- Departments of Neurosurgery, Graduate School of Medical and Dental Sciences, Kagoshima University, 8-35-7 Sakuragaoka, Kagoshima,
- Departments of Pathology, Graduate School of Medical and Dental Sciences, Kagoshima University, 8-35-7 Sakuragaoka, Kagoshima,
- Department of Clinical Pathology, Gunma University Hospital, 3-39-22 Showa Machi, Maebashi, Gunma, Japan.
Correspondence Address:
Nayuta Higa
Departments of Neurosurgery, Graduate School of Medical and Dental Sciences, Kagoshima University, 8-35-7 Sakuragaoka, Kagoshima,
DOI:10.25259/SNI_97_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Muhammad Kamil, Nayuta Higa, Hajime Yonezawa, Shingo Fujio, Jun Sugata, Tomoko Takajo, Tsubasa Hiraki, Junko Hirato, Kazunori Arita, Koji Yoshimoto. A sellar neuroblastoma showing rapid growth and causing syndrome of inappropriate secretion of antidiuretic hormone: A case report. 27-Jun-2020;11:165
How to cite this URL: Muhammad Kamil, Nayuta Higa, Hajime Yonezawa, Shingo Fujio, Jun Sugata, Tomoko Takajo, Tsubasa Hiraki, Junko Hirato, Kazunori Arita, Koji Yoshimoto. A sellar neuroblastoma showing rapid growth and causing syndrome of inappropriate secretion of antidiuretic hormone: A case report. 27-Jun-2020;11:165. Available from: https://surgicalneurologyint.com/surgicalint-articles/10101/
Abstract
Background: Sellar neuroblastoma is a very rare entity. We report a rare case of arginine vasopressin (AVP)- producing sellar neuroblastoma presumed to have originated from the lower part of sellar turcica, which grew very rapidly.
Case Description: A 33-year-old woman was found to have a sellar lesion with a diameter of 18 mm invading into the bilateral cavernous sinus on magnetic resonance imaging (MRI) performed for dizziness. Six years later, when she visited the clinic due to bilateral visual disturbance, MRI showed a rapid growth of the tumor, with a maximal diameter of 56 mm at the current state, strongly compressing the optic nerve and chiasm. Transsphenoidal decompression of the optic chiasm revealed an intact pituitary gland on the top of the tumor. The tumor was composed of neoplastic cells that were immunohistochemically positive for neuronal markers and arginine vasopressin (AVP), but negative for all anterior pituitary hormones, glial fibrillary acidic protein, or thyroid transcription factor-1; these findings were suggestive of sellar neuroblastoma. She underwent 50-Gy radiation therapy, which has controlled the growth for the past 3 years.
Conclusion: Awareness of rare sellar neuroblastomas will allow the accumulation of clinicopathologic information that may facilitate the understanding of their origin, clinical features, neuroimaging characteristics, and pertinent adjuvant treatment.
Keywords: Neuroblastoma, Rapid growth, Sellar, Syndrome of inappropriate antidiuretic hormone secretion
INTRODUCTION
Neuroblastomas are the most common extracranial solid tumors occurring in infants and children. They originate from the sympathetic ganglion and adrenal medulla.[
CASE REPORT
A 33-year-old woman with the dizziness as a chief complain. Magnetic resonance imaging (MRI) found a sellar lesion with a diameter of 18 mm invading into the bilateral cavernous sinus [
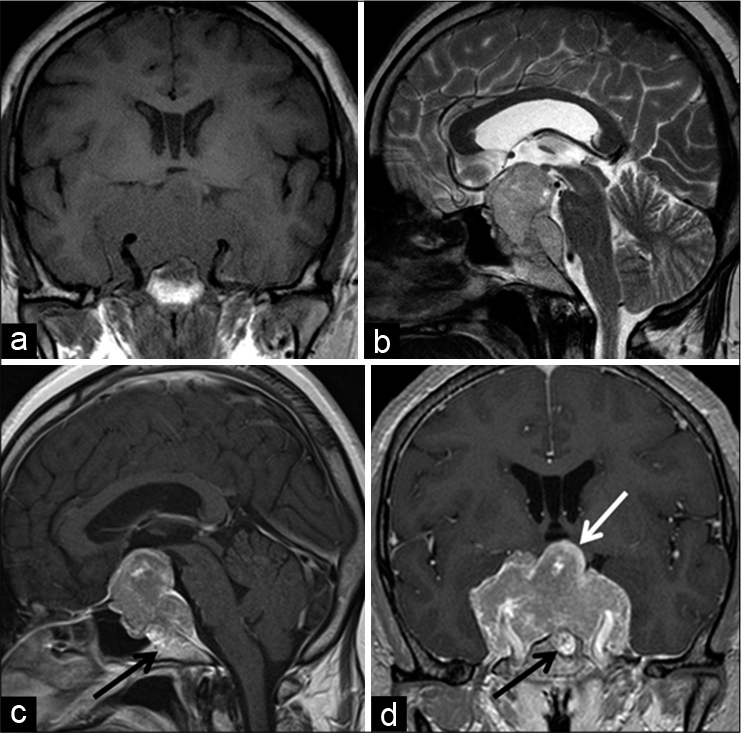
Figure 1:
Magnetic resonance imaging (MRI) at the first visit showing an incidentally found sellar tumor. The posterior pituitary gland was located superoposterior to the tumor (open arrowhead in a). The pituitary gland seemed to be on top of the tumor (arrows in c and d). Cavernous sinus invasion is indicated by arrowheads (b and d) (a) Sagittal T1-weighted image (WI). (b) Coronal T1WI. (c) Sagittal gadolinium-enhanced image (GEI). (d) Coronal GEI.
On admission to Kagoshima University Hospital, the patient had clear consciousness. Her vision was 0.3 in the right and 0.7 on the left. Bitemporal hemianopia was found on perimetry. Her serum sodium (Na) level was 127 meq/L. MRI showed a large tumor with a maximal diameter of 56 mm, involving the sellar, suprasellar, retroclival, and bilateral cavernous sinus regions and strongly compressing the optic nerve and optic chiasm [
Figure 2:
A magnetic resonance imaging (MRI) study for 6 years after the first visit demonstrated an extensively grown tumor occupying the sellar and wide juxtasellar regions (a-d). The white arrow indicates a relatively well-enhanced area, supposed to be the pituitary gland, on top of the tumor (d). The black arrow indicates an invasion of the front of the tumor into the clivus (c and d). (a) Coronal T1WI, (b) sagittal T2WI, (c) sagittal GEI, (d) coronal GEI.
The assessments of the anterior pituitary hormonal function showed hyperprolactinemia (53.9 ng/mL) and growth hormone deficiency, but the other four hormonal axes showed normal secretory functions. There was no manifestation of diabetes insipidus.
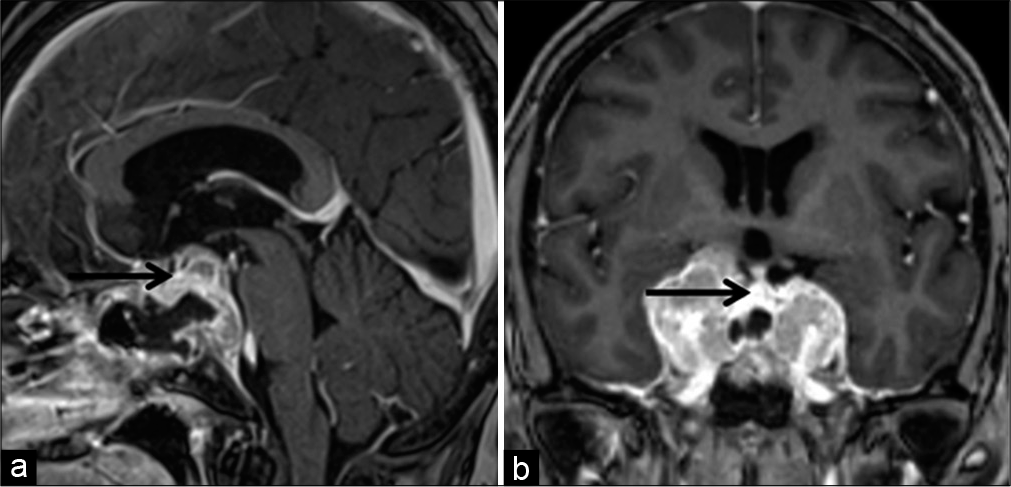
Decompression surgery of the optic chiasm was conducted through an endoscopic transsphenoidal approach. The removal of the thickened sella and thinned fibrous tissue presumed to be a remnant of the dura mater revealed the tumor, which was basically fibrous and easy to bleed. Tumor removal was continued until the suprasellar arachnoid membrane and tough tissue consistent with a pituitary gland appeared, leaving the tumor in the cavernous sinuses. Postoperative MRI demonstrated the sufficient decompression of the optic chiasm and an intact pituitary gland [
Pathologically, the tumor was composed of a proliferation of round to oval neoplastic cells with scant to moderate cytoplasm and chromatin-rich short rod-like nuclei against a background of intercellular neuropil-like fibrillary matrix [
Figure 5:
Histologic features of the specimen obtained by transsphenoidal surgery. The tumor was composed of neoplastic cells with scant to moderate cytoplasm and chromatin-rich oval or short rod-like nuclei. The neoplastic cells partly formed a lobular arrangement (×200, a). A few ganglion-like cells were seen (×600, b). On immunohistochemistry, the tumor was diffusely positive for synaptophysin (×200, c), neurofilament protein (×200, d), NeuN (×200, e), chromogranin A (×200, f), and arginine vasopressin (×400, g). S-100 protein was positive mainly in the spindle cells surrounding a fibrovascular stroma (×200, h). Bar = 100 μm.
A postoperative whole-body CT scan revealed a right ovarian cyst, which was later pathologically proven to be a simple cyst; otherwise, no other abnormalities were found. She underwent 50-Gy of intensity-modulated radiation therapy to the residual tumor due to hospital’s standard operating procedure, which has controlled its growth for the 3 years [
DISCUSSION
This is an unfortunate case, in which the recommendation of regular follow-up was ignored, but, ironically, the natural course of the sellar neuroblastoma still occurred in 6 years. [
Lach et al. assumed that the tumor was attributable to the transformation of the pituitary epithelium into neuronal cells based on the colocalization of prolactin-immunopositive granules in the neoplastic neuronal cells.[
The unstretched pituitary gland located on top of the tumor, relatively preserved anterior pituitary function, thickened sellar floor, and tattered dura mater on the sellar floor in our case hinted that the tumor arose from the lower part of sella turcica, including the dura mater, subdural space, and base of the pituitary gland.
The median age of patients with similar cases reported previously was 40 years (range: 29–71 years). These patients were much younger than the patients with nonfunctioning adenomas (n = 166, mean age: 62 years, and range: 19–84 years), who we treated for the last 10 years. The major manifestation was visual impairment, including temporal hemianopia. Cavernous sinus symptoms were seen in three out of the 11 cases, which are rarely seen in pituitary adenomas even with cavernous sinus invasion.[
Hyperprolactinemia, due to the stalk effect, was recorded in six cases. However, the impairment of other hormones was relatively low, which was recorded in only four cases. The anterior pituitary provocation test, in our case, found a well- preserved secretory function, considering the large tumor volume; only the GH secretion was compromised.[
SIADH, by definition, is a condition of excessive secretion or action of AVP irrespective of hyponatremia and inappropriate urinary concentration. There are various causes of SIADH: central nervous system disease such as meningitis, brain tumor, and cerebral hemorrhage; lung diseases such as pneumonia, lung tumor, and tuberculosis; iatrogenic such as vincristine, clofibrate, and carbamazepine drug adverse effects; and ectopic ADH- producing tumors such as small cell lung carcinoma and pancreatic cancer.[
In general, MRI revealed the aggressive nature of the disease. Among the 11 reported cases (including the present case), the suprasellar extension was seen in ten, cavernous sinus invasion in seven with five bilateral cases, clival involvement in four, sphenoid sinus involvement in three, and cerebrospinal fluid dissemination in one case. Eight cases showed multiple lobar appearances. Calcification was noted in only two cases.[
As in the previously reported cases, our case displayed immunohistochemical positivity for neuronal markers, including neurofilament protein, chromogranin, and synaptophysin, but negativity for all anterior pituitary hormones.[
The natural course of the pituitary neuroblastoma remains not well known. In our case, the tumor had grown rapidly in 6 years, showing an aggressive nature of this tumor. Mariani et al.[
At present, there has been no consensus established regarding the treatment of sellar neuroblastomas. Postoperative radiation, including gamma-knife, was conducted in the ten reported cases, which provided good control of the tumor during the 5-year follow-up period, except for one case [
In conclusion, although sellar neuroblastomas seem extremely rare, awareness of this special entity will promote the accumulation of clinicopathologic information, which may facilitate the understanding of its origin, clinical features, neuroimaging characteristics, and pertinent adjuvant treatment.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Al Ahwal M, Jha N, Nabholtz JM, Hugh J, Birchall I, Nguyen GK. Olfactory neuroblastoma: Report of a case associated with inappropriate antidiuretic hormone secretion. J Otolaryngol. 1994. 23: 437-9
2. Berger MS, Edwards MS, Wara WM, Levin VA, Wilson CB. Primary cerebral neuroblastoma. Long-term follow-up review and therapeutic guidelines. J Neurosurg. 1983. 59: 418-23
3. Dulguerov P, Allal AS, Calcaterra TC. Esthesioneuroblastoma: A meta-analysis and review. Lancet Oncol. 2001. 2: 683-90
4. Horten BC, Rubinstein LJ. Primary cerebral neuroblastoma. A clinicopathological study of 35 cases. Brain. 1976. 99: 735-56
5. Jakumeit HD. Neuroblastoma of the olfactory nerve. Acta Neurochir (Wien). 1971. 25: 99-108
6. Lach B, Rippstein P, Benott BG, Staines W. Differentiating neuroblastoma of pituitary gland: Neuroblastic transformation of epithelial adenoma cells. Case report. J Neurosurg. 1996. 85: 953-60
7. Lin JH, Tsai DH, Chiang YH. A primary sellar esthesioneuroblastomas with unusual presentations: A case report and reviews of literatures. Pituitary. 2009. 12: 70-5
8. Mariani L, Schaller B, Weis J, Ozdoba C, Seiler RW. Esthesioneuroblastoma of the pituitary gland: A clinicopathological entity? Case report and review of the literature. J Neurosurg. 2004. 101: 1049-52
9. Matthay KK, Maris JM, Schleiermacher G, Nakagawara A, Mackall CL, Diller L. Neuroblastoma. Nat Rev Dis Primers. 2016. 2: 16078-
10. Mete O, Lopes MB, Roncaroli F, Tihan T, Yamadha S, Lloyd RV, Osamura RY, Kloppel G, Rossi J.editors. Tumours of the posteior pituitary. WHO Classification of Tumours of Endocrine Organs. Lyon: IARC; 2017. p. 52-4
11. Nakano T, Motoshita J, Sawada F, Okabe M, Tamae A, Hiramatsu S. Syndrome of inappropriate antidiuretic hormone secretion in a case of olfactory neuroblastoma without anti-diuretic hormone immunoreactivity: A case report and review of the literature. Auris Nasus Larynx. 2017. 44: 771-4
12. Oyama K, Yamada S, Usui M, Kovacs K. Sellar neuroblastoma mimicking pituitary adenoma. Pituitary. 2005. 8: 109-14
13. Radotra B, Apostolopoulos V, Sandison A, Hatfield EC, Mendoza N, Moss J. Primary sellar neuroblastoma presenting with syndrome of inappropriate secretion of anti-diuretic hormone. Endocr Pathol. 2010. 21: 266-73
14. Roy A, Timothy J, Anthony R, Chakrabarty A. Correspondence: Aesthesioneuroblastoma arising in pituitary gland. Neuropathol Appl Neurobiol. 2000. 26: 177-9
15. Sajko T, Rumboldt Z, Talan-Hranilovic J, Radic I, Gnjidic Z. Primary sellar esthesioneuroblastoma. Acta Neurochir (Wien). 2005. 147: 447-8
16. Sarwar M. Primary sellar-parasellar esthesioneuroblastoma. AJR Am J Roentgenol. 1979. 133: 140-1
17. Schmalisch K, Psaras T, Beschorner R, Honegger J. Sellar neuroblastoma mimicking a pituitary tumour: Case report and review of the literature. Clin Neurol Neurosurg. 2009. 111: 774-8
18. Wirsig-Wiechmann CR, Wiechmann AF, Eisthen HL. What defines the nervus terminalis? Neurochemical, developmental, and anatomical criteria. Prog Brain Res. 2002. 141: 45-58
19. Yamamuro S, Fukushima T, Yoshino A, Yachi K, Ogino A, Katayama Y. Primary sellar neuroblastoma in an elderly patient: Case report. NMC Case Rep J. 2015. 2: 57-60
20. Yaris N, Yavuz MN, Reis A, Yavuz AA, Okten A. Primary cerebral neuroblastoma: A case treated with adjuvant chemotherapy and radiotherapy. Turk J Pediatr. 2004. 46: 182-5