- Department of Neurosurgery, University of Rochester, Strong Memorial Hospital, Rochester, New York, United States.
- Department of Neuropathology, University of Rochester, Strong Memorial Hospital, Rochester, New York, United States.
Correspondence Address:
Gabrielle Santangelo, Department of Neurosurgery, University of Rochester, Strong Memorial Hospital, Rochester, New York, United States.
DOI:10.25259/SNI_750_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Gabrielle Santangelo1, Jonathan Stone1, Mahlon Johnson2, Kevin Walter1. Lumbar cistern Candida intradural abscess following epidural anesthesia. 23-Sep-2022;13:435
How to cite this URL: Gabrielle Santangelo1, Jonathan Stone1, Mahlon Johnson2, Kevin Walter1. Lumbar cistern Candida intradural abscess following epidural anesthesia. 23-Sep-2022;13:435. Available from: https://surgicalneurologyint.com/surgicalint-articles/11883/
Abstract
Background: This report describes a case of an immunocompetent patient with an intradural abscess from Candida dubliniensis. The majority of fungal spine infections, although rare in general, are due to Aspergillus or C. albicans through systemic fungemia. To date, there have only been two reports of spondylodiscitis from C. dubliniensis.
Case Description: A 37-year-old immunocompetent female patient presented to the neurosurgical service for worsening headaches with nausea, vomiting, vision changes, and weight loss. MRI studies showed diffuse leptomeningeal enhancement of the distal spinal cord, conus medullaris, and nerve roots of the cauda equina extending beyond the neural foramina bilaterally. She had persistent symptoms and no clear diagnosis on lumbar puncture or systemic testing therefore L5-S1 laminectomy for an intradural tissue biopsy was performed. During surgery, cultures were taken and grew colonies of C. dubliniensis.
Conclusion: This organism has been reported rarely in the literature as being an infectious agent, thus diagnosing remains a challenge but should be considered in patients with a suggestive history.
Keywords: Candida dubliniensis, Cauda equina, Intradural abscess, Spinal abscess
INTRODUCTION
In this report, the investigators describe a patient with an intradural abscess comprised Candida dubliniensis in an intravenous drug user following epidural anesthesia. This is the third report of C. dubliniensis causing intradural extramedullary spinal infection. However, this case differs from the previous reports in many aspects. This is the first case of Candida dubliniensis extramedullary spine infection occurring in the context of a confirmed competent immune system. The patient is a female in her third decade of life with no clear immunodeficiency, a positive history for intravenous drug use, and intrapartum epidural anesthesia coinciding with the onset her symptoms. The diagnosis and management are described in detail.
CASE REPORT
History and examination
A 37-year-old female patient who received epidural anesthesia during childbirth presented to the neurosurgical service eleven months postpartum for worsening headaches with nausea, vomiting, vision changes, and weight loss. Here, a medical history was significant for peripheral neuropathies, unspecified mood disorder, herpes labialis, hepatitis C (HCV), intravenous substance abuse (including heroin and cocaine, now on buprenorphine), and noncompliance. Her medications before admission included gabapentin 300 mg daily, buprenorphine 2 mg daily, and topiramate 25 mg daily. She was never treated for hepatitis.
Delivery of her child the following month was uncomplicated, but she did require a second epidural injection during labor. After birth of her child, she had persistent headaches that were suspected to be secondary to a cerebrospinal fluid leak. She underwent epidural blood patch to treat the suspected leak, but her symptoms did not resolve. She began having shooting pain in her back that radiated into her legs. She described the headache as generalized, dull, nonradiating, and constant with a gradual onset that “worsen with laying down or standing up” and endorsed accompanying photophobia.
She had papilledema with diplopia, mild bilateral cranial nerve VI palsy, and decreased visual acuity. At this point, she was seen by ophthalmology and was admitted to the hospital. A lumbar puncture (LP) showed mildly elevated nucleated cell count of 179 with 50% PMNs, 42% lymphocytes, and 6% monocytes. Glucose was low at 36 mg/dL (serum glucose was 92 mg/dL) and protein was elevated at 287 mg/dL, consistent with a meningitis process and suggestive of bacterial meningitis. However, her CSF cultures were negative for histoplasma, tuberculosis, AFB, Cryptococcus, cytospin, fungus, and viruses. Additional testing showed HIV negativity, hepatitis A and C positivity and negative serology for listeria, Brucella, VDRL, syphilis, and lyme.
Her general chemistry panel on admission and throughout her hospital stay was normal. Liver function tests, CRP, IgG, and thrombocyte count were unremarkable. She was negative for antinuclear antibody, anti-LA/anti-RO, and antineutrophil cytoplasmic antibodies. Her CRP was normal with a mildly elevated ESR at the time of presentation in April, consistent with a largely resolved inflammatory process.
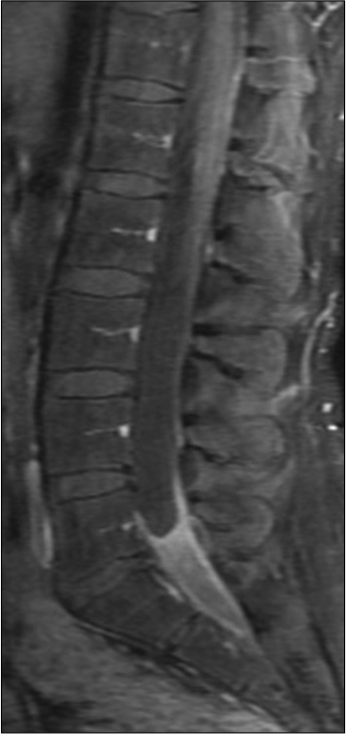
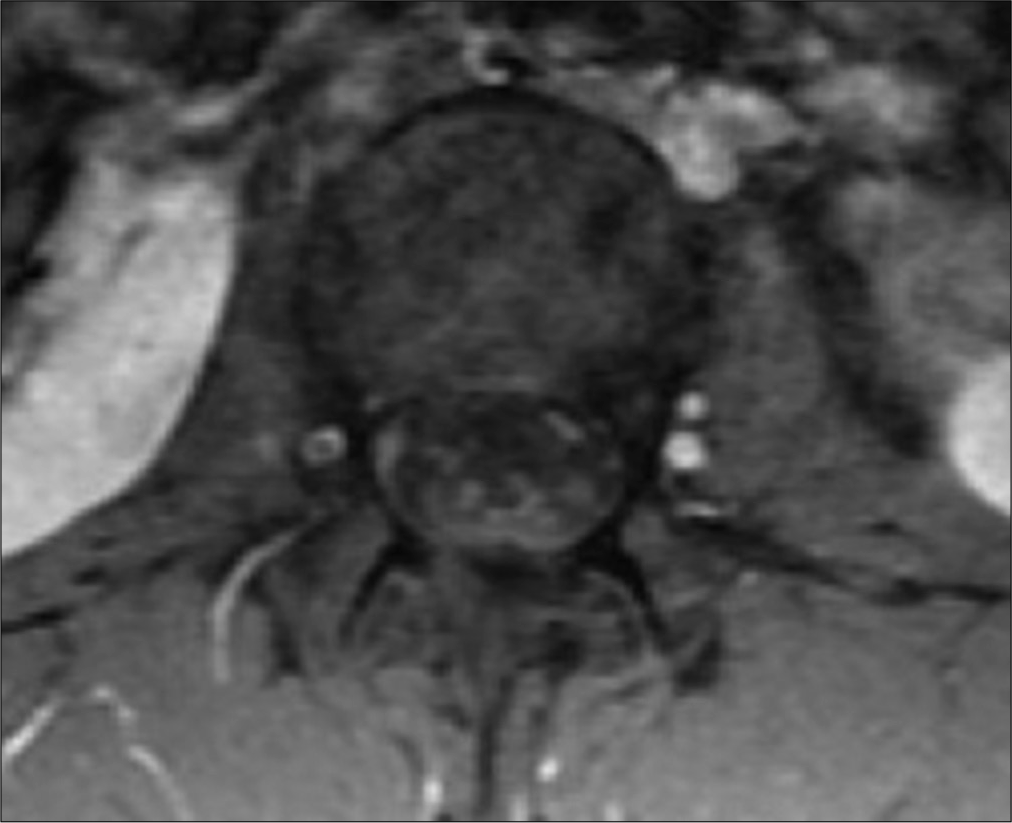
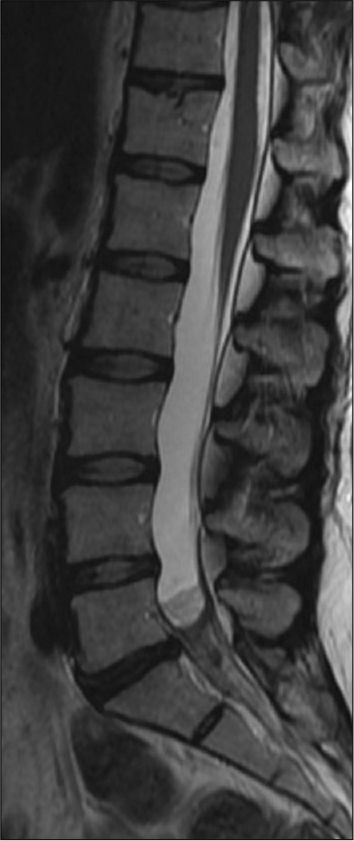
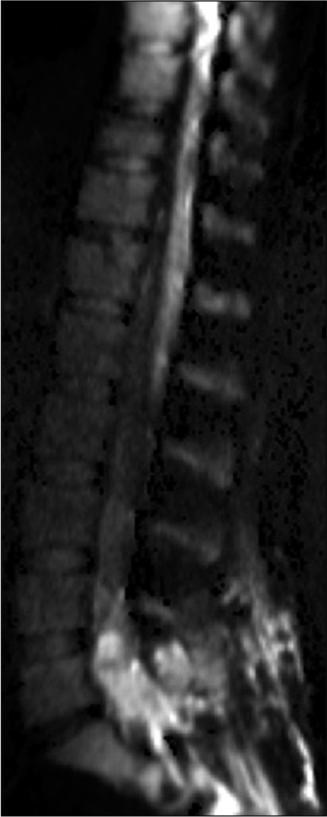
MRI studies showed diffuse leptomeningeal enhancement of the distal spinal cord, conus medullaris, and nerve roots of the cauda equina extending beyond the neural foramina bilaterally in T1 contrasted images [
Infectious disease was consulted and suggested chronic neutrophilic meningitis in the setting of an underlying autoimmune/inflammatory condition given the nature of her pain and her complicated medical history. She was discharged after 1 week on methylprednisolone (1 g/day for 5 days) that resulted in a near resolution of her back pain, headaches, and vision changes except for when she missed doses. She had no fevers, chills, or night sweats but reported a 10 lb weight loss with poor appetite. Her steroids were tapered to 50 mg, daily and when the dose was tapered, she had symptom return.
Infectious disease continued following and a lack of resolution of her pain prompted additional screening. Repeat MRI at a month post-partum showed no significant changes from the previous scan and continued leptomeningeal enhancement of the distal spinal cord and cauda equina nerve roots consistent with arachnoiditis. Her second LP showed a worsening inflammatory picture with 2682 nucleated cells (94% PMNs), low glucose at 26, elevated protein at 297, and 13 bands. The patient’s WBC was elevated at 23.2 from 16.4 three months prior although she remained afebrile. Her back pain rating at this time was 6–7/10.
Operation
Given her persistent symptoms and lack of diagnosis, she was scheduled for an L5-S1 laminectomy for an intradural tissue biopsy. During surgery, cultures were taken at every level of the exposure. After the laminectomy was complete, the epidural space appeared normal, but the dura was thickened and difficult to open inferiorly. The dura incision was carried cranially and once the subarachnoid space was entered, a copious amount of white, soft purulence material was released. The nerve roots were adherent to surrounding dura and to each other. There were no obvious signs of a capsule; however, the infection was likely loculated based on its appearance and indolent course. The intradural compartment was irrigated until the CSF ran clear. There were no intraoperative complications and she was started on postoperative antibiotics, including 500 mg metronidazole q8 h, 1 g vancomycin q12 h, IV fluconazole 400 mg, daily, and 2 g cefepime q12 h. After the procedure, the patient remained afebrile, without aberration in vitals, and was ambulatory.
Pathological findings
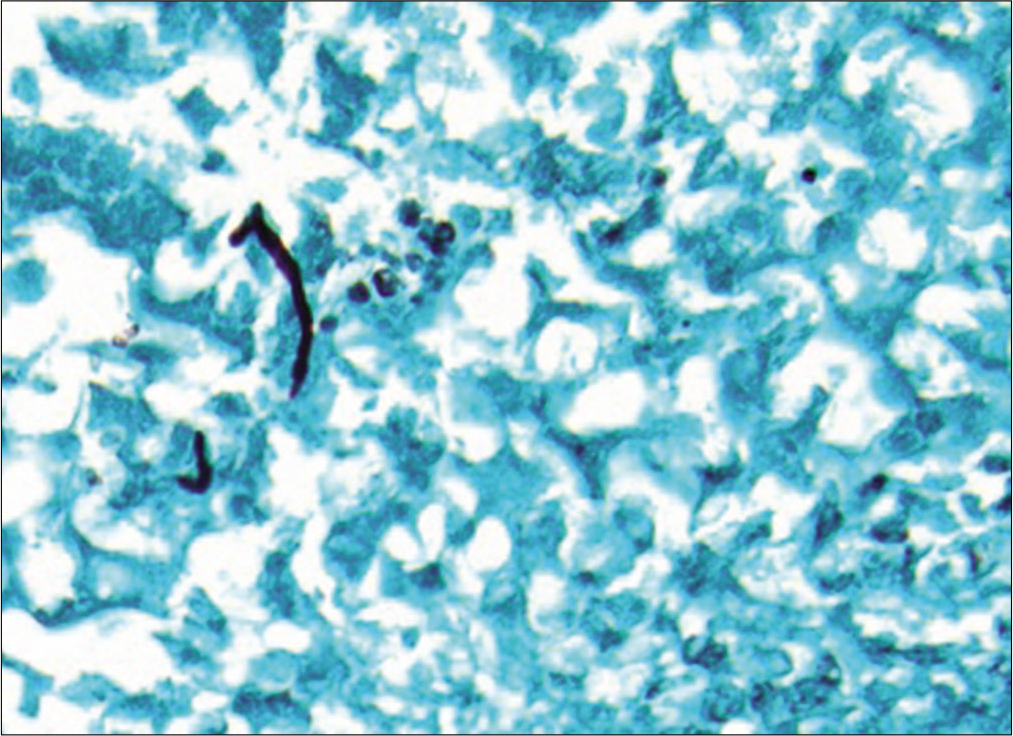
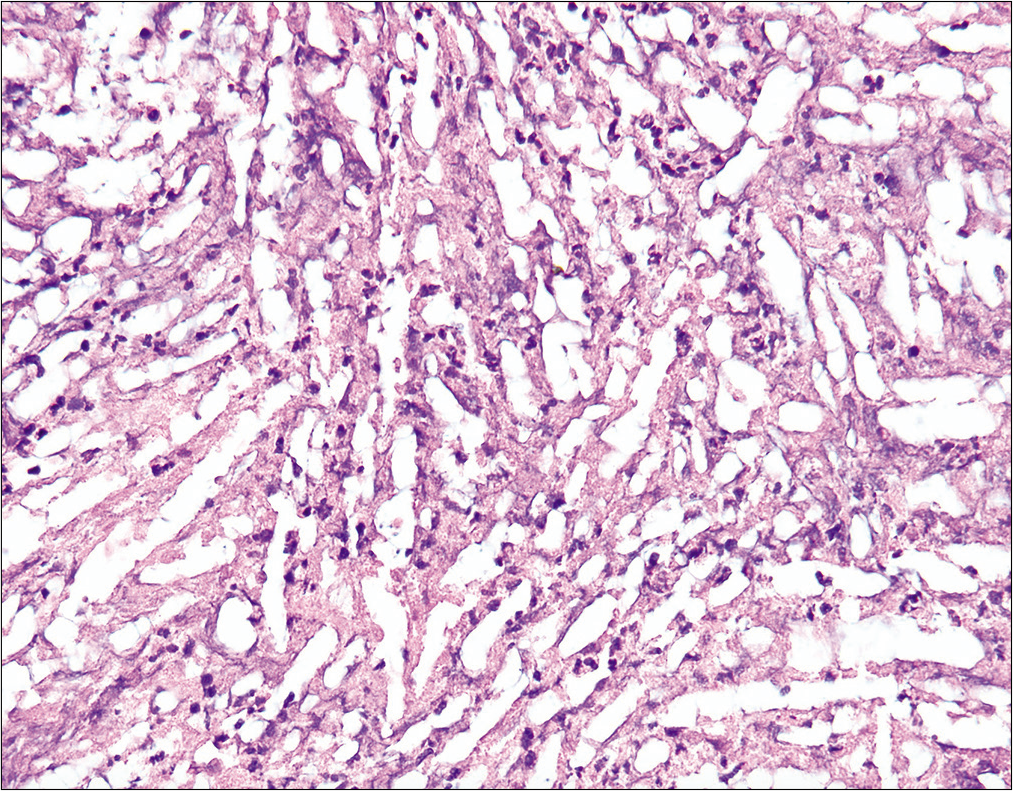
The histology showed rare nonseptate pseudohyphae and blastospores without a broad-based bud, indicative of a Candida species [
Postoperative course
Cefepime, metronidazole, vancomycin, and IV fluconazole were discontinued and the patient was started on 400 mg fluconazole PO daily (rather than outpatient peripherally inserted central catheter due to IV drug abuse) for a planned minimum of 4 weeks. After surgery, she endorsed only mild low back pain at the incision site and with position changes.
At 3-week postoperative follow-up, her back pain was worsening and the operative site was infected with induration of the surrounding skin but remained afebrile. She reported a significant amount of purulent drainage produced from her incision, which resulted in improvement of her symptoms. She was started on cephalexin. She is currently scheduled for follow-up with infectious disease and for a repeat MRI of her lumbar spine.
Of note, throughout the patient’s entire hospital course and at her follow-up appointments, she remained without neurological deficit. Her mental status examination, cranial nerves, strength, sensory perception, coordination, and reflexes were all within normal limits and without significant change at any time point.
DISCUSSION
The majority of fungal spine infections, although rare in general, are due to Aspergillus or C. albicans through a systemic fungemia. To date, there have only been two reports of spondylodiscitis from C. dubliniensis.[
To accurately diagnose and plan for intervention for intradural abscess, it has been shown that MRI with DWI is the most useful imaging to obtain, particularly in evaluating for dissemination.[
While both of the previous reports described a hematogenous spread of the infection, the patient in the present case report showed no systemic involvement of the fungus.[
The etiology of the fungus remains unclear in this case. While Candida dubliniensis has been reported as an oral colonizer in HIV-positive patients, it is unclear how this fungus was introduced into this patient.[
The complication of soft-tissue infection at the surgical site may be due to potential seeding during closure. The maintenance of intact neurological examination without focal findings is understandable considering the location of the abscess and the lack of fungal foci within the brain. However, this may be misleading and falsely reassuring without functional deficit as to the severity of infection.
Management of these cases is not clearly delineated in the literature. It is believed that treatment after diagnosis is relatively simple given limited involvement of skeletal structures and involves a 2–4 week treatment of fluconazole with following amphotericin b if not yet resolved.[
CONCLUSION
This report described an indolent course of intradural C. dubliniensis infection in an immunocompetent woman. This pathogen is most frequently seen in cases of oral candidiasis, candidemia, and endophthalmitis and spinal involvement is extremely rare.[
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Cheon JE, Yang HJ, Chung YN, Park SB. Pyogenic intradural abscess of lumbar spine : A case report. 2015. 11: 18-21
2. Chia SL, Tan BH, Tan CT, Tan SB. Candida spondylodiscitis and epidural abscess: Management with shorter courses of anti-fungal therapy in combination with surgical debridement. J Infect. 2005. 51: 17-23
3. Kauffman CA, Malani AN. Fungal infections associated with contaminated steroid injections. Microbiol Spectr. 2016. 4: 1-13
4. Kulkarni AG, Chu G, Fehlings MG. Pyogenic intradural abcess: A case report. Spine (Phila Pa 1976). 2008. 32: E354-7
5. Moritani T, Kim J, Capizzano AA, Kirby P, Kademian J, Sato Y. Pyogenic and non-pyogenic spinal infections: Emphasis on diffusion-weighted imaging for the detection of abscesses and pus collections. Br J Radiol. 2014. 87: 20140011
6. Oksi J, Finnilä T, Hohenthal U, Rantakokko-Jalava K. Candida dubliniensis spondylodiscitis in an immunocompetent patient. Case report and review of the literature. Med Mycol Case Rep. 2014. 3: 4-7
7. Rowe IF, Wright ED, Higgens CS, Burnie JP. Intervertebral infection due to Candida albicans in an intravenous heroin abuser. Ann Rheum Dis. 1988. 47: 522-5
8. Saigal G, Post MJ, Kozic D. Thoracic intradural Aspergillus abscess formation following epidural steroid injection. Am J Neuroradiol. 2004. 25: 642-4
9. Salzer HJ, Rolling T, Klupp EM, Schmiedel S. Hematogenous dissemination of Candida dubliniensis causing spondylodiscitis and spinal abscess in a HIV-1 and HCV-coinfected patient. Med Mycol Case Rep. 2015. 8: 17-20
10. Singh N, Perfect JR. Immune reconstitution syndrome and exacerbation of infections after pregnancy. Clin Infect Dis. 2007. 45: 1192-9