- Department of Neurosurgery, Albany Medical Center, Albany, New York, United States.
- Department of Radiology, Albany Medical Center, Albany, New York, United States.
- Department of Pathology, Albany Medical Center, Albany, New York, United States.
- Department of Otolaryngology and Head and Neck Surgery, Albany Medical Center, Albany, New York, United States.
Correspondence Address:
Adedamola Adepoju
Department of Neurosurgery, Albany Medical Center, Albany, New York, United States.
DOI:10.25259/SNI_489_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Adedamola Adepoju1, Ananth Narayan2, Mahmoud Aldyab3, David Foyt4, Maria Peris-Celda1. Absence of contrast enhancement in a petroclival meningioma: Case report and systematic literature review. 04-Dec-2020;11:418
How to cite this URL: Adedamola Adepoju1, Ananth Narayan2, Mahmoud Aldyab3, David Foyt4, Maria Peris-Celda1. Absence of contrast enhancement in a petroclival meningioma: Case report and systematic literature review. 04-Dec-2020;11:418. Available from: https://surgicalneurologyint.com/surgicalint-articles/10427/
Abstract
Background: Meningioma is one of the most common intracranial tumors with well-established radiologic features such as contrast enhancement, dural tail, and hyperostosis on computed tomography and magnetic resonance imaging. Contrast enhancement is usually homogeneous or heterogeneous based on tumor vascularity and underlying histopathology. Even in this context, faint or nonenhancing meningioma is exceedingly rare.
Case Description: A 57-year-old male presented with progressive right hearing loss, disequilibrium, occasional difficulty swallowing, and facial numbness. Imaging demonstrated an extensive hypodense, nonenhancing right cerebellopontine angle mass extending from the interpeduncular, and ambient cisterns to the foramen magnum. The pathological analysis demonstrated a microcystic meningioma WHO Grade I. There are few reported case reports or series of minimal or nonenhancing meningiomas, and a systematic review was performed for these cases. Seven peer-reviewed articles with 14 verifiable cases were identified and reviewed for radiologic features, tumor location, and tumor classification. The majority of minimal or nonenhancing meningiomas were microcystic, and most of them located at the convexity. This is the second case reported of a nonenhancing meningioma at the cerebellopontine angle and petroclival region
Conclusion: Meningioma should be considered a differential diagnosis for nonenhancing lesion at the cerebellopontine and petroclival regions.
Keywords: Cerebellopontine angle, Glial fibrillary astrocytic protein, Meningioma, Microcystic meningioma, Petroclival
INTRODUCTION
Meningioma is one of the most common benign intracranial tumors. The use of neuroimaging has increased the diagnosis of meningioma, and there are classic radiologic features associated with the tumor, including contrast enhancement, dural tail, and hyperostosis of the adjacent bone. On computed tomography (CT), meningiomas are mostly isodense to hyperdense, and about 20% have calcifications.[
CASE PRESENTATION
History and physical examination
A 57-year-old right-handed male presented with progressive right hearing loss, disequilibrium, occasional difficulty swallowing, and right facial numbness. CT and MRI showed a right cerebellopontine angle tumor extending from the interpeduncular cistern to the foramen magnum. An audiogram revealed moderate right sensorineural hearing loss. Neurological examination demonstrated right decreased hearing, right facial numbness, and a positive Romberg sign.
Imaging
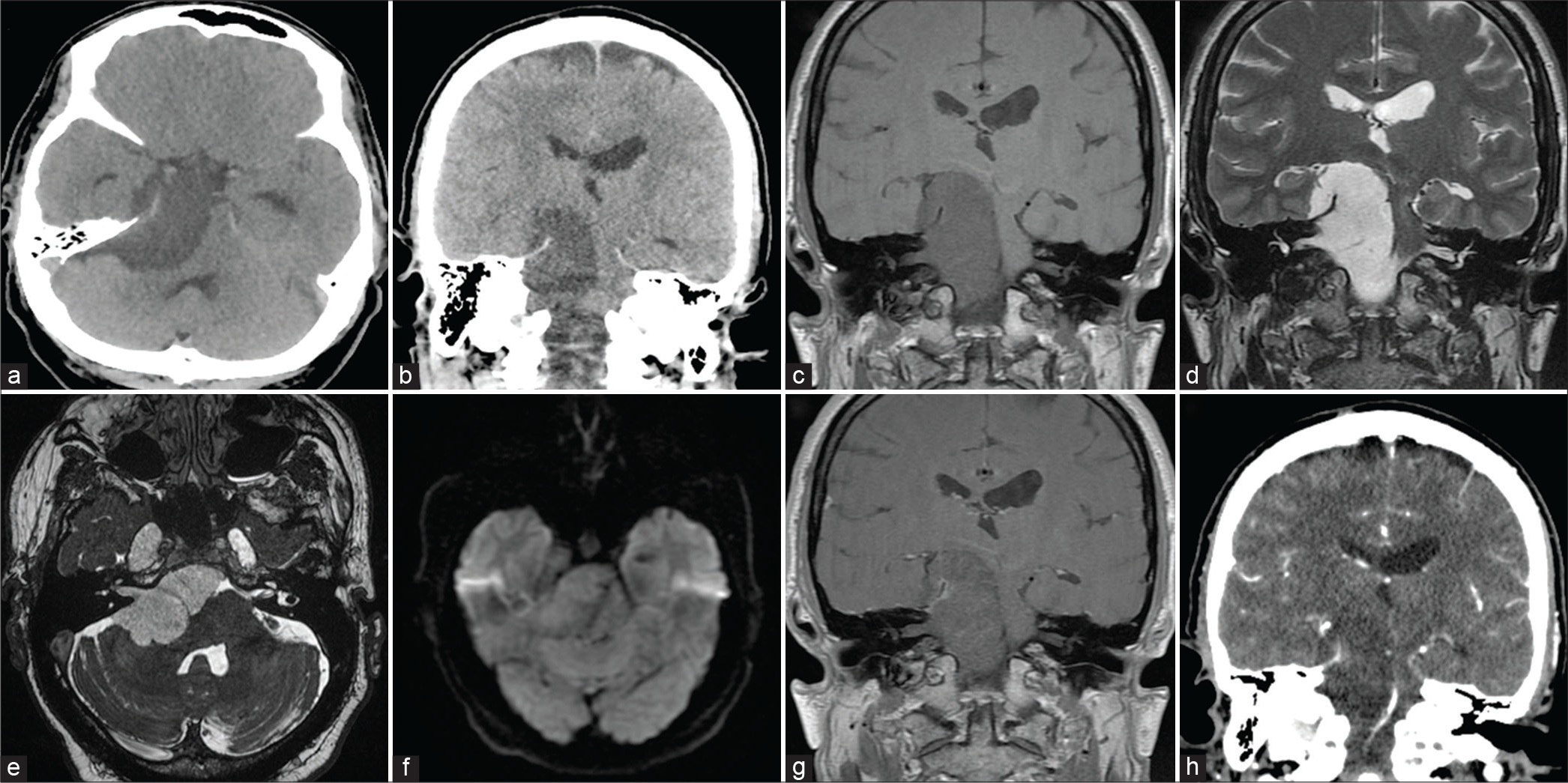
Noncontrast CT of the head revealed a hypodense right cerebellopontine angle mass extending from the interpeduncular and ambient cisterns to the foramen magnum [
Figure 1:
Preoperative images. Axial (a) and coronal (b) noncontrast CT image showing the hypodense cerebellopontine mass extending in the supratentorial space, interpeduncular and ambiens cisterns, and the right Meckel’s cave. (c and d) T1- and T2-weighted coronal MRI showing the extent of the tumor. (e) 3D-FIESTA sequence indicating the extension of the tumor into the internal acoustic canal. (f) Axial diffusion-weighted imaging demonstrating intermediate diffusion signal when compared with CSF. (g) Coronal view of T1-weighted with gadolinium demonstrating faint tentorial enhancement and no enhancement within the tumor. (h) Coronal view of a contrasted CT demonstrating the tentorial enhancement.
Operative procedure
A posterior petrosal retrolabyrinthine approach with retrosigmoid and temporal craniotomies was used for the surgical approach in a staged operation (approach and resection in different days). After orotracheal intubation, the patient was placed in a left lateral decubitus position. Bone landmarks and neuronavigation were used for surgical incision planning. Facial nerve monitoring was placed. Incision and opening were made in a layered fashion, and the retrosigmoid and temporal craniotomies were performed as described in Graffeo et al. (2018). With the aid of a surgical microscope, the presigmoid retrolabyrinthine part of the approach was performed. There was no dural opening. The bone flap was reattached, and the incision was closed accordingly. The patient was extubated and was observed on the neurosurgical floor.
Two days later, he returned to the operating room for resection of the tumor and was positioned as described above. Neuromonitoring, including brainstem auditory evoked potential, was utilized for ipsilateral facial, glossopharyngeal, and accessory nerves. The incision and the bone flap were reopened. The retrosigmoid dura was opened in a curvilinear fashion, and the CSF was drained from the cisterna magna to relax the posterior fossa. The tumor was identified as a greyish mass with moderate vascularity and soft consistency, and resection was performed with an ultrasonic aspirator. In the posterior fossa, the vestibulocochlear, facial, and lower cranial nerves were identified posterior to the tumor, whereas the abducens nerve was found within the tumor. The trigeminal nerve was displaced superiorly against the tentorium. The infratentorial portion of the tumor was completely resected through the retrosigmoid part of the approach. The presigmoid and the temporal dura mater were opened, and the tentorium was divided after ligation of the superior petrosal sinus.[
Postoperative course
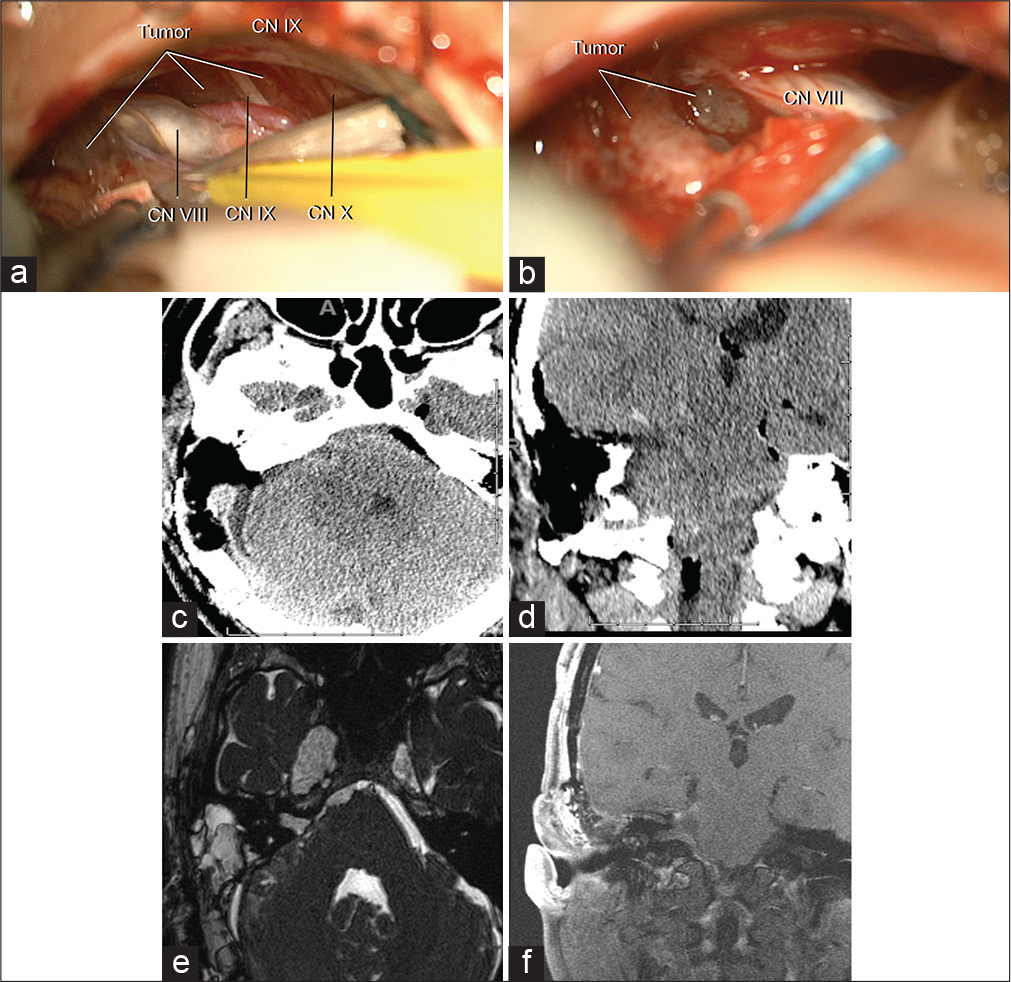
He had a right partial forth, and sixth nerve palsies and mild facial weakness (House-Brackman 2) after surgery. A postoperative CT scan is shown in [
Figure 2:
Intraoperative pictures and postoperative imaging after tumor resection. (a and b) intraoperative pictures demonstrating a grayish pink tumor of moderate vascularization anteromedial to cranial nerves VII, VIII, XI, and X. The cranial nerves IV and VI were embedded within the tumor and preserved. CN, cranial nerves. Axial (c) and coronal (d) non contrast postoperative CT images showing the posterior petrosal approach. Axial FIESTA (e) and coronal (f) T1-weighted with contrast postoperative MRI images showing the extent of tumor resection, a small portion of meningioma was left in Meckel’s cave.
Pathology
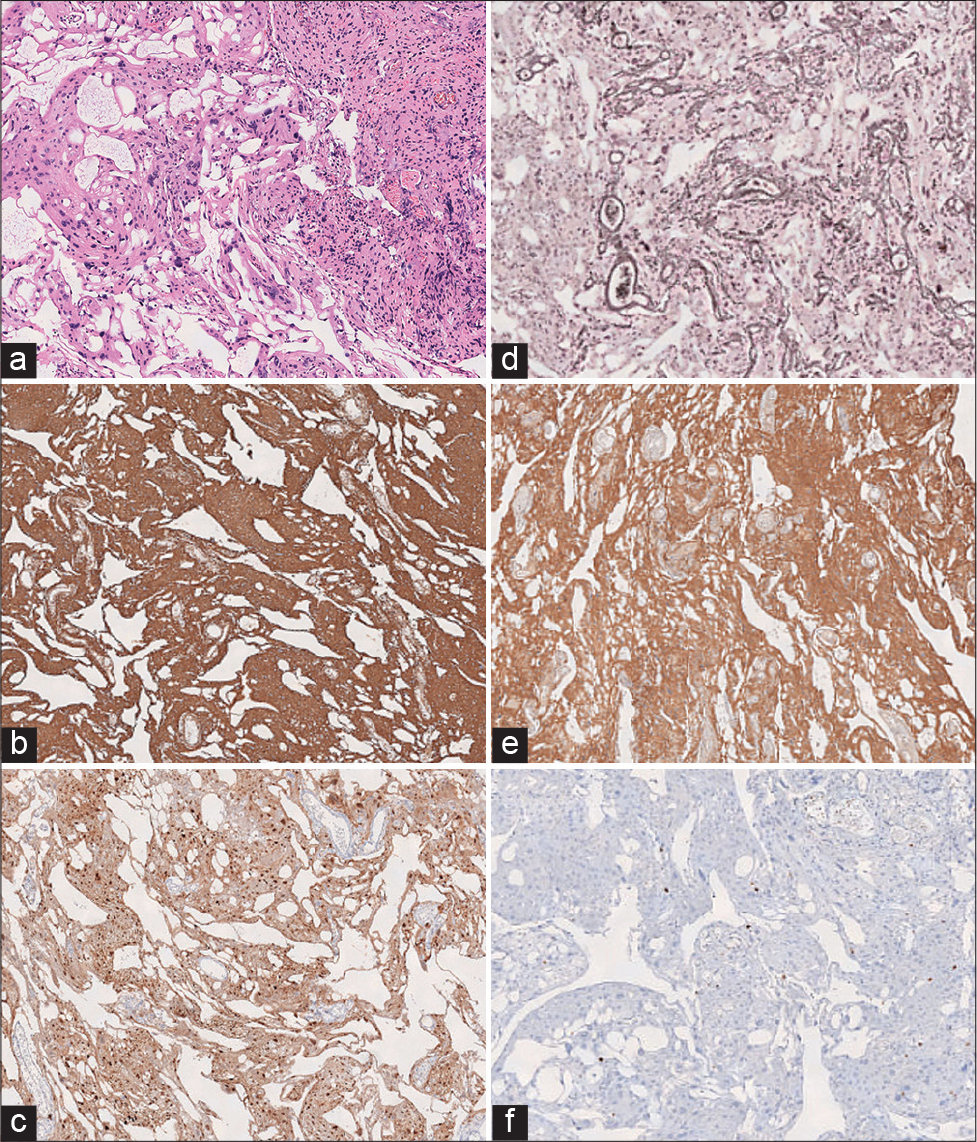
Grossly, the tumor appeared tan to pink. Microscopically, the tissue had lobulated architecture, mixed with prominent loose myxoid microcystic background and minor angiomatous changes. The lobulated component contained meningothelial whorls [
Figure 3:
(a) Hematoxylin and eosin (×4) stained fragments of tissue with the proliferation of bland ovoid to epithelioid cells, with pink cytoplasm and indistinct cell borders. The whorled appearance is better appreciated. Nuclei appear to have some size variability, but no significant atypia or anaplasia is identified. The cystic pattern and angiomatous changes are also evident (×40). (b) Vimentin: Immunohistochemical staining for Vimentin shows a strong, diffuse cytoplasmic staining pattern. (c) Cyclin-D1: Immunohistochemical staining for Cyclin-D1 shows a diffuse staining pattern. (d) Reticulin: Immunohistochemical staining for reticulin shows an epithelioid staining pattern. (e) GFAP: Immunohistochemical staining for GFAP shows a strong, diffuse cytoplasmic staining pattern. (f) KI-67: Immunohistochemical staining for GFAP shows scattered positive staining, corresponding to a low proliferation index estimated to be <2%.
Systematic review
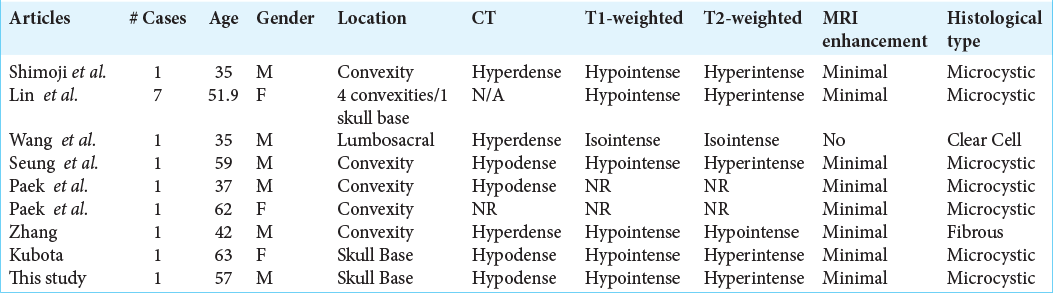
A systematic review of the literature for minimal or nonenhancing meningioma was conducted using the Preferred Reporting Items for Systematic Reviews and Meta-analysis guidelines. Search items including “nonenhancing meningioma,” “unenhanced meningioma,” and more broadly “enhancing meningioma” were entered into the online databases PubMed and web of science. The search returned 652 total peer-reviewed articles, and each item was reviewed to evaluate for cases with minimal or nonenhancing meningioma. Screening guidelines included recoverable English-written articles with basic demographic and radiographic information, including images to support the interpretation of radiographic features. The last query was 04 April 2020. Selection and reporting biases are acknowledged. Cystic and necrotic meningioma was excluded from the review. Minimal enhancement was defined as faint or hardly noticeable contrast enhancement on radiologic images or interpretation. Seven articles, including case reports and case series, had 14 verifiable cases included in the systemic review [
DISCUSSION
Meningioma is a common intradural tumor that avidly enhances with contrast. Almost all meningiomas either enhance homogeneously or heterogeneously. The meningioma presented in this report had faint enhancement with contrast, which is very rare. Dural tail, found in 72% of meningioma,[
The histology and the immunostaining demonstrated that the tumor was a microcystic meningioma. The tumor stained for typical meningioma markers such as Vimentin, Reticulin, and Cyclin D1.[
Meningioma is one of the most incidentally diagnosed intracranial lesions.[
Microcystic meningioma is rare WHO Grade 1 meningioma, and it constitutes about 1.6% of all intracranial meningiomas. Although the microcystic type comprised the majority of the minimally enhancing meningiomas, only 10.2% of microcystic meningiomas had a faint enhancement in MRI, and thus, the majority of them had typical radiological features of meningiomas.[
CONCLUSION
Meningioma should be considered a differential diagnosis for a non-enhancing lesion at the cerebellopontine and petroclival regions.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Aoki S, Sasaki Y, Machida T, Tanioka H. Contrast-enhanced MR images in patients with meningioma: Importance of enhancement of the dura adjacent to the tumor. AJNR Am J Neuroradiol. 1990. 11: 935-8
2. Backer-Grøndahl T, Moen BH, Arnli MB, Torseth K, Torp SH. Immunohistochemical characterization of brain-invasive meningiomas. Int J Clin Exp Pathol. 2014. 7: 7206-19
3. Buetow MP, Buetow PC, Smirniotopoulos JG. Typical, atypical, and misleading features in meningioma. Radiographics. 1991. 11: 1087-106
4. Commins DL, Atkinson RD, Burnett ME. Review of meningioma histopathology. Neurosurg Focus. 2007. 23: E3
5. Eng LF. Glial fibrillary acidic protein (GFAP): The major protein of glial intermediate filaments in differentiated astrocytes. J Neuroimmunol. 1985. 8: 203-14
6. Graffeo CS, Peris-Celda M, Perry A, Carlstrom LP, Driscoll CL, Link MJ. Anatomical step-by-step dissection of complex skull base approaches for trainees: Surgical anatomy of the posterior petrosal approach. J Neurol Surg B Skull Base. 2019. 80: 338-51
7. Haberler C, Jarius C, Lang S, Rossler K, Gruber A, Hainfellner JA. Fibrous meningeal tumours with extensive non-calcifying collagenous whorls and glial fibrillary acidic protein expression: The whorling-sclerosing variant of meningioma. Neuropathol Appl Neurobiol. 2002. 28: 42-7
8. Huang RY, Bi WL, Weller M, Kaley T, Blakeley J, Dunn I. Proposed response assessment and endpoints for meningioma clinical trials: Report from the response assessment in neurooncology working group. Neuro Oncol. 2019. 21: 26-36
9. Kim SH, Kim DG, Kim CY, Choe G, Chang KH, Jung HW. Microcystic meningioma: The characteristic neuroradiologic findings. J Korean Neurosurg Soc. 2003. 35: 401-6
10. Kubota Y, Ueda T, Kagawa Y, Sakai N, Hara A. Microcystic meningioma without enhancement on neuroimaging--case report. Neurol Med Chir (Tokyo). 1997. 37: 407-10
11. Lin Z, Zhao M, Li X, Wang J, Qiu P, Lan F. Characteristic features and proposed classification in 69 cases of intracranial microcystic meningiomas. Neurosurg Rev. 2019. 42: 443-53
12. Ng HK, Tse CC, Lo ST. Microcystic meningiomas--an unusual morphological variant of meningiomas. Histopathology. 1989. 14: 1-9
13. Paek SH, Kim SH, Chang KH, Park CK, Kim JE, Kim DG. Microcystic meningiomas: Radiological characteristics of 16 cases. Acta Neurochir (Wien). 2005. 147: 965-72
14. Pieper DR, Al-Mefty O, Hanada Y, Buechner D. Hyperostosis associated with meningioma of the cranial base: Secondary changes or tumor invasion. Neurosurgery. 1999. 44: 742-6
15. Shimoji K, Yasuma Y, Mori K, Eguchi M, Maeda M. Unique radiological appearance of a microcystic meningioma. Acta Neurochir (Wien). 1999. 141: 1119-21
16. Su M, Ono K, Tanaka R, Takahashi H. An unusual meningioma variant with glial fibrillary acidic protein expression. Acta Neuropathol. 1997. 94: 499-503
17. Wang XQ, Huang MZ, Zhang H, Sun FB, Tao BB, Feng BH. Clear cell meningioma: Clinical features, CT, and MR imaging findings in 23 patients. J Comput Assist Tomogr. 2014. 38: 200-8
18. Wanschitz J, Schmidbauer M, Maier H, Rossler K, Vorkapic P, Budka H. Suprasellar meningioma with expression of glial fibrillary acidic protein: A peculiar variant. Acta Neuropathol. 1995. 90: 539-44
19. Whittle IR, Smith C, Navoo P, Collie D. Meningiomas. Lancet. 2004. 363: 1535-43
20. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010. 99: 307-14
21. Zhang Q, Wang X. A distinctive pathologic meningioma completely without enhancement and dural tail sign on imaging findings. World Neurosurg. 2017. 101: 815