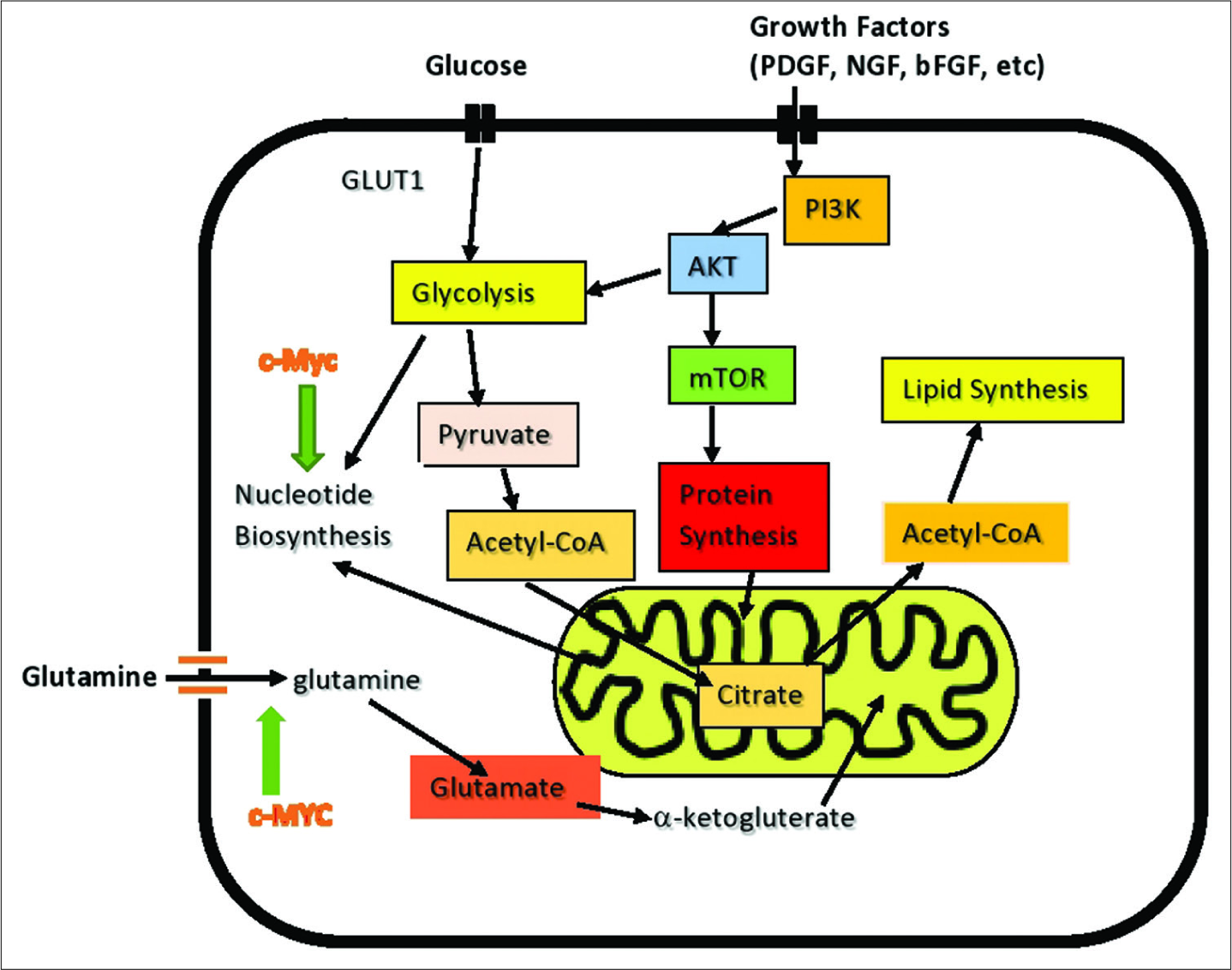
Cancer cell metabolism involving its two major fuels, glucose and glutamine. Demonstrates role of c-MYC in induction of glutamine addiction. Myc consists of a family of regulator genes (proto-oncogenes) coding for transcription factors. The Myc family consists of c-myc, l-myc, and n-myc and also demonstrates the influence of growth factors on cancer cell signaling.
Tumor microenvironment hypoxia is known to make cancer significantly more aggressive, mainly by activating hypoxia- inducible factor-1α. This factor is also a major inducer of angiogenesis. Oncomodulatory viral proteins are also known to potently stimulate the production of angiogenic factors. HCMV is unique in promoting the release of a powerful secretome from cells in the tumor microenvironment, which contains the tumor invasion factor matrix metalloproteinase and the growth factors such as vascular endothelial growth factor, basic fibroblast growth factor (FGF), FGF-binding protein, and platelet-derived growth factor. In addition, HCMV enhances the formation of telomerase (human telomerase reverse transcriptase), which promotes tumor cell survival.
VIRUSES ALTERING THE TUMOR CELL METABOLISM
Finally, oncomodulatory viruses also affect the metabolism of cancer cells [
Figure 6:
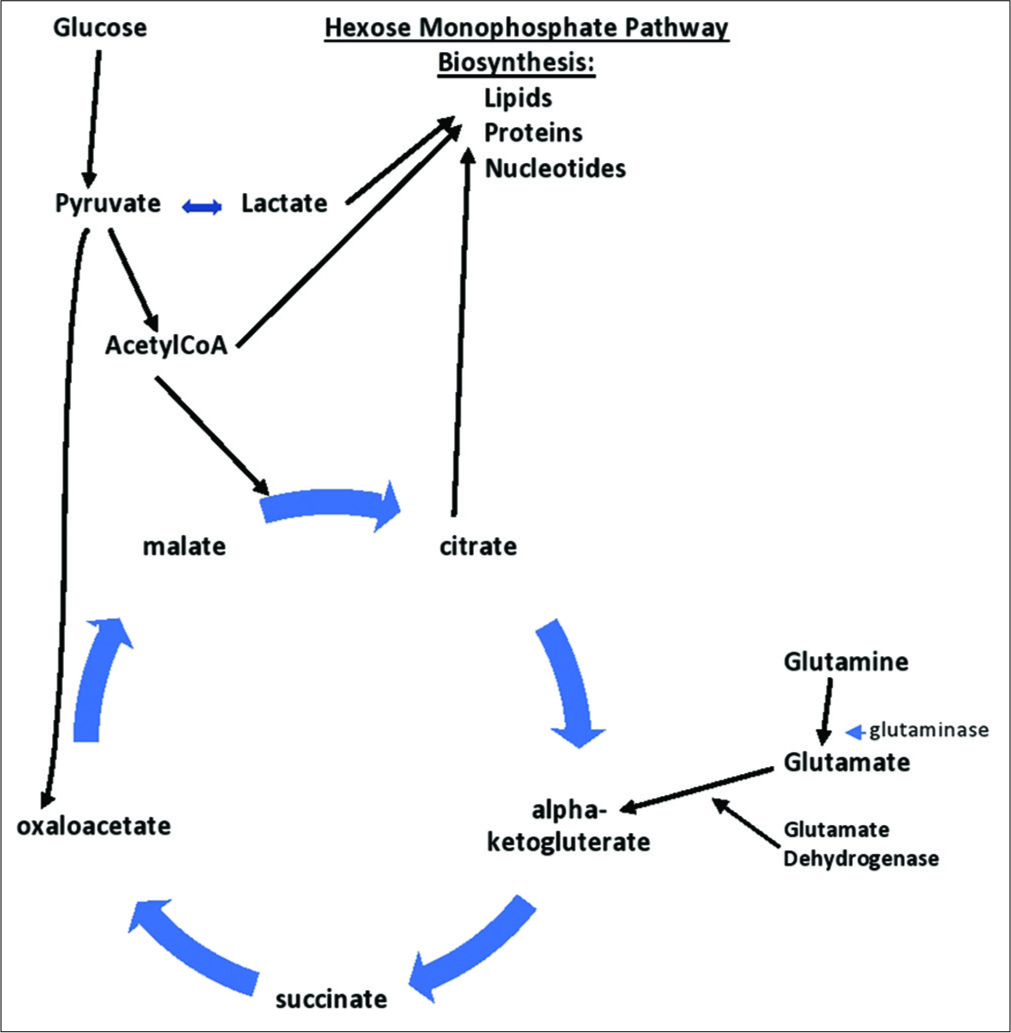
Warburg effect altering cellular metabolism to maximize use of glycolysis and Kreb’s cycle intermediates for biosynthesis of lipids, proteins, and nucleotides for viral and cancer cell reproduction. Demonstrating glutaminolysis pathway for converting glutamine to glutamate and then to alpha-ketoglutarate within Kreb’s cycle, used for biosynthesis of substrates.
It has been shown that HCMV infections can switch infected cells’ metabolism toward a Warburg-like metabolism, much like cancer cells. While cancer cells can become dependent on glucose as a major fuel, recent evidence suggests that an alternate fuel may be of more importance – glutamine. In a process called glutaminolysis, glutamine is increasingly taken up by cancer cells, where it is converted to glutamate by the enzyme glutaminase and then further metabolized into alpha-ketoglutarate by the enzyme glutamate dehydrogenase [
GLUTAMINE STIMULATES TUMOR CELL GROWTH
Glioblastomas are known to have high levels of glutamine consumption and are glutamine addicted. The oncogene, c-myc, plays a major role in converting cancer cells to glutamate/glutamine dependence. Tumors with the highest c-myc levels also have the highest glutamate levels.
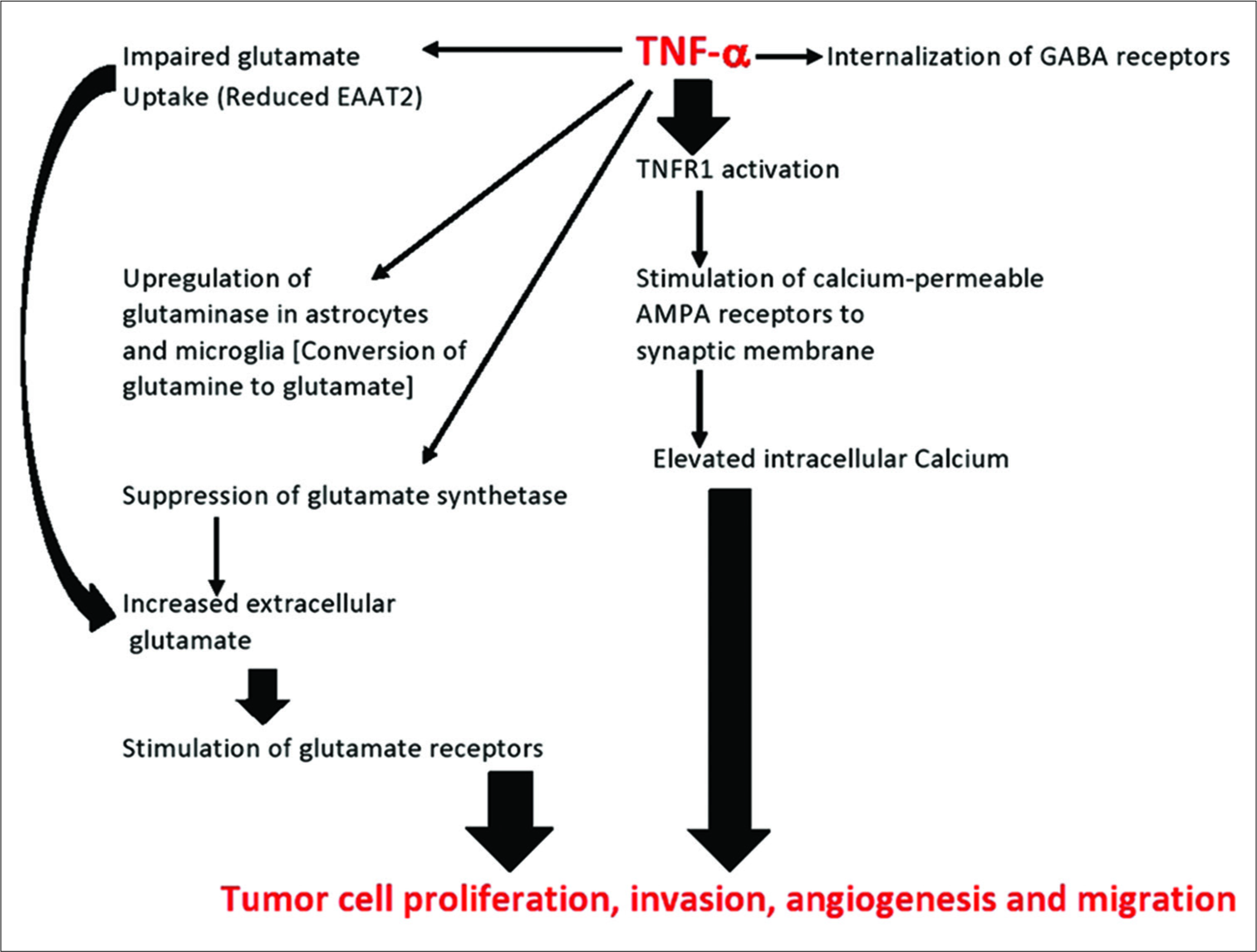
Interestingly, glutaminolysis is not the major source of glutamate in tumors. Recent studies have shown that most of the extracellular glutamate within the tumor microenvironment comes from the cystine/glutamate Xc antiporter, a membrane exchange system that exchanges extracellular cystine for intracellular glutamate. Once inside the cell, the cystine is metabolized to form glutathione, which protects the cell. The extruded glutamate raises the extracellular glutamate levels, which could trigger a cascade of neurodestruction. This is normally prevented by the presence of glutamate transport proteins that move the extracellular glutamate inside astrocytes and microglia, where it is safe. The most abundant of these transfer proteins is GLT-1 (EAAT2), which is absent in gliomas, thus allowing glutamate to rise to excitotoxic levels in the extracellular space.
While glutamate stimulates tumor growth by glutaminolysis, it also does so by the stimulation of glutamate receptors [
TUMOR CELL MIGRATION EFFECTED BY GLUTAMATE ACTIVATED GLUTAMATE RECEPTORS ON THE TUMOR CELL
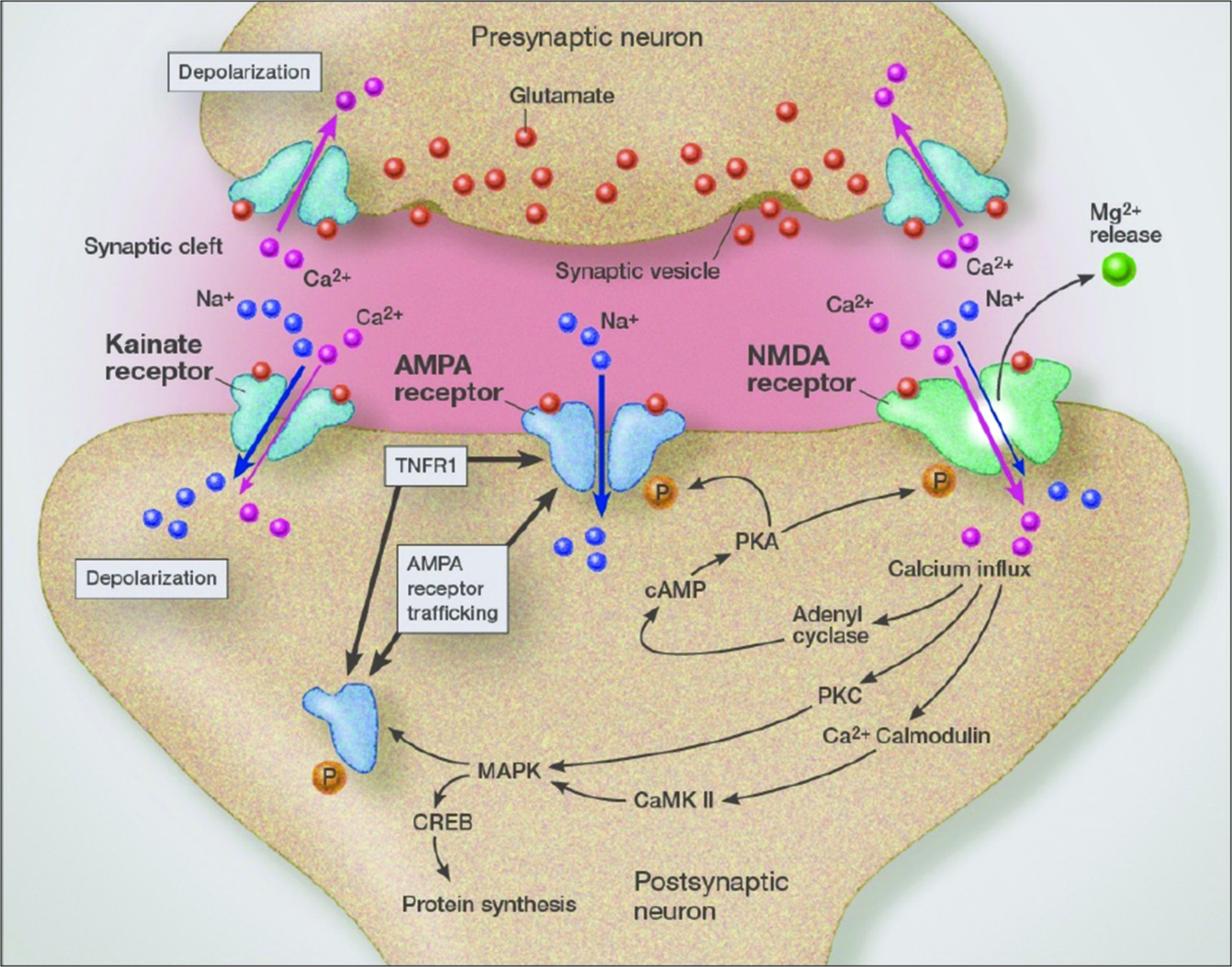
Tumor cells migrate much in the same way as embryonic cells, which are driven by gradients of glutamate. Studies have shown that migration of both types of cells, embryonic and cancer cells, is dependent on calcium oscillation (varying gradients of calcium), which are determined by calcium-controlling glutamate receptors [
Under most normal conditions, the AMPA-type glutamate receptors are calcium impermeable, meaning they do not regulate calcium entry into the cell. Under inflammatory conditions, as with cancers, special calcium-permeable AMPA receptors are recruited to the synaptic plate [
TREATMENT OF VIRUSES BY ANTIVIRAL MEDICATIONS AND NATURAL COMPOUNDS
As one can see, these oncomodulatory viruses can play a major role in tumor behavior, potentially converting an otherwise curable cancer into an incurable one. A number of studies have shown that treating these viruses with antiviral medications and similar substances can reduce the malignant potential of many types of tumors. It is also encouraging to note that several natural compounds, each with a high margin of safety, can also dramatically reduce viral loads, potentially having the same beneficial effects as the antiviral medications. Most such compounds also have a number of other anticancer effects and are much safer than antiviral medications.
DNA MUTATIONS NOT NECESSARY FOR CANCER CELL DEVELOPMENT: CHRONIC INFLAMMATION AND VIRUSES CAN CAUSE EXCESSIVE STIMULATION OF CELL-SIGNALING PATHWAYS THAT CAN PRODUCE CANCER CELLS
One important observation that has changed our thinking in the field of oncology is that DNA mutations are not absolutely necessary for cellular transformation into a cancer. New studies have shown that some cancers have no DNA mutations and other studies have shown that hyperactivity of certain cell-signaling pathways alone can transform cells into cancer cells. Hyperactivity of these pathways comes by excessive stimulation of membrane receptors – cytokine/chemokine receptors, DAMPS, PAMPS, glutamate receptors, and Toll-like receptors. This is one reason for the strong connection to inflammation in carcinogenesis – the inflammation activates the inflammatory cytokine receptors chronically. Ironically, both chemotherapy and radiation are very inflammatory and can stimulate the release of factors that drive cancer cell signaling. This may be why it takes so long to eventually fully transform these exposed cells. That is, a series of coordinated cell-signaling events must take place to allow survival of the transformed cells. This is the importance of the oncomodulation – unlike haphazard free radical damage to chromosomes or inflammation alone, the viruses are expert at activation of just the proper cell-signaling pathways that favor both viron survival and cancer cell survival and proliferation. This may be the future target in oncology – controlling these oncomodulatory viruses. Indeed, several studies have shown that suppressing these latent viruses does improve, often dramatically, the control of many cancers. Oncomodulation is directed by the viral proteins that are released into the tumor microenvironment and not by the virus itself. This negates the idea that all or most of the cells in a tumor must be infected by the virus to cause tumor development. Only a small percentage of the 200 viral proteins released by cytomegalovirus has anything to do with viral replication – most alter cell signaling.
CONCLUSION
Over the past 30 years, we have learned a great deal about the incredibly complex mechanisms involved in the transformation of normal cells into cancer cells, especially as regards cell-signaling pathways. While mutations and epigenetic overexpression of genes can result in this transformation, increasing evidence suggest that overactivation of critical cell signaling pathways can also result in transformation of cells toward malignancy. Universal to both genetic and cell-signaling induced malignant transformation is the inflammatory process, especially chronic, low-grade inflammation. Inflammation not only increases free radical generation and lipid peroxidation products that can damage and mutate oncogenes but can also directly overactivate cell-signaling pathways essential to carcinogenesis and tumor behavior.
Recent evidence demonstrates a much higher incidence of viral infection of a number of cancerous tumors than previously appreciated, based on greatly improved detection systems and the realization that all tumor cells do not have to be infected to make the link. Recent studies have shown that infection of a small number of cells, especially cancer stem cells and progenitor cells, can result in the release of a large number of viral proteins that can infect numerous tumor and stromal cells in the microenvironment, and as a result, trigger carcinogenesis and of equal importance, can drive a number of tumor mechanisms involved in tumor cell metabolism, immune resistance, proliferation, tumor invasion, and tumor cell migration. Potentially, this can convert relatively benign, mostly localized cancers to become more aggressive, rapidly growing, more invasive, and more likely to metastasize than they would otherwise. The ability of infectious organisms to alter tumor biology has been termed oncomodulation.