- Department of Neurosurgery, Parkash Hospital, Bhatinda, India.
- Department of Surgery, Government Medical College, Rajindra Hospital, Patiala, India.
- Department of Neurosurgery, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
- Department of Neurosurgery, Dayanand Medical College and Hospital, Ludhiana, Punjab, India.
Correspondence Address:
Saurabh Sharma, Department of Neurosurgery, Dayanand Medical College and Hospital, Ludhiana, Punjab, India.
DOI:10.25259/SNI_985_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Tarun Garg1, Raghav Sood2, Akshay Chaudhary3, Shivender Sobti4, Saurabh Sharma4, Ashwani K. Chaudhary4, Hanish Bansal4, Jagminder Singh4, Manish Sharma4. A prospective study to determine the risk factors associated with post traumatic seizures: A single institution experience. 21-Apr-2023;14:143
How to cite this URL: Tarun Garg1, Raghav Sood2, Akshay Chaudhary3, Shivender Sobti4, Saurabh Sharma4, Ashwani K. Chaudhary4, Hanish Bansal4, Jagminder Singh4, Manish Sharma4. A prospective study to determine the risk factors associated with post traumatic seizures: A single institution experience. 21-Apr-2023;14:143. Available from: https://surgicalneurologyint.com/surgicalint-articles/12280/
Abstract
Background: Post traumatic seizures (PTS) are a known sequel of traumatic brain injury (TBI). Incidence of PTS is dependent on many factors including study design and characteristics of the study population. As incidence of TBI increases and death due to TBI decreases, more individuals will be at risk of developing and living with chronic complications. The objective of the present study was to determine the frequency and risk factors for PTS following TBI.
Methods: A prospective study was conducted on patients admitted with TBI from April 1, 2019, to May 31, 2020, to determine the frequency, time to event, and risk factors for PTS following TBI. We classified the severity of head injury using a standard criterion, into mild, moderate and severe injury. Follow-up of 3 months was undertaken for all patients. Variables include age, sex, trauma severity, Glasgow coma scale, onset of PTS, and neuroradiological finding.
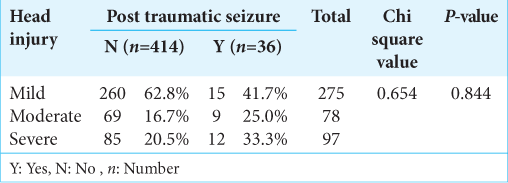
Results: We enrolled 450 post traumatic subjects, out of which 36 (8%) developed seizures. Of the total of 36 patients detected to have hemorrhagic contusion on computerized tomography scan, 12 patients developed seizures. We found that the independent risk factors associated with occurrence of PTS were frontal— temporal lobar contusion and severity of head injury. All these findings were statistically significant.
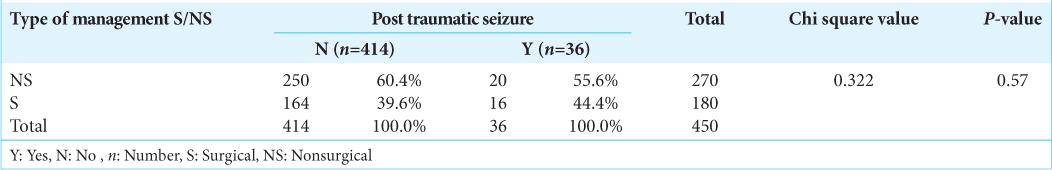
Conclusion: We found that the independent risk factors associated with occurrence of PTS were frontal-temporal lobar contusion and severity of head injury. Type of management (Operative vs. Non operative) does not affect the outcome of PTS.
Keywords: Antiepileptic, Head injury, Post traumatic seizures, Traumatic brain injury
INTRODUCTION
Traumatic brain injury (TBI) is a significant public health concern and is a major cause of morbidity and mortality, especially in patients <45 years.[
Individuals with more severe injury are particularly affected by chronic conditions associated with TBI. Among individuals who survive a severe TBI, disability rates are estimated as high as 77%.[
Despite decades of research, there are no effective pharmacological interventions to prevent post-traumatic seizures (PTS) that develop after 7 days post injury and, it does not appear that rates of PTS are decreasing.[
MATERIALS AND METHODS
A prospective study was conducted on patients admitted with TBI in a Tertiary care Centre in North India from April 1, 2019, to May 31, 2020. Prior ethics committee permission was obtained and written informed consent was taken from each participant before including them in the study.
Inclusion criterion
All patients who presented with acute TBI in the hospital were included in the study.
Exclusion criterion
(1) A preceding history of brain injury or major mental disorder, (2) Pregnancy, (3) Predisposition to seizures (such as a history of brain tumor or stroke), (4) Family history of seizures.
In this study, we used the classification of head injury severity as defined by Annegers et al.[
Statistical analysis
All statistical calculations were done using 17 Statistical Package for the Social Science 21 version statistical program for Microsoft Windows.
RESULTS
In our study, 450 post traumatic patients were included in the study. Thirty-six (8%) patients developed seizures. Patients with seizures were classified into three categories based on the GCS score. The group with GCS score of 3–6 had 15 patients developing seizures out of 275 patients. The group with GCS score of 7–12 had nine patients developing seizures out of 78 patients. The group with GCS score of 13–15 had 12 numbers of patients developing seizures out of 97 patients. Mean GCS of patients developing seizure is 12.28 ± 3.551 [
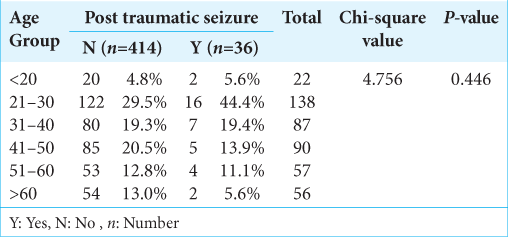
In our study, total number of patients were 450, 22 patients below the age group of 20 out of which two developed seizures (5.6%). One hundred and thirty-eight patients in the age group of 21–30 years, out of which 16 developed seizures (44.4%). Eighty-seven patients were in the age group of 31–40, out of which seven developed seizures (19.4%). Ninety patients were in the age group of 41–50, out of which five developed seizures (13.9). Fifty-seven patients were in the age group of 51–60, out of which four developed seizures (11.1%). Fifty-six patients had age more than 60 year, out of which two developed seizures (5.6%). Mean age of patients developing seizure is 36.14 ± 13.7 [
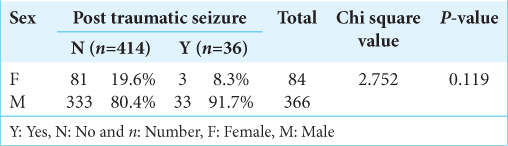
In our study, out of 450 patients, 366 were males and 84 were females. Thirty-three males and three females developed seizures [
Patients were also classified on the basis of computerized tomography (CT) findings. Of the total of 36 patients detected to have hemorrhagic contusion on CT scan, 12 patients developed seizures (33%). Of the total of eight patients diagnosed diffuse axonal injury, one patient developed seizures (12.5%). Of the total of 70 patients detected to have depressed fracture on CT scan, seven developed seizures (10%). Forty-seven patients who were diagnosed as a case of extradural hemorrhage on CT scan, one patient developed seizures (2%). Of the total of 61 patients diagnosed subarachnoid hemorrhage on CT scan, six patients developed seizures (9.8%), of the total of 131 patients detected to have subdural hemorrhage on CT scan, nine patients developed seizures (6.8%). Of the two patients diagnosed with intraparenchymal hemorrhage, none developed seizures. Of the total 450 patients, 270 were managed conservatively and 180 patients were operated. Twenty patients out of 270 patients had seizures.
One hundred and eighty patients were operated, 16 had seizures [
DISCUSSION
In our study, 450 patients with diagnosis of TBI were evaluated. The patients underwent detailed clinical evaluation and radiological investigation as and when indicated. In a study conducted by Lee et al., total of 4232 consecutively treated adult patients with mild closed head injury were examined. They ranged in age from 16 to 88 years mean (32.82 years); the male: Female ratio was 3.8:1. The most common causes of trauma are motorcycle accidents (53.78%) and pedestrian injury (28.89%). In 100 patients (2.36) who developed seizures within the1st week after head injury, the male: female was 7:1, the mean age was 29.74 years.[
In this study, subjects that were 21–30 years at age were more likely to develop PTS than subjects >30 years of age, but no significant difference between subjects 21–50 and those >50 years. Immaturity of the brain, which has been offered as the explanation for the greater susceptibility of children to early PTS, may explain the greater susceptibility of the younger age groups for seizures.
In our study, the overall incidence of PTS was found to be 8%. The incidence in normal civilian is 1.8–5%.[
Several reports (Annegers et al.; Angeleri et al.; Englander et al.) have shown cerebral contusion was indeed a risk factor.[
A possible explanation for this may be the following; Cortical neurons are injured following contusions and this would lead the iron from the blood deposited in the cortex; the deposited iron would lead to iron induced membrane peroxidation and would make the seizure spread to the subcortical areas (hippocampus mainly); the occurrence and evolution of the epileptiform activity in the subcortical structures would in parallel with that in the cortical focus.[
TBI severity measured by GCS score is also a risk factor in this study. The patients with lower GCS scores (more severe injury) have more risk to develop PTS.[
In our study, we used GCS score to measure the TBI severity and the results were consistent with the former studies. We found that the patients with GCS scores from 3 to 8 had the highest seizure frequency (19.9%) and the highest risk to develop PTS (4.103 times risk rate). However, some studies showed that the GCS score is not a risk factor. Englander et al.[
CONCLUSION
In this study of TBI patients, frontal-temporal lobar contusion and severity of TBI measured by initial GCS were significant in the multivariate model analysis. The reason PTS happens still needs to be investigated. Thorough follow-up of patients with PTS and antiepileptic therapy may attain rehabilitation goals.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Agrawal A, Timothy J, Pandit L, Manju M. Posttraumatic epilepsy: An overview. Clin Neurol Neurosurg. 2006. 108: 433-9
2. Andriessen TM, Horn J, Franschman G, van der Naalt J, Haitsma I, Jacobs B. Epidemiology, severity classification, and outcome of moderate and severe traumatic brain injury: A prospective multicenter study. J Neurotrauma. 2011. 28: 2019-31
3. Andriessen TM, Jacobs B, Vos PE. Clinical characteristics and pathophysiological mechanisms of focal and diffuse traumatic brain injury. J Cell Mol Med. 2010. 14: 2381-92
4. Angeleri F, Majkowski J, Cacchio G. Posttraumatic epilepsy risk factors: One-year prospective study after head injury. Epilepsia. 1999. 40: 1222-30
5. Annegers JF, Hauser WA, Coan SP, Rocca WA. A population based study of seizures after traumatic brain injuries. N Engl J Med. 1998. 338: 20-4
6. Brooks JC, Strauss DJ, Shavelle RM, Paculdo DR, Hammond FM, Harrison-Felix CL. Long-term disability and survival in traumatic brain injury: Results from the National Institute on Disability and Rehabilitation Research Model Systems. Arch Phys Med Rehabil. 2013. 94: 2203-9
7. Bruns J, Hauser WA. The epidemiology of traumatic brain injury: A review. Epilepsia. 2003. 44: 2-10
8. Dakin KA, Weaver DF. Mechanisms of post-traumatic seizures: A quantum pharmacological analysis of the molecular properties of an epileptogenic focus following iron-induced membrane peroxidation. Seizure. 1993. 2: 21-33
9. Englander J, Bushnik T, Duong TT, Cifu DX, Zafonte R, Wright J. Analyzing risk factors for late posttraumatic seizures: A prospective, multicenter investigation. Arch Phys Med Rehabil. 2003. 84: 365-73
10. Ferguson PL, Smith GM, Wannamaker BB. A population-based study of risk of epilepsy after hospitalization for traumatic brain injury. Epilepsia. 2010. 51: 891-8
11. Frey LC. Epidemiology of posttraumatic epilepsy: A critical review. Epilepsia. 2003. 44: 11-7
12. Haltiner AM, Temkin NR, Dikmen SS. Risk of seizure recurrence after the first late posttraumatic seizure. Arch Phys Med Rehabil. 1997. 78: 835-40
13. Harrison-Felix C, Pretz C, Hammond FM, Cuthbert JP, Bell J, Corrigan J. Life expectancy after inpatient rehabilitation for traumatic brain injury in the United States. J Neurotrauma. 2015. 32: 1893-901
14. Jennett B. Early traumatic epilepsy. Incidence and significance after non-missile injuries. Arch Neurol. 1974. 30: 394-8
15. Kharatishvili I, Nissinen JP, McIntosh TK, Pitkänen A. A model of posttraumatic epilepsy induced by lateral fluid-percussion brain injury in rats. Neuroscience. 2006. 140: 685-97
16. Kirmani BF, Robinson DM, Fonkem E, Graf K, Huang JH. Role of anticonvulsants in the management of posttraumatic epilepsy. Front Neurol. 2016. 7: 32
17. Lee ST, Lui TN, Wong CW, Yeh YS, Tzuan WC, Chen TY. Early seizures after severe closed head injury. Can J Neurol Sci. 1997. 24: 40-3
18. Lowenstein DH. Epilepsy after head injury: An overview. Epilepsia. 2009. 50: 4-9
19. , editors. Report to Congress on Traumatic Brain Injury in the United States: Epidemiology and Rehabilitation. Atlanta, GA: Centers for Disease Control and Prevention; 2014. p.
20. Rockhill CM, Jaffe K, Zhou C, Fan MY, Katon W, Fann JR. Health care costs associated with traumatic brain injury and psychiatric illness in adults. J Neurotrauma. 2012. 29: 1038-46
21. Salazar AM, Jabbari B, Vance SC, Grafman J, Amin D, Dillon JD. Epilepsy after penetrating head injury. I. Clinical correlates: A report of the Vietnam Head Injury Study. Neurology. 1985. 35: 1406-14
22. Schütze M, Dauch WA, Güttinger M, Hämpel-Christiansen M, Firsching R. Risk factors for posttraumatic fits and epilepsy. Zentralbl Neurochir. 1999. 60: 163-7
23. Sharma V, Babu PP, Singh A, Singh S, Singh R. Iron-induced experimental cortical seizures: Electroencephalographic mapping of seizure spread in the subcortical brain areas. Seizure. 2007. 16: 680-90
24. Temkin NR, Dikmen SS, Anderson GD, Wilensky AJ, Holmes MD, Cohen W. Valproate therapy for prevention of posttraumatic seizures: A randomized trial. J Neurosurg. 1999. 91: 593-600
25. Willmore LJ, Triggs WJ. Effect of phenytoin and corticosteroids on seizures and lipid peroxidation in experimental posttraumatic epilepsy. J Neurosurg. 1984. 60: 467-72