- Department of Neurosurgery, Japanese Red Cross Kyoto Daini Hospital, Kyoto, Japan.
- Department of Neurosurgery, Kyoto Prefectural University of Medicine, Graduate School of Medical Sciences, Kyoto, Japan.
Correspondence Address:
Saki Kotani, Department of Neurosurgery, Japanese Red Cross Kyoto Daini Hospital, Kyoto, Japan.
DOI:10.25259/SNI_1010_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Saki Kotani1, Nobukuni Murakami1, Tomoyuki Doi1, Takahiro Ogawa1, Naoya Hashimoto2. Acute epidural vertex hematoma with good hemostasis using delayed surgery after monitoring of coagulation and fibrinolytic parameters: A case report. 24-Feb-2023;14:73
How to cite this URL: Saki Kotani1, Nobukuni Murakami1, Tomoyuki Doi1, Takahiro Ogawa1, Naoya Hashimoto2. Acute epidural vertex hematoma with good hemostasis using delayed surgery after monitoring of coagulation and fibrinolytic parameters: A case report. 24-Feb-2023;14:73. Available from: https://surgicalneurologyint.com/surgicalint-articles/12165/
Abstract
Background: The appropriate timing and method of surgery for vertex epidural hematoma (VEDH) are uncertain due to the presentation and slow symptomatic exacerbation caused by bleeding from a venous origin involving the injured superior sagittal sinus (SSS). Coagulation and fibrinolytic disorders that occur after traumatic brain injury also worsen bleeding. For these reasons, it is challenging to decide the surgical procedure and timing of surgery.
Case Description: A 24-year-old man involved a car accident and was transported to our emergency department. He was unconscious but not lethargic. Computed tomography showed VEDH overlying the SSS, and hematoma increased temporarily. Due to abnormal coagulation and fibrinolysis at admission, he underwent intentionally delayed surgery after control of coagulation and fibrinolysis. Bilateral parasagittal craniotomy was chosen to ensure hemostasis from the torn SSS. The patient improved without complications and was discharged with no neurological deficit. This case indicates that this surgical strategy is favorable for VEDH with slow symptomatic progression.
Conclusion: VEDH is mostly caused by bleeding from the injured SSS secondary to diastatic fracture of sagittal suture. Intentionally delayed surgical intervention using bilateral parasagittal craniotomy after stabilization of coagulation and fibrinolysis is favorable for prevention of further hemorrhage and good hemostasis.
Keywords: Coagulation, Epidural vertex hematoma, Fibrinolysis, Parasagittal craniotomy, Traumatic brain injury
INTRODUCTION
Vertex epidural hematoma (VEDH) is a rare condition that accounts for 1–8% of all intracranial extradural hematomas.[
CASE REPORT
Clinical presentation
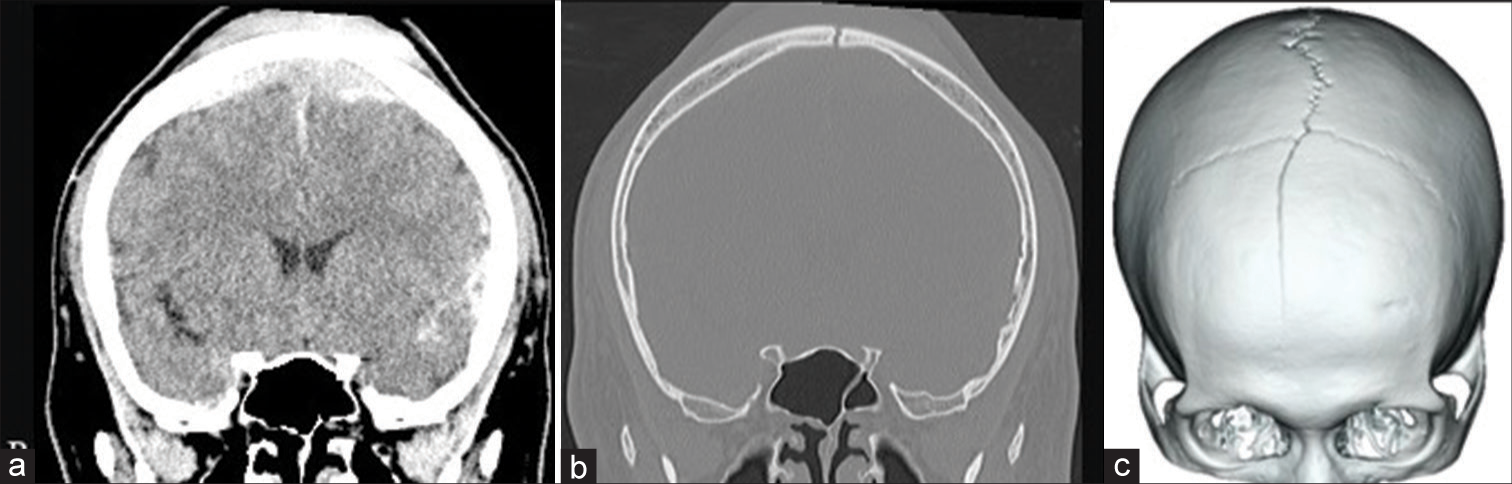
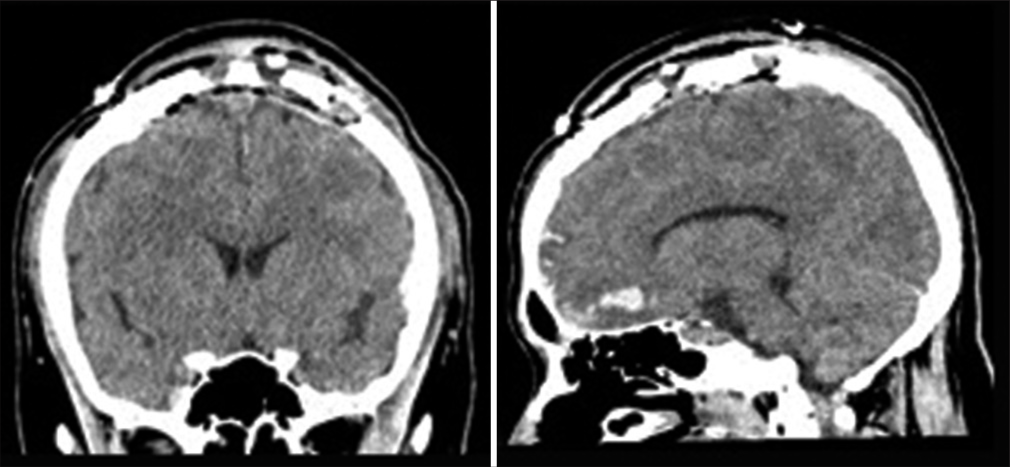
A 24-year-old man had an accident involving a vehicle while walking and was transported to our emergency department. On arrival, his level of consciousness had deteriorated (Glasgow Coma Scale: 13 [E3V4M6]). He was not lethargic, and his pupils were equal and round, and reactive to light and movement. Computed tomography (CT) showed traumatic subarachnoid hemorrhage, a 1-cm thick VEDH overlying the SSS, and a parietal skull fracture reaching a diastatic fracture of the sagittal suture [
Surgical procedure
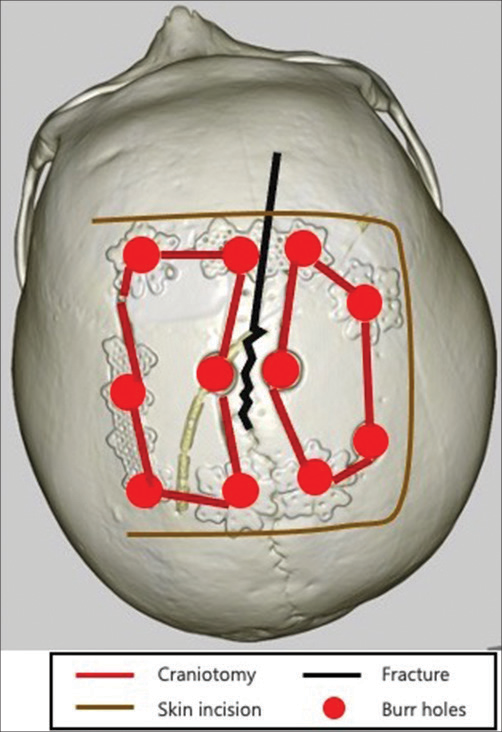
Under general anesthesia, the patient was placed in the supine position with the head in a neutral position and fixed in a Mayfield clamp. A horseshoe skin incision overlying the lesion was sketched [
Postoperative course
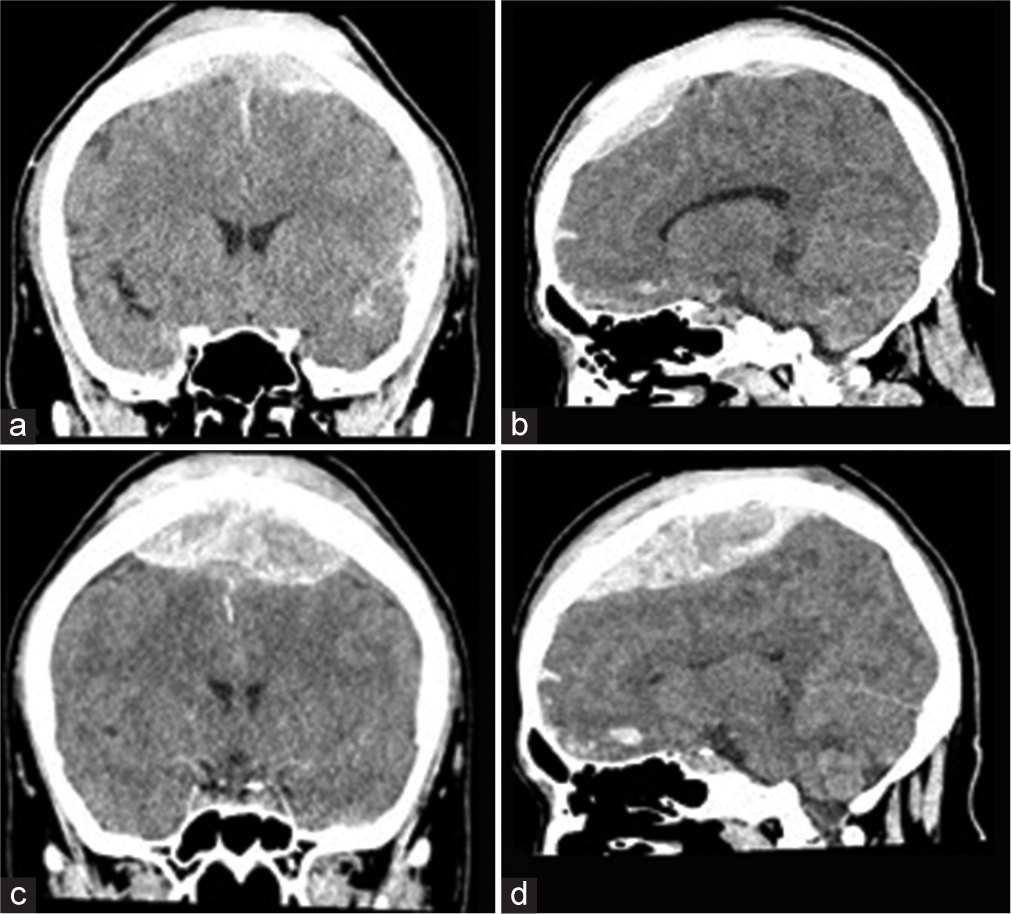
After surgery, the patient was returned to the intensive care unit with intubation and sedation. Postoperative head CT showed no residual hematoma and no recurrence of hemorrhage and contusion [
DISCUSSION
VEDH accounts for 1–8% of intracranial extradural hematomas.[
VEDH is usually caused by venous bleeding from the SSS and slowly progresses. Most cases initially have no symptoms, but delayed neurological symptoms frequently appear. Kim et al. found that 71.4% of cases were initially managed with observation without surgery, but that delayed neurological deterioration then occurred in 44% of these patients and that 40% eventually required surgical intervention.[
Among patients with traumatic brain injury (TBI), 63% have coagulopathy on admission, and this is associated with progressive intracranial brain contusions and contributes to a poor outcome.[
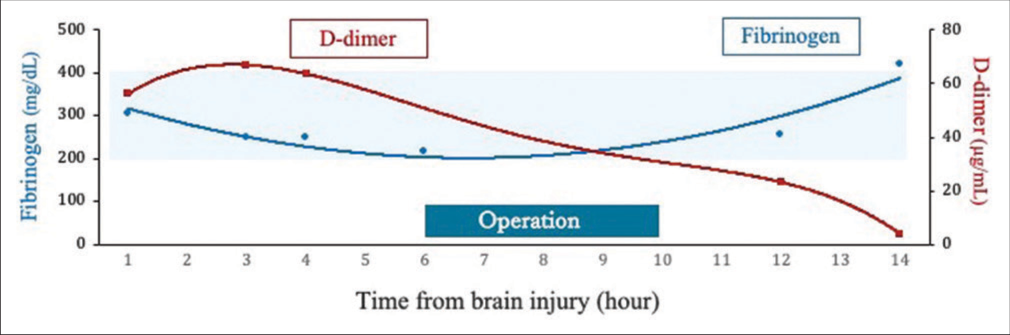
Hemostatic disorders that occur following TBI increase within 3 h after brain injury and a hypercoagulable stage follows 6 h after injury.[
Surgical intervention in the acute phase increases the risk of hemostasis and progression of intracranial brain contusions due to coagulation and fibrinolytic disorders. In a case of VEDH with slow symptomatic exacerbation, it is preferable to perform intentionally delayed surgery after stabilizing coagulation and fibrinolysis. There are two main options for the surgical technique: single-flap vertex craniotomy or bilateral parasagittal craniotomy. Kim et al. reported that there was no significant difference between these methods in 40 cases.[
CONCLUSION
VEDH is mostly caused by bleeding from the injured SSS secondary to diastatic fracture of sagittal suture. Surgical management is difficult due to exacerbation of hemorrhage in the perioperative period. Intentionally delayed surgical intervention using bilateral parasagittal craniotomy after stabilization of coagulation and fibrinolysis is favorable for prevention of further hemorrhage and good hemostasis.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Borzone M, Rivano C, Altomonte M, Capuzzo T. Acute traumatic vertex epidural haematomas surgically treated. Acta Neurochir (Wien). 1998. 93: 55-60
2. Fernandes-Cabral DT, Kooshkabadi A, Panesar SS, Celtikci E, Borghei-Razavi H, Celtikci P. Surgical management of vertex epidural hematoma: Technical case report and literature review. World Neurosurg. 2017. 103: 475-83
3. Kim JH, Yoon WK, Kwon TH, Kim JH. Clinical features and treatment strategies for vertex epidural hematoma: A systematic review and meta-analysis from individual participant data. Neurosurg Rev. 2022. 45: 819-30
4. Maegele M, Schöchl H, Menovsky T, Maréchal H, Marklund N, Buki A. Coagulopathy and haemorrhagic progression in traumatic brain injury: Advances in mechanisms, diagnosis, and management. Lancet Neurol. 2017. 16: 630-47
5. Nakae R, Takayama Y, Kuwamoto K, Naoe Y, Sato H, Yokota H. Time course of coagulation and fibrinolytic parameters in patients with traumatic brain injury. J Neurotrauma. 2016. 33: 688-95
6. Nakae R, Yokobori S, Takayama Y, Kanaya T, Fujiki Y, Igarashi Y. A retrospective study of the effect of fibrinogen levels during fresh frozen plasma transfusion in patients with traumatic brain injury. Acta Neurochir (Wien). 2019. 161: 1943-53
7. Takayama Y, Yokota Y, Sato H, Naoe Y, Araki T. Pathophysiology, mortality, treatment of acute phase of haemostatic disorders of traumatic brain injury. Jpn J Neurosurg (Tokyo). 2013. 22: 837-41