- Department of Neurology, Functional Neurosurgery Division, School of Medicine, University of São Paulo, São Paulo, Brazil
- Department of Neurology, Movement Disorders Center, School of Medicine, University of São Paulo, São Paulo, Brazil
- Department of Neurology, Pain Center, Hospital das Clínicas FMUSP, São Paulo, Brazil.
Correspondence Address:
Juliete Melo Diniz, Department of Neurology, Functional Neurosurgery Division, School of Medicine, University of São Paulo, São Paulo, Brazil.
DOI:10.25259/SNI_338_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Juliete Melo Diniz1, Rubens Gisbert Cury2, Ricardo Ferrareto Iglesio1, Guilherme Alves Lepski1, Carina Cura França2, Egberto Reis Barbosa2, Daniel Ciampi de Andrade3, Manoel Jacobsen Teixeira1, Kleber Paiva Duarte1. Dentate nucleus deep brain stimulation: Technical note of a novel methodology assisted by tractography. 09-Aug-2021;12:400
How to cite this URL: Juliete Melo Diniz1, Rubens Gisbert Cury2, Ricardo Ferrareto Iglesio1, Guilherme Alves Lepski1, Carina Cura França2, Egberto Reis Barbosa2, Daniel Ciampi de Andrade3, Manoel Jacobsen Teixeira1, Kleber Paiva Duarte1. Dentate nucleus deep brain stimulation: Technical note of a novel methodology assisted by tractography. 09-Aug-2021;12:400. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11028
Abstract
Background: The cerebellum has emerged as an attractive and promising target for neuromodulation in movement disorders due to its vast connection with important cortical and subcortical areas. Here, we describe a novel technique of deep brain stimulation (DBS) of the dentate nucleus (DN) aided by tractography.
Methods: Since 2015, patients with movement disorders including dystonia, ataxia, and tremor have been treated with DN DBS. The cerebellar target was initially localized using coordinates measured from the fastigial point. The target was adjusted with direct visualization of the DN in the susceptibility-weighted imaging and T2 sequences of the MRI and finally refined based on the reconstruction of the dentatorubrothalamic tract (DRTT).
Results: Three patients were treated with this technique. The final target was located in the anterior portion of DN in close proximity to the DRTT, with the tip of the lead on the white matter and the remaining contacts on the DN. Clinical outcomes were variable and overall positive, with no major side effect.
Conclusion: Targeting the DN based on tractography of the DRTT seems to be feasible and safe. Larger studies will be necessary to support our preliminary findings.
Keywords: Deep brain stimulation, Dentate nucleus, Dentatorubrothalamic tract, Tractography
INTRODUCTION
A key anatomical feature of the cerebellum is the deep cerebellar nuclei.[
The DN has connections with different cortical and subcortical areas, making it a strategic target.[
The use of the DN as a target for the treatment of different movement disorders dates back to 1935, when DelmasMarsalet and Von Bogaert performed the first ablation of the DN in a Parkinsonian patient.[
Recently, cerebellar modulation has reemerged as a promising therapy in the movement disorders field. Noninvasive stimulation of the DN showed encouraging results. Despite this, there are few centers with experience in deep brain stimulation (DBS) of this target and its effect has been evaluated only in single cases. Thus far, small improvements were noted, DN DBS was effective for treating ataxia in SCA type 3, cerebellar stroke, dystonia, and essential tremor.[
References in the literature to electrode placement in DN use atlas coordinates rather than direct MRI visualization, probably a result of the difficulty of seeing the DN on conventional imaging. This fact may have limited the interest for the target.[
With the evolution of technique and image quality, the ability to identify the DN has improved.[
In this paper, we detailed for the 1st time the surgical technique used by our group to perform DN DBS based on specific MRI sequences associated with tractography of the dentatorubrothalamic tract (DRTT).
METHODS
Between July 2015 and September 2020, three patients with refractory movement disorders including tremor, dystonia, ataxia, and stroke were treated with unilateral or bilateral DN DBS at the University of São Paulo, Brazil. Data were obtained from patients included in a research protocol approved by the ethics committee of the mentioned institution. Epidemiological characteristics and etiological diagnosis are summarized in [
Before surgery, contrast-enhanced volumetric T1, T2, susceptibility-weighted imaging (SWI), and fluid-attenuated inversion recovery, MRI scans were obtained in axial sections with a 1.5 T Siemens Espree scanner (Siemens, Munich, Germany). Diffusion tensor imaging (DTI) was acquired with the following specifications: single-shot, 2-dimensional; echo-planar imaging; repetition time (TR), 6700 ms; echo time (TE), 95 ms; diffusion values, b=0 s/mm2 and b=1000 s mm2; diffusion directions, 20; slice count, 50; voxel size, 2.6×2.6×2.7 mm3; and acquisition time, 9:07 min.
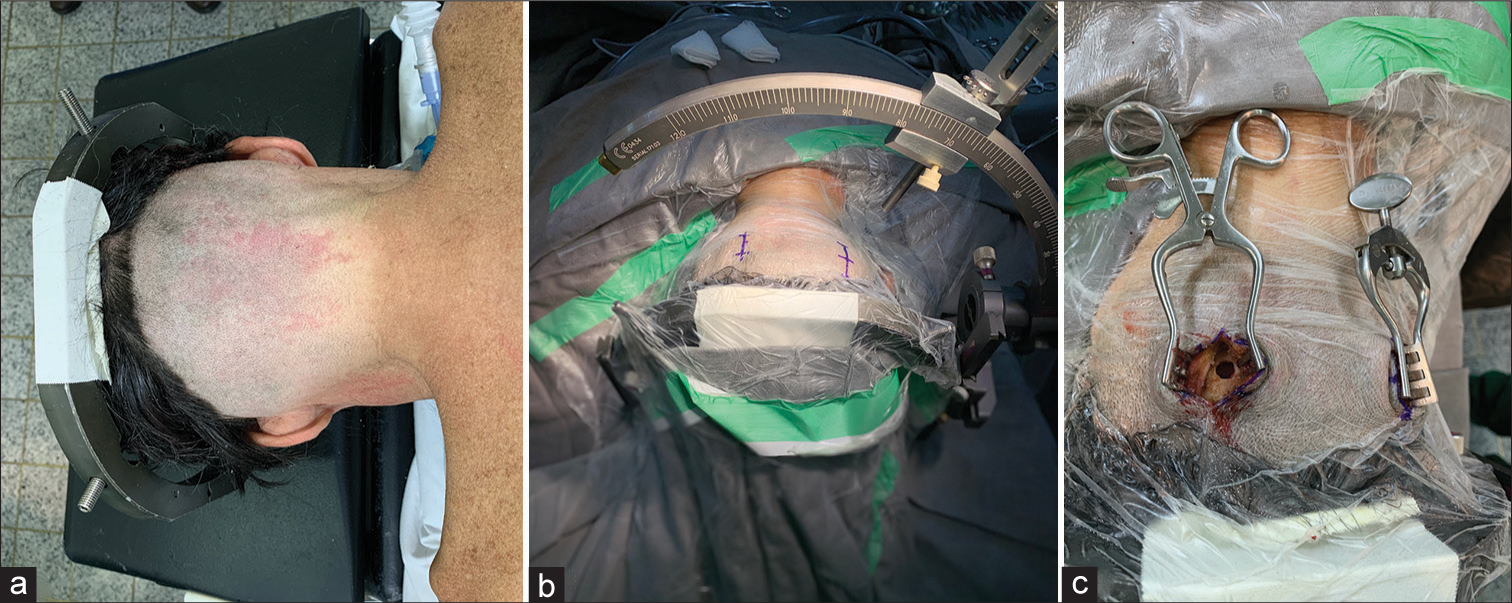
On the day of surgery, a stereotactic frame (Aimsystem, Micromar, Brazil) was placed on the patient’s head under local anesthesia and stereotactic contrast-enhanced computed tomography scan was performed.
Registration of image sets and target planning was subsequently performed using the MNPS Software (MEVIS neurosurgery planning system, MEVIS, Brazil) and deterministic tractography was performed using DSI studio software (
Tractography, based on diffusion-weighted imaging (DWI), is a technique with great potential to characterize the in vivo anatomical position and integrity of white matter tracts (Basser et al., 2000; Behrens et al., 2003; Mori et al., 1999). Tractography has proven its worth in neuroscience as well as in neurology and neurosurgery (Bick et al., 2012; Dimou et al., 2013; Potgieser et al., 2014). It is an invaluable tool in investigating structure–function relationship.
Target planning
Coordinates described by Weigel et al.[
The target position was then adjusted using direct visualization of the DN in the SWI and T2 sequences previously to coregistration to stereotomography and T1 contrast-enhanced volumetric MRI. The target was refined to be located in the anterior and upper third of the DN, region related to motor function.
In the next step, adjustment was performed to keep the target within the DN, being as close and parallel as possible to the DRTT fibers [
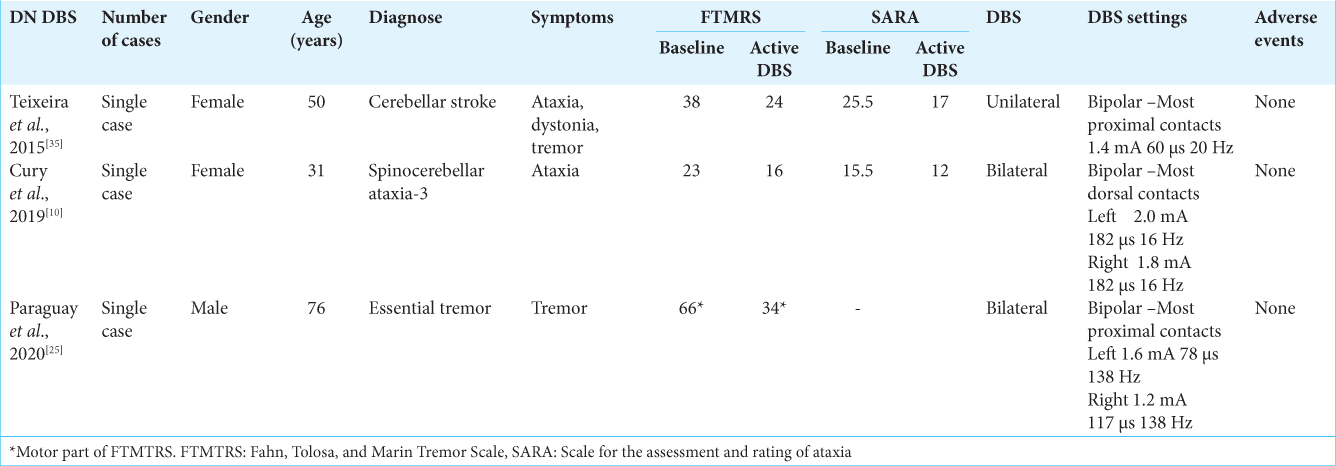
Figure 1:
Planning target. (a) Axial T2 MRI showing bilateral dentate nucleus with electrodes position simulated. (b) Axial T2 MRI with regions of interest in dentate nucleus (brown and yellow volumes) and VIM (red and blue volumes). (c) Axial T2 MRI showing the planned position of the electrodes in the dentate nucleus and the relation with DRTT in the left side.
The entry points were planned to provide a safe trajectory through suboccipital bone, avoiding proximity to the venous sinuses and vessels visualized on the contrasted images.
Procedure
After planning the trajectory, the patients underwent a surgical procedure to implant the cerebellar electrodes. All cases were operated under general anesthesia and were positioned in prone with the skull fixed in flexion to facilitate access to the posterior fossa [
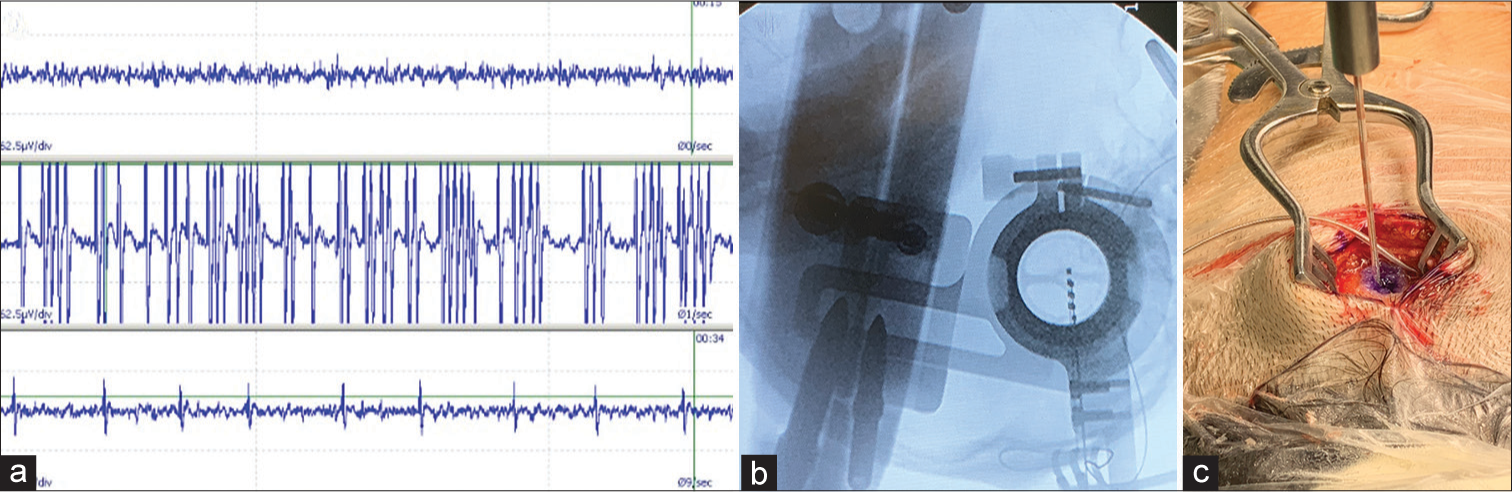
Microelectrodes of 130 mm length, 0.5 mm diameter, and 2 mm uninsulated recording tips were used (Inomed, Emmendingen, Germany). The record started 10 mm before the target and was performed up to 3 mm beyond the planned target point. The DN was identified on the MER by a region with intense electrical activity and continuous bursts of big spikes.[
Quadripolar leads with 1.5 mm active contacts, 3 mm active tip, and 1.5 spacing (model 6145, Abbott, Memphis, USA) were implanted on the planned targets adjusted by the MER findings, ensuring the tip of the lead is located on the white matter and the remaining contacts on the DN. The electrodes location was confirmed intraoperatively with X-rays and then fixed in place with cyanoacrylate glue [
In a subsequent procedure, the frame was removed, the patient was repositioned in supine position, electrodes were internalized and connected to a programmable implantable pulse generator (Libra XP, Abbott, Memphis, USA). The electrodes remained turned off after the procedure. Determination of optimal stimulation parameters for each electrode was based on a detailed test – stimulation protocol implemented on the postoperative follow-up.
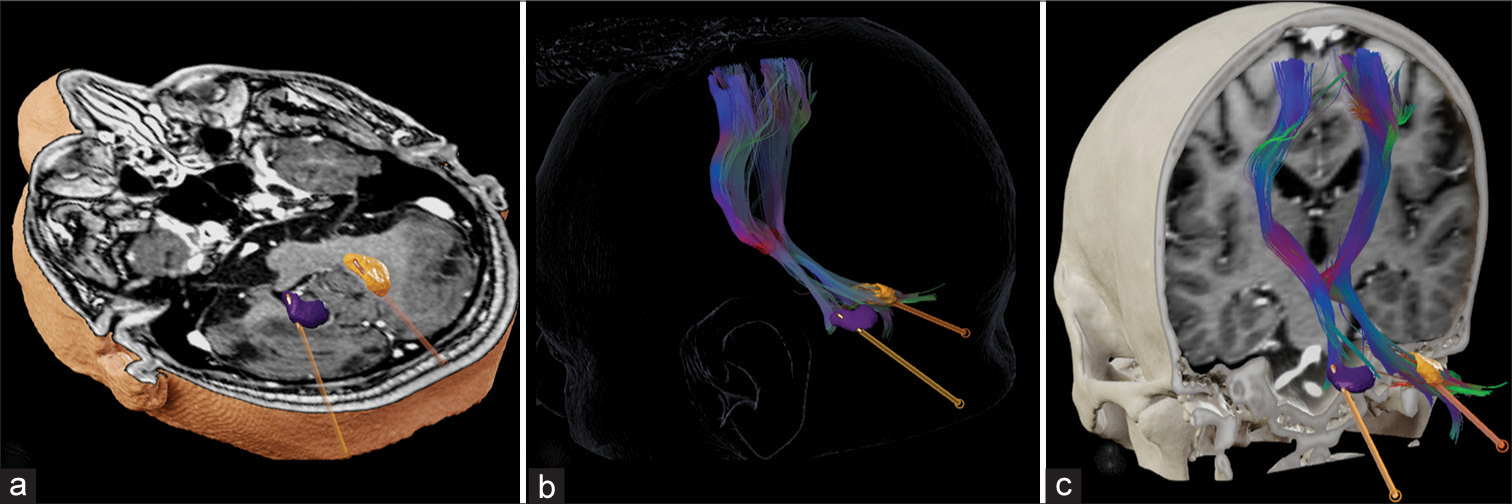
Accurate lead placement was confirmed with a postoperative CT scan registered to preoperative target planning using Elements Software (Brainlab AG, Germany) [
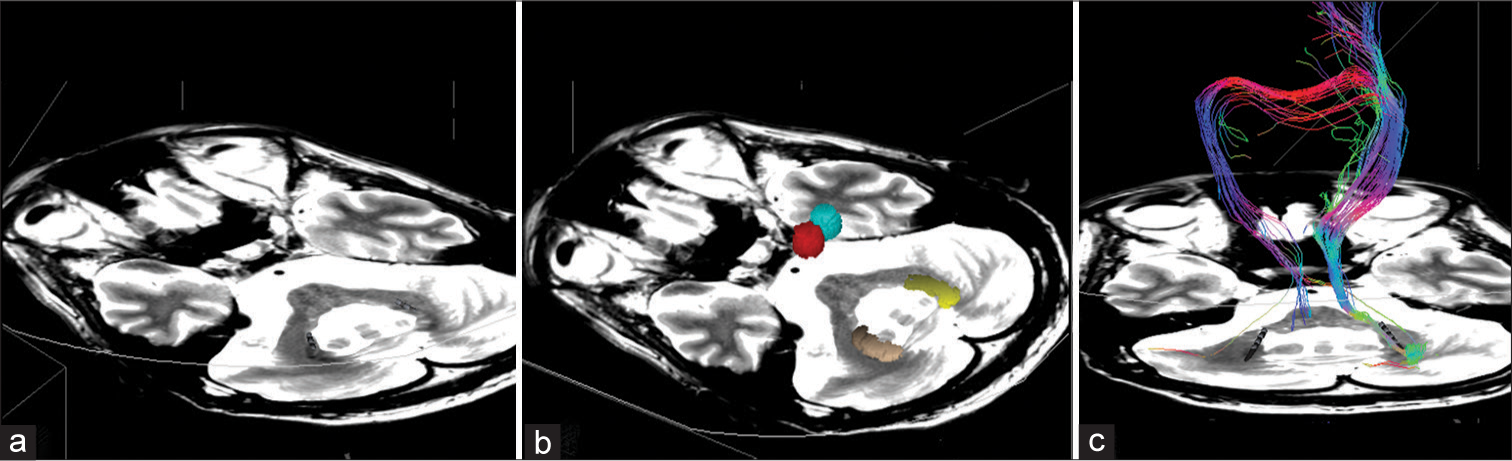
Figure 4:
Postoperative image control. (a) Axial T1 gadolinium-weighted MRI reconstruction showing the adequate position of the electrodes in the dentate nucleus. (b) Three-dimensional reconstruction of DBS electrodes in bilateral dentate nucleus (purple and orange volumes) showing close relationship with DRTT (c) Coronal T1 gadolinium-weighted MRI reconstruction to evidence the bilateral electrodes, dentate nucleus, and DRTT. The 3D reconstructions and tractography were performed with Elements Software (Brainlab AG, Germany).
Clinical outcomes
The clinical outcomes from the cohort have been published elsewhere[
DISCUSSION
DBS is a neurosurgical technique well established to treat different disorders.[
The DN is the largest of the deep cerebellar nuclei and plays a critical role in movement, cognition, autonomic functions, and behavior.[
The use of the DN as target to treat movement disorders is not new. In 1935, Delmas-Marsalet performed the first surgical procedure on the DN in humans to treat a patient with Parkinson’s disease.[
The DN has connections with different cortical and subcortical areas, making it a strategic target. It receives afferent fibers from the premotor cortex, supplementary motor cortex, and also from the spinocerebellar tract.[
Even with the current recognition of the role of the DN in movement disorders, studies evaluating the effects of cerebellar modulation are still rare. Horisawa et al.[
The rationale related to the modulation of DN and DRTT is based in the fact that cerebellar lesions in some cases may decrease intracortical inhibition in the contralateral motor cortex, culminating in interhemispheric asymmetry of the cortical excitability, which could contribute to the motor impairment. Modulating the healthy DN and DRTT could increase contralateral intracortical facilitation, restoring cortical excitability symmetry.[
In the cases of ischemic cortical stroke, deafferentation and reduced activity of the cerebellum would happen, resulting in decreased excitatory output from the cerebellum to the affected cortex through the reciprocal dentatothalamocortical pathway. This phenomenon is a critical component of the rationale underlying the selection of the DRTT at the DN as a target.[
Our group has been one of the few to use DN to treat different pathologies. One of the limitations for targeting the DN is the difficulty for its proper identification using conventional imaging techniques. References in the literature to electrode placement describe the use of atlas coordinates rather than direct MRI targeting and it could lead to inaccurate electrode placement. Although the DN is properly seen on SWI, the field distortion limits its use. We chose to merge volumetric images of T1 and T2, as well as T2 and SWI, aiming targeting based on the direct visualization of the DN.
Despite the possibility of adjusting the stimulation with DBS in relation to dentatotomy, another critical point of the procedure is to achieve adequate location of the contacts in relation to the DN. In our surgical technique, we aim at the dorsal and medial anterior region of the DN that appears to be more related to motor function. Brown et al.[
The DRTT is a bundle that originates from the DN, runs in the SCP, and then mainly decussates to the contralateral RN to ascend to the thalamus and finally to the cortex.[
It is accepted that DBS can have influence over widespread areas of the brain, which have implications beyond the inhibition of a local gray matter structure. One of the mechanisms of DBS is to modulate circuit activity and fiber pathways in the vicinity of the electrodes. Considering this, the use of tractography during targeting has been advocated in some applications of this technique.[
King et al.[
In this paper, we systematically describe the technique used to target the DN assisted by tractography. The authors are not aware of the use of a similar technique. As limitations of this work, we have the restricted number of cases, the use of DTI with 50 slices and deterministic tractography. A detailed discussion about the merits and limitations of tractography is beyond the scope of this paper. Besides, the best target into the cerebellum DN or the DRTT or both has yet to be defined. Larger studies will be necessary to support our preliminary findings.
CONCLUSION
The interest in the neuromodulation of the DN for the treatment of movement disorders has reemerged with better knowledge of its multiple connections with cortical and subcortical areas. Besides that, novel data have suggested that much of the benefit achieved from DBS is due to indirect stimulation of white matter pathways in close proximity to the active contact of the stimulating electrode. Considering this, we presented a feasible methodology to targeting the DN using the direct visualization with the help of specific sequences of MRI and based on the relationship with the DRTT.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Abdulbaki A, Kaufmann J, Galazky I, Buentjen L, Voges J. Neuromodulation of the subthalamic nucleus in Parkinson’s disease: The effect of fiber tract stimulation on tremor control. Acta Neurochir (Wien). 2021. 163: 185-95
2. Akakin A, Peris-Celda M, Kilic T, Seker A, Gutierrez-Martin A, Rhoton A. The dentate nucleus and its projection system in the human cerebellum: The dentate nucleus microsurgical anatomical study. Neurosurgery. 2014. 74: 401-25
3. Bond KM, Brinjikji W, Eckel LJ, Kallmes DF, McDonald RJ, Carr CM. Dentate update: Imaging features of entities that affect the dentate nucleus. AJNR Am J Neuroradiol. 2017. 38: 1467-74
4. Brown EG, Bledsoe IO, Luthra NS, Miocinovic S, Starr PA, Ostrem JL. Cerebellar deep brain stimulation for acquired hemidystonia. Mov Disord Clin Pract. 2020. 7: 188-93
5. Chan HH, Wathen CA, Mathews ND, Hogue O, Modic JP, Kundalia R. Lateral cerebellar nucleus stimulation promotes motor recovery and suppresses neuroinflammation in a fluid percussion injury rodent model. Brain Stimul. 2018. 11: 1356-67
6. Coenen VA, Allert N, Mädler B. A role of diffusion tensor imaging fiber tracking in deep brain stimulation surgery: DBS of the dentato-rubro-thalamic tract (drt) for the treatment of therapy-refractory tremor. Acta Neurochir (Wien). 2011. 153: 1579-85
7. Coenen VA, Allert N, Paus S, Kronenbürger M, Urbach H, Mädler B. Modulation of the cerebello-thalamo-cortical network in thalamic deep brain stimulation for tremor: A diffusion tensor imaging study. Neurosurgery. 2014. 75: 657-69
8. Cooper IS, Amin I, Upton A, Riklan M, Watkins S, McLellan L. Safety and efficacy of chronic cerebellar stimulation. Appl Neurophysiol. 1977. 40: 124-34
9. Cury RG, França C, Barbosa ER, Galhardoni R, Lepski G, Teixeira MJ. Dentate nucleus stimulation in a patient with cerebellar ataxia and tremor after cerebellar stroke: A long-term follow-up. Parkinsonism Relat Disord. 2019. 60: 173-5
10. Cury RG, França C, Silva V, Barbosa ER, Capato TT, Lepski G. Effects of dentate nucleus stimulation in spinocerebellar ataxia Type 3. Parkinsonism Relat Disord. 2019. 69: 91-3
11. Cury RG, Teixeira MJ, Galhardoni R, Barboza VR, Alho E, Seixas CM. Neuronavigation-guided transcranial magnetic stimulation of the dentate nucleus improves cerebellar ataxia: A sham-controlled, double-blind n=1 study. Parkinsonism Relat Disord. 2015. 21: 999-1001
12. Delmas-Marsalet P, Von bogaert L. On a case of continuous rhythmic myoclonus determined by surgery on the brainstem. Journal Neurol. 1935. 64: 728-40
13. Deoni SC, Catani M. Visualization of the deep cerebellar nuclei using quantitative T1 and rho magnetic resonance imaging at 3 Tesla. Neuroimage. 2007. 37: 1260-6
14. França C, de Andrade DC, Silva V, Galhardoni R, Barbosa ER, Teixeira MJ. Effects of cerebellar transcranial magnetic stimulation on ataxias: A randomized trial. Parkinsonism Relat Disord. 2020. 80: 1-6
15. Gaytán-Tocavén L, Olvera-Cortés ME. Bilateral lesion of the cerebellar-dentate nucleus impairs egocentric sequential learning but not egocentric navigation in the rat. Neurobiol Learn Mem. 2004. 82: 120-7
16. Gortvai P, Teruchkin S. The position and extent of the human dentate nucleus. Acta Neurochir (Wien). 1974. p. 101-10
17. Henderson JM. “Connectomic surgery”: Diffusion tensor imaging (DTI) tractography as a targeting modality for surgical modulation of neural networks. Front Integr Neurosci. 2012. 6: 15
18. Horisawa S, Arai T, Suzuki N, Kawamata T, Taira T. The striking effects of deep cerebellar stimulation on generalized fixed dystonia: Case report. J Neurosurg. 2019. 132: 712-6
19. Ji Q, Edwards A, Glass JO, Brinkman TM, Patay Z, Reddick WE. Measurement of projections between dentate nucleus and contralateral frontal cortex in human brain via diffusion tensor tractography. Cerebellum. 2019. 18: 761-9
20. Küper M, Thürling M, Maderwald S, Ladd ME, Timmann D. Structural and functional magnetic resonance imaging of the human cerebellar nuclei. Cerebellum. 2012. 11: 314-24
21. Meola A, Comert A, Yeh FC, Sivakanthan S, Fernandez-Miranda JC. The nondecussating pathway of the dentatorubrothalamic tract in humans: Human connectome-based tractographic study and microdissection validation. J Neurosurg. 2016. 124: 1406-12
22. Nádvorník P, Sramka M, Lisý L, Svicka I. Experiences with dentatotomy. Confin Neurol. 1972. 34: 320-4
23. Nicholson CL, Coubes P, Poulen G. Dentate nucleus as target for deep brain stimulation in dystono-dyskinetic syndromes. Neurochirurgie. 2020. 66: 258-65
24. Nowacki A, Schlaier J, Debove I, Pollo C. Validation of diffusion tensor imaging tractography to visualize the dentatorubrothalamic tract for surgical planning. J Neurosurg. 2018. 130: 99-108
25. Paraguay IB, França C, Duarte KP, Diniz JM, Galhardoni R, Silva V. Dentate nucleus stimulation for essential tremor. Parkinsonism Relat Disord. 2020. 82: 121-2
26. Petersen KJ, Reid JA, Chakravorti S, Juttukonda MR, Franco G, Trujillo P. Structural and functional connectivity of the nondecussating dentato-rubro-thalamic tract. Neuroimage. 2018. 176: 364-71
27. Ramos A, Chaddad-Neto F, Dória-Netto HL, de CamposFilho JM, Oliveira E. Cerebellar anatomy as applied to cerebellar microsurgical resections. Arq Neuropsiquiatr. 2012. 70: 441-6
28. Rodrigues NB, Mithani K, Meng Y, Lipsman N, Hamani C. The emerging role of tractography in deep brain stimulation: Basic principles and current applications. Brain Sci. 2018. 8: 23
29. See AA, King NK. Improving surgical outcome using diffusion tensor imaging techniques in deep brain stimulation. Front Surg. 2017. 4: 54
30. Slaughter DG, Nashold BS, Somjen GG. Electrical recording with micro-and macroelectrodes from the cerebellum of man. J Neurosurg. 1970. 33: 524-8
31. Steele CJ, Anwander A, Bazin PL, Trampel R, Schaefer A, Turner R. Human cerebellar sub-millimeter diffusion imaging reveals the motor and non-motor topography of the dentate nucleus. Cereb Cortex. 2017. 27: 4537-48
32. Sultan F, Hamodeh S, Baizer JS. The human dentate nucleus: A complex shape untangled. Neuroscience. 2010. 167: 965-8
33. Sweet JA, Walter BL, Gunalan K, Chaturvedi A, McIntyre CC, Miller JP. Fiber tractography of the axonal pathways linking the basal ganglia and cerebellum in Parkinson disease: Implications for targeting in deep brain stimulation. J Neurosurg. 2014. 120: 988-96
34. Swinkin E, Lizárraga KJ, Algarni M, Dominguez LG, Baarbé JK, Saravanamuttu J. A distinct EEG marker of celiac disease-related cortical myoclonus. Mov Disord. 2021. 36: 999-1005
35. Teixeira MJ, Cury RG, Galhardoni R, Barboza VR, Brunoni AR, Alho E. Deep brain stimulation of the dentate nucleus improves cerebellar ataxia after cerebellar stroke. Neurology. 2015. 85: 2075-6
36. Teixeira MJ, Schroeder HK, Lepski G. Evaluating cerebellar dentatotomy for the treatment of spasticity with or without dystonia. Br J Neurosurg. 2015. 29: 772-7
37. Wathen CA, Frizon LA, Maiti TK, Baker KB, Machado AG. Deep brain stimulation of the cerebellum for poststroke motor rehabilitation: From laboratory to clinical trial. Neurosurg Focus. 2018. 45: E13
38. Weigel K, Mundinger F. Computerized tomography-guided stereoctatic dentatotomy. Appl Neurophysiol. 1986. 49: 301-6