- Department of Neurosurgery, Kasr Alainy Faculty of Medicine, Cairo University, Giza, Egypt.
- Department of Neurosurgery, Cairo University Kasr Alainy Faculty of Medicine, Children Cancer Hospital, Cairo, Egypt.
DOI:10.25259/SNI_318_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ayman Tarek Mahmoud1, Abdelrhman Enayet2, Ahmed Mohamed Ahmed Alselisly1. Surgical considerations for maximal safe resection of exophytic brainstem glioma in the pediatric age group. 28-Jun-2021;12:310
How to cite this URL: Ayman Tarek Mahmoud1, Abdelrhman Enayet2, Ahmed Mohamed Ahmed Alselisly1. Surgical considerations for maximal safe resection of exophytic brainstem glioma in the pediatric age group. 28-Jun-2021;12:310. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10926
Abstract
Background: Brainstem glioma is the leading cause of morbidity and mortality among all central nervous system tumors, especially in childhood as it represents about 20% of all pediatric brain tumors. Therefore, this study aimed to present our experience in a tertiary center in a developing country with limited resources for the surgical management of exophytic brainstem gliomas.
Methods: This retrospective study included pediatric patients with brainstem (midbrain, pontine, or medullary) focal or diffuse gliomas whether low or high grade that had dorsal, ventral, or lateral exophytic component who were presented to our hospitals from January 2019 to January 2021. The patients’ data were collected, such as age, sex, preoperative and postoperative clinical condition, radiological data, surgical approach, extent of tumor removal, histopathology, follow-up period, and adjuvant therapy.
Results: A total of 23 patients were included in this study. The telovelar approach was used in 17 patients, the supracerebellar infratentorial approach in three patients, and the retrosigmoid, transcerebellar, and occipital transtentorial approach once for each patient. Twenty patients underwent near-total excision, and three underwent subtotal excision. Two-thirds of our cases (17 patients) were low-grade gliomas, with the remaining one-third comprising entirely of either anaplastic astrocytoma (five patients) or glioblastoma multiforme (one patient). The follow-up period of the patients extended from 3 months to 24 months.
Conclusion: Exophytic brainstem glioma surgery can result in good outcomes with minimal complications when near-total excision is attempted through a properly chosen approach and adherence to some surgical techniques and considerations.
Keywords: Brainstem glioma, Cerebrospinal fluid, Extent of resection, Intraoperative ultrasonography
INTRODUCTION
Brainstem glioma is the leading cause of morbidity and mortality among all central nervous system tumors, especially in childhood. Its management is one of the challenges faced by neurosurgeons, considering that it represents 20% of all pediatric brain tumors.[
MATERIALS AND METHODS
This retrospective study after approval from the Institutional Review Board included pediatric patients with exophytic brainstem glioma who were presented to the Cairo University Hospitals from January 2019 to January 2021. We included patients with brainstem (midbrain, pontine, or medullary) focal or diffuse gliomas whether low or high grade that had dorsal, ventral, or lateral exophytic component. The age and sex of the patients were registered. The preoperative assessment of the patients was performed, including the analysis of the complaint, full neurological examination, CT, and MRI brain with contrast, to evaluate the hydrocephalus, origin of the tumor within the brainstem, and direction of growth either through the fourth ventricle, CSF cistern, or other tissues and the tumoral epicenter. If hydrocephalus was present, it is managed through CSF diversion either by endoscopic third ventriculostomy (ETV) or ventriculoperitoneal shunt (VPS). Surgical approach was tailored for every case according to the origin of the tumor within brainstem, its direction of growth, targeting to go through a natural corridor allowing maximal tumor exposure, especially to its epicenter with minimal normal tissue injury, and retraction by trying to get benefit from gravity using proper position and drainage of CSF from surrounding cisterns or lumbar drain, using fine microneurosurgical skills in tissue handling. Ultrasonic aspirator was used as needed, especially with low settings at parts close to the brainstem, if the manual sucker could not remove the pathological tissue. The extent of resection (EOR) for every case was registered based on the intraoperative findings and the results of the intraoperative ultrasonography (US) and follow-up radiology, either total, near-total (leaving a thin sheet on the brainstem), subtotal (more than 50%), or partial (<50%). New neurological deficits and histopathology of the tumor were also registered. The follow-up of the cases was done periodically, determining the need of adjuvant therapy for every case individually depending on the EOR and histopathology.
RESULTS
From January 2019 to January 2021, 23 patients (12 males and 11 females) aged between 1 and 17 years were included in this study. The median age was 9 years at presentation and the mean age was 6.6 years. The most common clinical presentation included manifestations of increased intracranial pressure, unsteadiness, difficulty of swallowing, long tract deficits, and cranial nerve dysfunctions.
Hydrocephalus was associated with 18 out of the 23 patients (78%) preoperatively. Overall, 13 patients were treated with VPS and 5 with ETV, with no complications associated with these procedures and the resolution of hydrocephalus based on the follow-up images.
In 15 patients (65%), the tumor originated from the floor of the fourth ventricle. In one patient, the tumor originated from the pons. In 7 patients (30%), the tumor originated from the pontomesencephalic region. Directions of extensions of the exophytic parts are detailed in [
The telovelar approach was used for the excision of the tumor in 17 patients, the supracerebellar infratentorial approach in three patients, and the retrosigmoid, transcerebellar, and occipital transtentorial approaches once for each patient.
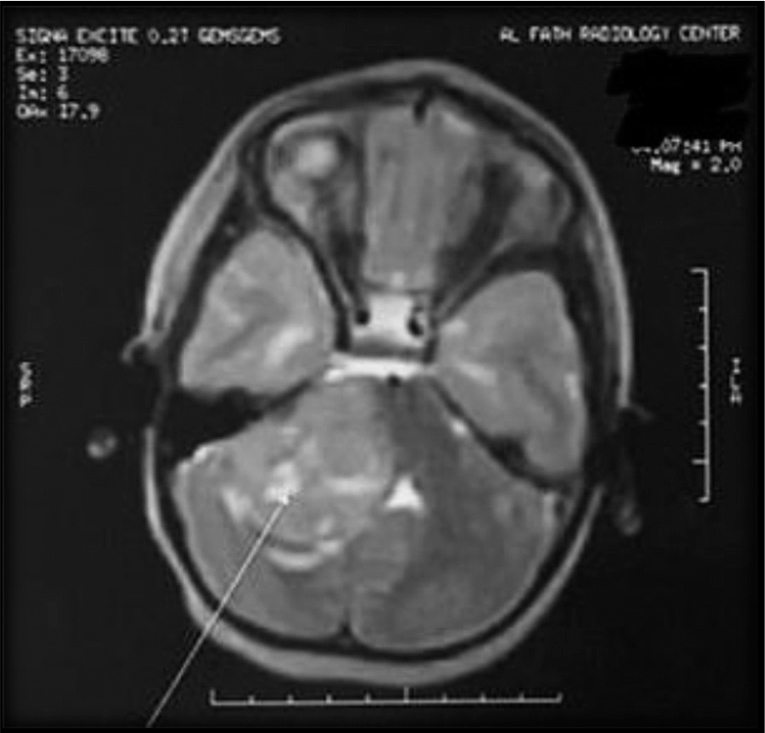
With regard to the EOR, 20 patients underwent near-total excision, leaving only a thin sheet of tumor over the brainstem. Three patients underwent subtotal excision of the tumor (more than 50%). In one of them, the tumor was extending from the pontomesencephalic region to invade the thalamus [
Two-thirds of all tumors (17 patients) were low-grade gliomas, with the remaining one-third (6 patients) comprising entirely of high-grade gliomas. The different surgical pathological results for the lesions are listed in [
In this study, 16 patients were followed without adjuvant therapy (underwent near-total excision with low-grade pathologies). One patient who underwent subtotal excision with low-grade pathology received chemotherapy. Four patients with high-grade pathologies received chemotherapy and radiotherapy. One patient with high-grade pathology was younger than 3 years old and received chemotherapy only with meticulous follow-up with a possibility to receive radiotherapy if the tumor progressed at an older age. One patient who underwent the subtotal removal of high-grade tumor died without receiving any adjuvant therapy. Furthermore, the follow-up period of patients extended from 3 months to 24 months (mean follow-up period was 11.8 months). During this period, no tumor progression was observed.
Postoperative morbidity and mortality
A case of perioperative death was observed in this series, wherein vascular injury occurred during the excision of the lesion. Surgery was aborted with difficult hemostasis, on which the patient was not fully recovered and could not be weaned from the ventilator with marked bulbar dysfunction. The patient then developed ventilator-dependent pneumonia and died after 3 weeks of surgery. Four patients developed marked bulbar dysfunction postoperatively and gradually improved over 2 months, in which three of them needed temporary tracheostomy. Other neurological morbidities were generally limited to the transient exacerbation of preoperative ataxia, dysmetria, nystagmus, and cranial nerve deficits (particularly sixth and seventh nerve paresis). In two patients, hemiparesis worsened immediately after surgery and started to improve after 2 weeks of surgery. No patients developed wound infection, meningitis, or CSF leak from the wound. No cases of mutism were reported. Furthermore, we did not observe any permanent complications in this series. [
DISCUSSION
The main goal of the surgical management of the exophytic brainstem gliomas is to reach the maximum EOR as it is one of the most significant prognostic factors affecting the prognosis of pediatric patients with exophytic brainstem glioma.[
Choosing the best suitable approach that gives the proper maximum and safe tumor exposure was one of the main considerations we focused on. We aim to select whenever possible the shortest approach through a natural existing corridor wherein the tumor was extended besides assuming the epicenter of the lesion in the planning of the approach. Hence, among the 15 cases of tumors originating mainly from the floor of the fourth ventricle (68% of cases), the telovelar approach was used in 14 cases to fully expose the tumor wherein the opening of the tela choroidea and inferior medullary velum and the retraction of the tonsils laterally were sufficient to resect the tumors in nine cases, while lateral extensive splitting of the cerebellomedullary fissure allowed a maximum exposure in the other five cases with lateral extension [
Figure 2:
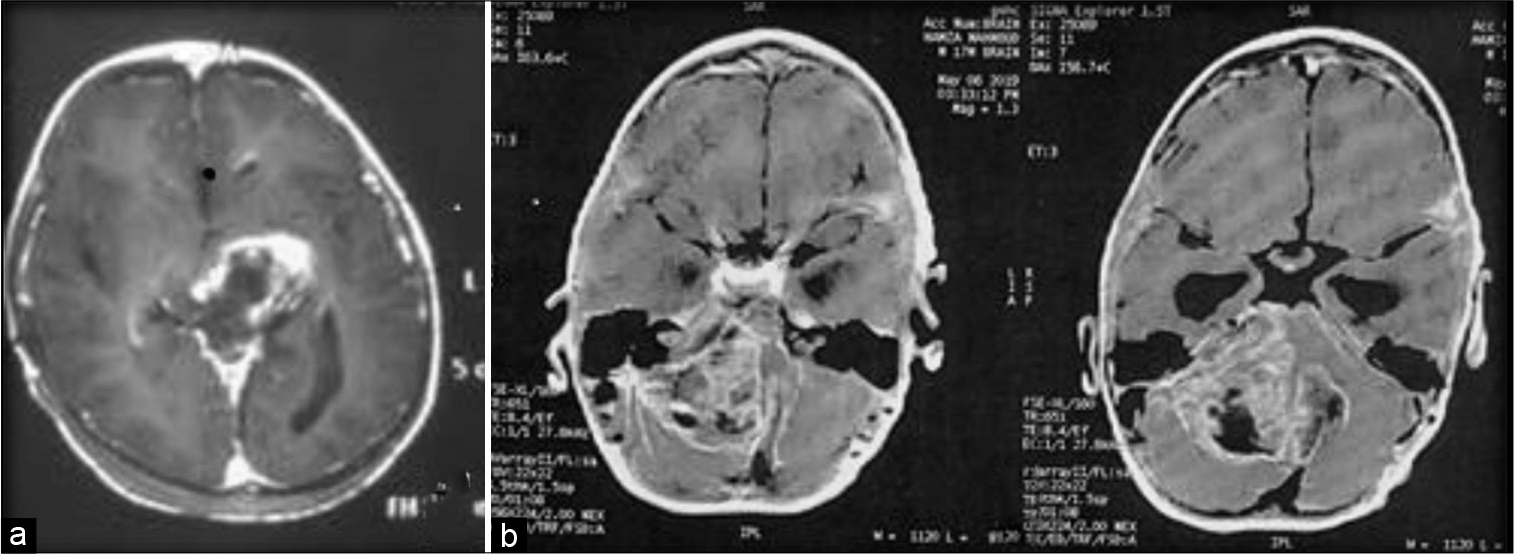
(a) Preoperative MRI showing a tumor originating from the floor of the fourth ventricle filling the fourth ventricle and the lateral extension filling the cerebellomedullary cistern. (b) Postoperative CT after near-total excision through the telovelar approach with extensive opening of the cerebellomedullary fissure.
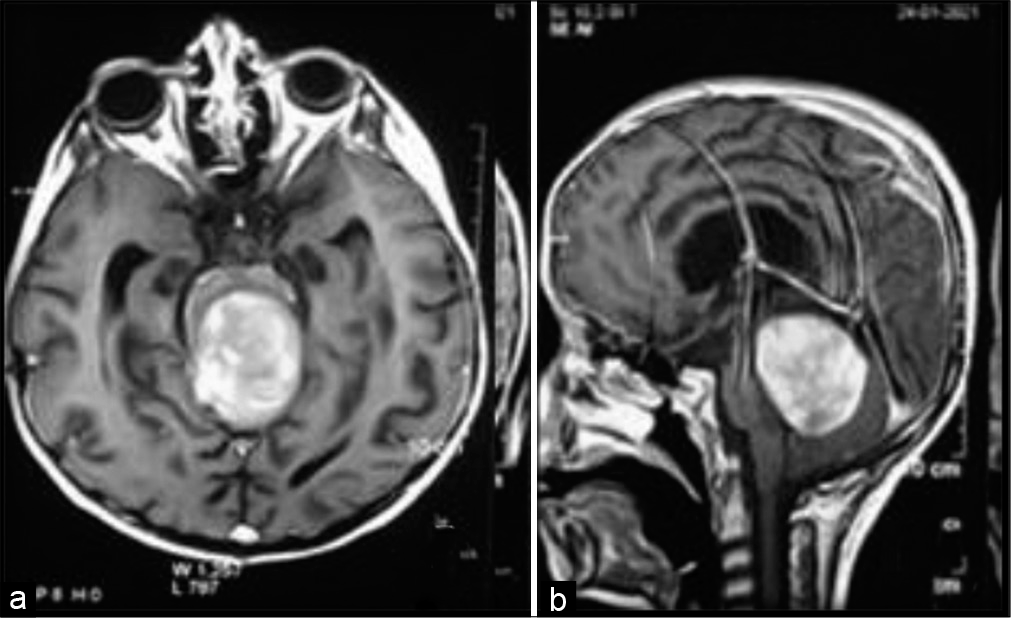
In seven patients, the tumors originated from the pontomesencephalic region. In three of them, the tumors mainly grew in an inferior rather than a posterior direction where its epicenter was located within the fourth ventricle, as the tumor attained a large size filling it, so the decision to attack these tumors through the telovelar approach was made [
Similar to the present study, Cavalheiro et al. described the surgical approaches to the brainstem tumors according to their origin and direction of growth and divided the brainstem into seven portions: midbrain (anterior, central, and posterior), pons (ventral and dorsal), and medulla (ventral and dorsal).[
One of the important tools to be considered in the surgical excision of exophytic brainstem gliomas is the use of Cavitron Ultrasonic Surgical Aspirator (CUSA), especially for the parts attached to the brainstem where safe resection can be achieved with less rocking and manipulations on the brainstem and eloquent neural tissues, less dissection, and less coagulation. Moreover, CUSA is safer than the other traditional tissue excision techniques in avoiding injury to nearby high-strength tissues, such as veins and neural tissue, during the resection of low-strength tissues. Working cautiously tangentially to the brainstem using low ultrasonic settings and moderate suction forces will result in better outcomes. The technological advancement provided surgeons with smaller bayoneted hands that allowed the CUSA to reach deep and far corners to increase the EOR.[
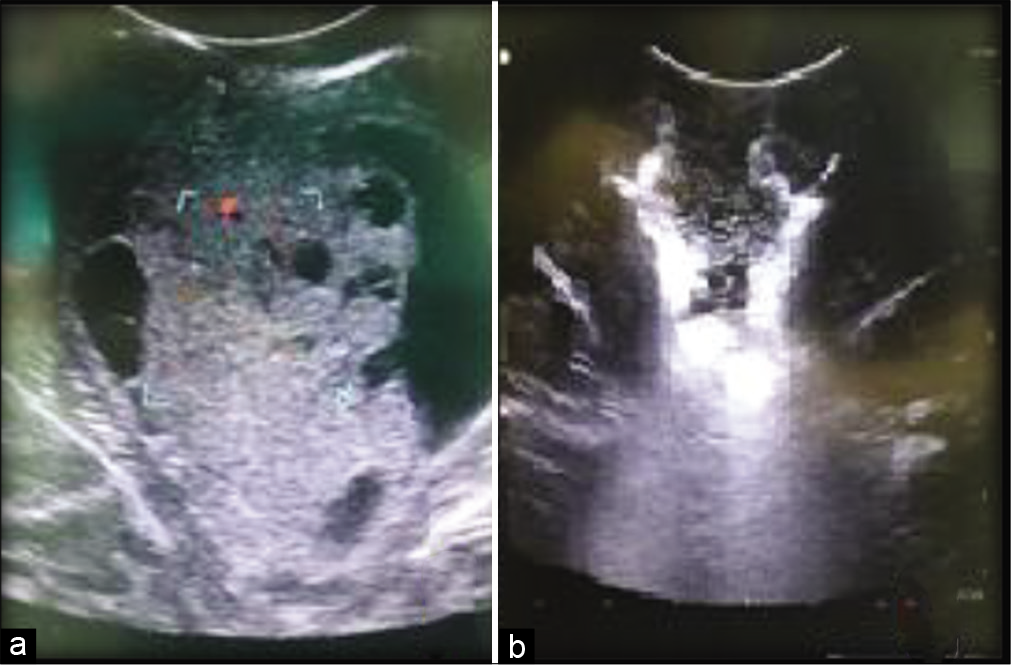
Many studies concluded that intraoperative US is more effective than the other types of image-guided navigation systems, such as MR-based neuronavigation and intraoperative MRI, especially when working with limited resources, as US is cheaper and accurate and could be used repetitively before starting tumor excision and frequently during excision to have an estimate about the proximity of the brainstem and to ensure maximal possible resection [
Leaving a sheet of tumor on the brainstem and near-total resection (as we did in 20 patients in our study) is another surgical strategy that can be followed when managing exophytic brainstem gliomas, especially if the tumor is seen violating the brainstem or when neuromonitoring is not available, as the viability of the neural structures of the brainstem should be respected to avoid extensive perioperative morbidity (e.g. intraoperative hemodynamic instability and mechanical ventilator dependency) or mortality, even if the radical tumor excision of nondiffuse low-grade brainstem glioma is curative and radical excision of high-grade nondiffuse brainstem glioma results in better survival.[
Some other considerations that should be kept in mind include the suitable positioning of the patient (trying to benefit from gravity), CSF drainage from the surrounding cisterns or lumbar drain, maximal arachnoid untethering, and the use of fine microneurosurgical skills in tissue handling to keep normal tissue moisture by continuous irrigation to guard against the thermal effect of microscope lights and vaporization of tissue fluid with exposure.[
According to Jallo et al., exophytic tumors that grow laterally and ventrally into the brainstem are tumors with a higher grade compared with those exophytic tumors that project dorsally into the fourth ventricle. Midbrain gliomas behave in a very low-grade nature.[
CONCLUSION
Exophytic brainstem glioma surgery can result in good outcomes with minimal complications when near-total excision is performed through a properly chosen approach in accordance with some surgical techniques and considerations. Choosing the shortest approach through a natural corridor that provides no retraction, assuming the epicenter of the lesion in the planning of the approach, using CUSA and intraoperative US, CSF drainage, maximal arachnoid untethering, and maintaining a normal tissue moisture by continuous irrigation can help improve the surgical outcomes.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Bharati SJ, Pandia MP, Rath GP, Bithal PK, Dash HH, Dube SK. Perioperative problems in patients with brainstem tumors and their influence on patient outcome. J Anaesthesiol Clin Pharmacol. 2016. 32: 172-6
2. Bricolo A. Surgical management of intrinsic brain stem gliomas. Oper Tech Neurosurg. 2000. 3: 137-54
3. Cavalheiro S, Yagmurlu K, da Costa MD, Nicácio JM, Rodrigues TP, Chaddad-Neto F. Surgical approaches for brainstem tumors in pediatric patients. Childs Nerv Syst. 2015. 31: 1815-40
4. Deshmukh VR, Figueiredo EG, Deshmukh P, Crawford NR, Preul MC, Spetzler RF. Quantification and comparison of telovelar and transvermian approaches to the fourth ventricle. Oper Neurosurg. 2006. 58: ONS-202-6
5. Ghodsi M, Mortazavi A, Shahjouei S, Hanaei S, Esmaeili A, Nejat F. Exophytic glioma of the medulla: Presentation, management and outcome. Pediatr Neurosurg. 2013. 49: 195-201
6. Guillamo JS, Monjour A, Taillandier L, Devaux B, Varlet P, Haie-Meder C. Brainstem gliomas in adults: Prognostic factors and classification. Brain. 2001. 124: 2528-39
7. Jallo GI, Biser-Rohrbaugh A, Freed D. Brainstem gliomas. Childs Nerv Syst. 2004. 20: 143-53
8. Jallo GI. CUSA EXcel ultrasonic aspiration system. Neurosurgery. 2001. 48: 695-7
9. Mussi AC, Rhoton AL. Telovelar approach to the fourth ventricle: Microsurgical anatomy. J Neurosurg. 2000. 92: 812-23
10. Pollack IF, Hoffman HJ, Humphreys RP, Becker L. The long-term outcome after surgical treatment of dorsally exophytic brain-stem gliomas. J Neurosurg. 1993. 78: 859-63
11. Preston JK, Masciopinto J, Salamat MS, Badie B. Tumour cell dispersion by the ultrasonic aspirator during brain tumour resection. Br J Neurosurg. 1999. 13: 486-9
12. Reyes-Botero G, Mokhtari K, Martin-Duverneuil N, Delattre JY, Laigle-Donadey F. Adult brainstem gliomas. Oncologist. 2012. 17: 388-97
13. Roujeau T, di Rocco F, Dufour C, Bourdeaut F, Puget S, Rose CS. Shall we treat hydrocephalus associated to brain stem glioma in children?. Childs Nerv Syst. 2011. 27: 1735-9
14. Tanriover N, Ulm AJ, Rhoton AL, Yasuda A. Comparison of the transvermian and telovelar approaches to the fourth ventricle. J Neurosurg. 2004. 101: 484-98
15. Teo C, Siu TL. Radical resection of focal brainstem gliomas: Is it worth doing?. Childs Nerv Syst. 2008. 24: 1307-14
16. Tomasello F, Conti A, Cardali S, La Torre D, Angileri FF. Telovelar approach to fourth ventricle tumors: Highlights and limitations. World Neurosurg. 2015. 83: 1141-7
17. Tuncer C, Polat O, Duru S. The use of tissue-selective ultrasonic aspirators in the surgical treatment of brain and spinal cord tumors. Duzce Med J. 2019. 21: 94-7
18. Upadhyay PK, Tiwary G. Real-time intraoperative ultrasonography in the surgical resection of brain lesions: A cheap, effective, and quick alternative. J Neurosci Rural Pract. 2010. 1: 127-8
19. Vougioukas VI, Omran H, Gläsker S, Velthoven V. Far lateral supracerebellar infratentorial approach for the treatment of upper brainstem gliomas: Clinical experience with pediatric patients. Childs Nerv Syst. 2005. 21: 1037-41
20. Yeole U, Singh V, Mishra A, Shaikh S, Shetty P, Moiyadi A. Navigated intraoperative ultrasonography for brain tumors: A pictorial essay on the technique, its utility, and its benefits in neuro-oncology. Ultrasonography. 2020. 39: 394-406
21. Yeole U, Singh V, Mishra A, Shaikh S, Shetty P, Moiyadi A. Navigated intraoperative ultrasonography for brain tumors: A pictorial essay on the technique, its utility, and its benefits in neuro-oncology. Ultrasonography. 2020. 39: 394-406