- Department of Neurosurgery, Kantonsspital Aarau, Tellstrasse 1, 5001 Aarau/Switzerland
- Department of Neurosurgery, Luzerner Kantonsspital, Spitalstrasse, 6000 Luzern/Switzerland
- Universität Basel, Petersplatz 1, 4001 Basel/Switzerland
- Department of Anatomy, Histology and Embryology, Medical University of Innsbruck (MUI), 6020 Innsbruck/Austria
Correspondence Address:
Alexander Spiessberger
Department of Anatomy, Histology and Embryology, Medical University of Innsbruck (MUI), 6020 Innsbruck/Austria
DOI:10.4103/sni.sni_298_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Alexander Spiessberger, Fabian Baumann, Alexandra Stauffer, Serge Marbacher, Karl F. Kothbauer, Javier Fandino, Bernhard Moriggl. Extended exposure of the petroclival junction: The combined anterior transpetrosal and subtemporal/transcavernous approach. 24-Dec-2018;9:259
How to cite this URL: Alexander Spiessberger, Fabian Baumann, Alexandra Stauffer, Serge Marbacher, Karl F. Kothbauer, Javier Fandino, Bernhard Moriggl. Extended exposure of the petroclival junction: The combined anterior transpetrosal and subtemporal/transcavernous approach. 24-Dec-2018;9:259. Available from: http://surgicalneurologyint.com/surgicalint-articles/9147/
Abstract
Background:The combined anterior transpetrosal and subtemporal/transcavernous (atsta) approach to the petroclival junction provides a wide exposure facilitating resection of large tumor lesions such as petroclival mengiomas, chondrosarcomas, or chordomas. In this article we provide technical instructions on the approach with anatomical consideration and a literature review of previous applications of this approach.
Methods:The combined approach was performed in two cadaveric specimen and relevant anatomical aspects were studied. Additionally, the authors performed a review of the literature focusing on indications, neurologic outcome, and complications associated with the technique.
Results:A combined atsta approach offers a wide exposure of the crus cerebrum, pons, basal temporal lobe, cranial nerves III to VII/VIII, posterior cerebral artery (PCA), superior cerebellar artery (SCA), basilar artery (BA), anterior inferior cerebellar artery (AICA), and posterior communicating artery (Pcom). It has been successfully applied with acceptable morbidity and mortality rates, mainly for (spheno-) petroclival meningiomas.
Conclusion:The combined approach studied here is a useful skull base approach to the petroclival junction and can be applied to treat large or complex pathologies of the region. Detailed anatomical knowledge is essential.
Keywords: Anterior petrosal approach, petroclival junction, subtemporal/transcavernous approach
INTRODUCTION
The junction between petrous apex and clivus is difficult to expose because of its deep and secluded location. Surgery in this area is complicated by the close proximity of important nervous and vascular structures, such as the basilar artery (BA) and its branching vessels, brainstem, and cranial nerves.[
In the past, the following approaches have been developed to treat lesions of the petroclival junction: Dolenc's, Hakuba's, and Kawase's interdural approach to the parasellar area, the cavernous sinus, its lateral wall, and Meckel's cave;[
All these techniques expose only a part of the entire anatomic region, however, in cases requiring a wider exposure a combined technique can be applied: the anterior transpetrosal and subtemporal/transcavernous (atsta) approach.
In this study, we aim to demonstrate the surgical techniques of the “atsta” approach and its anatomic exposure based on dissection on two human cadaver heads. Further we provide a review of published surgical case series applying the combined approach to discuss surgical outcome and possible complication.
MATERIALS AND METHODS
For this study two human cadaver heads were used to demonstrate the surgical anatomy and exposure of the combined “atsta” approach to the petroclival junction. The cadaver specimens were donated by people who had given informed consent for their use for scientific and educational purposes prior to death to the Medical University of Innsbruck, Division of Clinical and Functional Anatomy.[
The two cadaver heads (one male aged 75 years and one female aged 82 years) showed no gross anatomic distortions, however, the cause of death was unknown to us. Preservation was performed in accordance with the institute's standard.[
We used standard neurosurgical instruments, including bone drill, microinstruments, and microscope. All surgical steps were documented using a digital camera.
RESULTS
The surgical technique outlined in this article consists of a temporal craniotomy with transzygomatic extension and extradural anterior transpetrosal approach, as first described by Kawase et al.[
Each specimen was fixed in a head holder, 45° rotation to the contralateral side and moderate head tilt. A question mark skin incision was performed and a two layer skin/galea and muscle flap was elevated. To protect the frontal branches of the facial nerve the superficial temporal fascia was incised posterior to the interfascial fat pad vertically all the way from the superior temporal line until below the zygomatic arch. The superficial fascial layer and interfascial fat pad were dissected off the deep fascial layer and reflected anteriorly. Over the zygomatic arch the superficial temporal fascia is adherent with the bone and has to be peeled off thoroughly to protect the facial nerve branches. An osteotomy of the zygomatic arch is performed using a small side cutting burr and avoiding violation of the temporomandibular joint capsule at the root of the zygoma. After completion of the osteotomy the zygomatic arch remains in situ and is not freed from the masseteric muscle attachments at its inferior aspect. The temporalis muscle now is incised parallel to the superior temporal line leaving a small cuff for later re-fixation. Posteriorly the muscle was incised parallel to the course of muscle fibers and finally elevated off the temporal squama in a subperiostial fashion. Anteriorly sharp dissection is used to detach the muscle insertions from the bone around the frontozygomatic suture. Once the muscle has been mobilized it is reflected inferiorly together with the mobile zygomatic arch. By performing this maneuver the temporalis muscle bulk can be reflected into the gap that was created by the zygomatic arch osteotomy. The downward flected muscle is now flush with the middle cranial fossa. Now a large temporal craniotomy was performed extending up to the Sylvian fissure. After far downward flection of the temporalis muscle the craniotomy can be performed all the way anteriorly to the frontozygomatic sutureline and inferiorly to the middle fossa floor. After elevation of the bone flap a high speed diamond drill is used to drill off the remaining few millimeters of the temporal squama until flush with the middle cranial fossa. At this point the operating microscope is brought into the field.
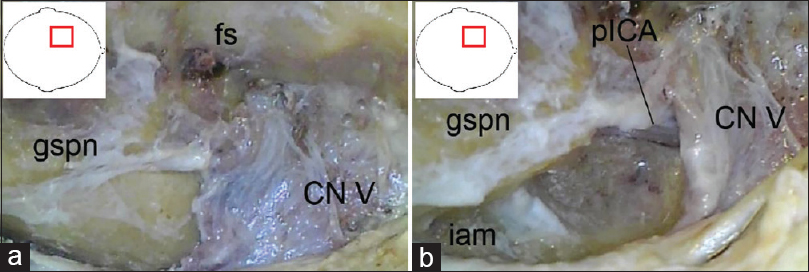
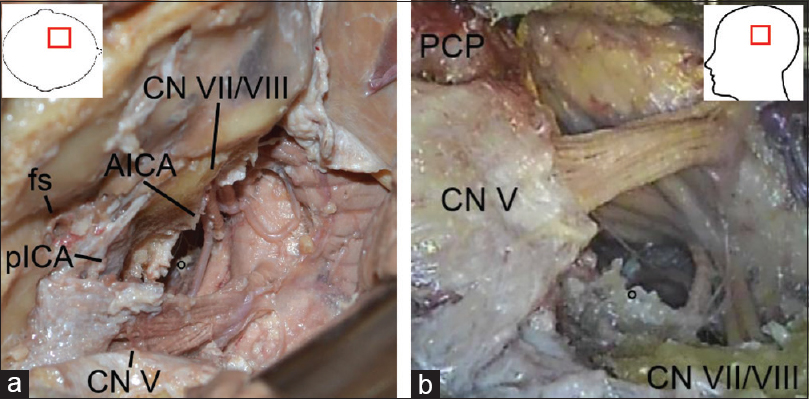
The dura was now peeled off the middle fossa using a dissector under preservation of the greater superficial petrosal nerve, see
Figure 2
(original) a: Left-sided anterior transpetrosal approach, intradural stage with final exposure of the paramedian brainstem and surrounding structures: AICA, anterior inferior cerebellar artery; CN V and VII/VIII, cranial nerve V and VII/VIII; fs, foramen spinosum; pICA, petrous segment of the internal carotid artery. b: Left-sided anterior transpetrosal approach with transtentorial/transcavernous extension, intradural stage with final exposure of the median brainstem: CN V and VII/VIII, cranial nerve V and VII/VIII; PCP, posterior clinoid process
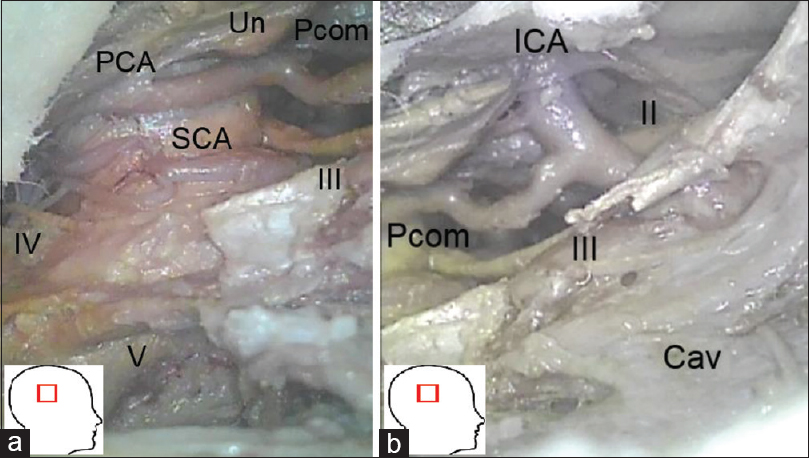
At this point the subtemporal corridor is further expanded. The dural incision is completed across the tentorium under preservation of the fourth cranial nerve at the level of the tentorial free edge. The tentorium and inner most dural layer is now peeled off the lateral wall of the cavernous sinus under the protection of the entry points of the third and fourth cranial nerves into the cavernous sinus wall (a small piece of tentorium was left in place covering those entry points).
Figure 3
(original): a and b: Right-sided subtemporal/transcavernous/transtentorial exposure of the petroclival junction: CN III, IV, V, cranial nerves III, IV, and V; Cav, lateral wall of the cavernous sinus; PCA, posterior cerebral artery; Pcom, posterior communicating artery; SCA, superior cerebral artery; Un, uncus
In summary the combined “atsta” approach exposes the areas and structures outlined in Figures
DISCUSSION
The “atsta” approach provides a wide anterolateral exposure of the brainstem, cerebral peduncles and adjacent neurovascular structures from the level of the internal auditory canal caudally to the level of the anterior clinoid process cranially, see
Aoyagi et al. applied the technique on seven patients with tumor lesions of the petroclival junctional area.[
Pathology-specific considerations
Meningiomas are frequent neoplasms that can be found in 3% of autopsies in patients >60 years and comprise 15% of intracranial neoplasms.[
Complete surgical resection, unlike radiosurgery relieves mass effects on adjacent structures and offers the possibility of long-term cure. It is well established that a complete resection leads to a significantly lower recurrence rate of 3–4% compared to a recurrence rate of 10–25% in case of subtotal resection. However, surgery of such lesions frequently can carry considerable risks. In a large systematic review including 1000 patients with petroclival menigiomas undergoing surgical treatment the rate of new cranial nerve deficits was 34% after surgery.[
For the rare clival chordomas and sphenopetroclival chondrosarcomas (incidence 0.02/100 000 per year) the effect of “extend of resection” on “progression-free survival” and “overall survival” are not well established.[
Potential Complications of the “atsta” approach
Temporal lobe retraction carries the risk of brain contusion. It has been shown that the temporal upward retraction in subtemporal approaches is more complication prone than a postero-lateral retraction, as applied with a pretemporal approach.[
A further potential complication can occur depending on the course of the superficial Sylvian vein. In some individuals this major vein runs along the base of the temporal lobe, rather than draining into the sphenoparietal sinus.[
The third and fourth cranial nerves are at particular risk during this combined “atsta” approach. Separating the dura from the lateral cavernous sinus wall poses a risk for postoperative dysfunction of the third and fourth cranial nerve.
CONCLUSION
The combined “atsta” approach studied in this article is a useful skull base approach to the petroclival junction and can be applied to treat large or complex pathologies such as sphenopetroclival meningiomas, chordomas, or chondrosarcomas. The technique enables the surgeon to achieve gross total resection in a significant proportion of patients. Even though cranial nerve deficits (especially cranial nerves III and IV) can be potential complications, the literature shows that these are temporary in a substantial proportion of patients.
Abbreviations
AICA anterior inferior cerebellar artery
atsta anterior transpetrosal and subtemporal/transcavernous
BA basilar artery
PCA posterior cerebral artery
Pcom posterior communicating artery
SCA superior cerebellar artery
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
The authors thank the individual who donated his body and tissues for the advancement of education and research.
References
1. Al-Mefty O, Ayoubi S, Gaber E. Trigeminal schwannomas: Removal of dumbbell-shaped tumors through the expanded Meckel cave and outcomes of cranial nerve function. J Neurosurg. 2002. 96: 453-63
2. Aoyagi M, Kawano Y, Tamaki M, Tamura K, Ohno K. Combined extradural subtemporal and anterior transpetrosal approach to tumors located in the interpeduncular fossa and the upper clivus. Acta Neurochir. 2013. 155: 1401-7
3. Bohman LE, Koch M, Bailey RL, Alonso-Basanta M, Lee JY. Skull base chordoma and chondrosarcoma: Influence of clinical and demographic factors on prognosis: A SEER analysis. World Neurosurg. 2014. 82: 806-14
4. Castellano F, Ruggiero G. Meningiomas of the posterior fossa. Acta Radiol Suppl. 1953. 104: 1-177
5. Chanda A, Nanda A. Retrosigmoid intradural suprameatal approach: Advantages and disadvantages from an anatomical perspective. Neurosurgery. 2006. 59: ONS1-
6. Colasanti R, Tailor AR, Zhang J, Ammirati M. Functional petrosectomy via a suboccipital retrosigmoid approach: Guidelines and topography. World Neurosurg. 2016. 87: 143-54
7. Diluna ML, Bulsara KR. Surgery for petroclival meningiomas: A comprehensive review of outcomes in the skull base surgery era. Skull Base. 2010. 20: 337-42
8. Dolenc VV, Skrap M, Sustersic J, Skrbec M, Morina A. A transcavernous-transsellar approach to the basilar tip aneurysms. British J Neurosurg. 1987. 1: 251-9
9. el-Kalliny M, van Loveren H, Keller JT, Tew JM. Tumors of the lateral wall of the cavernous sinus. JNeurosurg. 1992. 77: 508-14
10. Fournier HD, Mercier P, Roche PH. Surgical anatomy of the petrous apex and petroclival region. Adv Tech Stand Neurosur. 2007. 32: 91-146
11. Fournier HD, Mercier P, Velut S, Reigner B, Cronier P, Pillet J. Surgical anatomy and dissection of the petrous and peripetrous area. Anatomic basis of the lateral approaches to the skull base. Surg Radiol Anat. 1994. 16: 143-8
12. Giliberto G, Lanzino DJ, Diehn FE, Factor D, Flemming KD, Lanzino G. Brainstem cavernous malformations: Anatomical, clinical, and surgical considerations. Neurosurg Focus. 2010. 29: E9-
13. Hakuba A, Liu S, Nishimura S. The orbitozygomatic infratemporal approach: A new surgical technique. Surg Neurol. 1986. 26: 271-6
14. Harsh GR, Sekhar LN. The subtemporal, transcavernous, anterior transpetrosal approach to the upper brain stem and clivus. JNeurosurg. 1992. 77: 709-17
15. Hu P, Liang J, Bao Y, Li M, Ling F. The pterional transsylvian transtentorial approach to ventrolateral pontine cavernomas: Indications and techniques. World Neurosurg. 2014. 82: 1276-82
16. Ikushima I, Korogi Y, Kitajima M, Yamura M, Yamashita Y. Evaluation of drainage patterns of the major anastomotic veins on the lateral surface of the cerebrum using three-dimensional contrast-enhanced MP-RAGE sequence. Eur J Radiol. 2006. 58: 96-101
17. Kawase T, Shiobara R, Toya S. Anterior transpetrosal-transtentorial approach for sphenopetroclival meningiomas: Surgical method and results in 10 patients. Neurosurgery. 1991. 28: 869-
18. Kawase T, Toya S, Shiobara R, Mine T. Transpetrosal approach for aneurysms of the lower basilar artery. JNeurosurg. 1985. 63: 857-61
19. Kawase T, van Loveren H, Keller JT, Tew JM. Meningeal architecture of the cavernous sinus: Clinical and surgical implications. Neurosurgery. 1996. 39: 527-
20. Kobayashi M, Yoshida K, Kawase T. Inter-dural approach to parasellar tumors. Acta Neurochir. 2010. 152: 279-
21. Little KM, Friedman AH, Sampson JH, Wanibuchi M, Fukushima T. Surgical management of petroclival meningiomas: Defining resection goals based on risk of neurological morbidity and tumor recurrence rates in 137 patients. Neurosurgery. 2005. 56: 546-
22. Matsushima K, Komune N, Matsuo S, Kohno M. Microsurgical and endoscopic anatomy for intradural temporal bone drilling and applications of the electromagnetic navigation system: Various extensions of the retrosigmoid approach. World Neurosurg. 2017. 103: 620-30
23. Nakasu S, Hirano A, Shimura T, Llena JF. Incidental meningiomas in autopsy study. Surg Neurol. 1987. 27: 319-22
24. Platzer W, Putz R, Poisel S. [New system for the preservation and storage of anatomical matter]. Acta Anat (Basel). 1978. 102: 60-7
25. Riederer BM, Bolt S, Brenner E, Bueno-López JL, Circulescu ARM, Davies DC. The legal and ethical framework governing body donation in Europe – 1st update on current practice. Eur J Anat. 2012. 16: 1-21
26. Roche PH, Lubrano VF, Noudel R. How I do it: Epidural anterior petrosectomy. Acta Neurochir. 2011. 153: 1161-7
27. Samii M, Carvalho GA, Tatagiba M, Matthies C, Vorkapic P. Meningiomas of the tentorial notch: Surgical anatomy and management. JNeurosurg. 1996. 84: 375-81
28. Shibao S, Toda M, Orii M, Fujiwara H, Yoshida K. Various patterns of the middle cerebral vein and preservation of venous drainage during the anterior transpetrosal approach. JNeurosurg. 2016. 124: 432-9
29. Spetzler RF, Daspit CP, Pappas CT. The combined supra- and infratentorial approach for lesions of the petrous and clival regions: Experience with 46 cases. JNeurosurg. 1992. 76: 588-99
30. Starke R, Kano H, Ding D, Nakaji P, Barnett GH, Mathieu D. Stereotactic radiosurgery of petroclival meningiomas: A multicenter study. J Neurooncol. 2014. 119: 169-76
31. Taha JM, Tew JM, van Loveren HR, Keller JT, el-Kalliny M. Comparison of conventional and skull base surgical approaches for the excision of trigeminal neurinomas. JNeurosurg. 1995. 82: 719-25
32. Tripathi M, Deo RC, Suri A, Srivastav V, Baby B, Kumar S. Quantitative analysis of the Kawase versus the modified Dolenc-Kawase approach for middle cranial fossa lesions with variable anteroposterior extension. JNeurosurg. 2015. 123: 14-22
33. Tzortzidis F, Elahi F, Wright DC, Temkin N, Natarajan SK, Sekhar LN. Patient outcome at long-term follow-up after aggressive microsurgical resection of cranial base chondrosarcomas. Neurosurgery. 2006. 58: 1090-
34. Van Havenbergh T, Carvalho G, Tatagiba M, Plets C, Samii M. Natural history of petroclival meningiomas. Neurosurgery. 2003. 52: 55-
35. Yoneoka Y, Tsumanuma I, Fukuda M, Tamura T, Morii K, Tanaka R. Cranial base chordoma--long term outcome and review of the literature. Acta Neurochir. 2008. 150: 773-8
36. Zador Z, Lu DC, Arnold CM, Lawton MT. Deep bypasses to the distal posterior circulation: Anatomical and clinical comparison of pretemporal and subtemporal approaches. Neurosurgery. 2010. 66: 92-100