- Department of Surgical Specialties and Neurosurgery, Pedro Ernesto University Hospital, Rio de Janeiro,
- Laboratory of Microneurosurgical Anatomy, BP-A Beneficência Portuguesa de São Paulo, São Paulo, Brazil.
Correspondence Address:
Magno Rocha Freitas Rosa, Department of Surgical Specialties and Neurosurgery, Pedro Ernesto University Hospital, Rio de Janeiro, Brazil.
DOI:10.25259/SNI_232_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Magno Rocha Freitas Rosa1, Flavio Nigri1, Vanessa Milanese Holanda Zimpel2, Mateus Reghin Neto2. Middle fossa triangles – A step-by-step dissection. 28-Apr-2023;14:150
How to cite this URL: Magno Rocha Freitas Rosa1, Flavio Nigri1, Vanessa Milanese Holanda Zimpel2, Mateus Reghin Neto2. Middle fossa triangles – A step-by-step dissection. 28-Apr-2023;14:150. Available from: https://surgicalneurologyint.com/surgicalint-articles/12302/
Abstract
Background: The anatomy and surgical approach to the cavernous sinus and the middle fossa can constitute a considerable challenge, specially for young surgeons. Although their surgical explorations have gone through a popular phase in the past, to this date, they remain an uncomfortable subject for many neurosurgeons. The aim of this paper is to systematically review its anatomy and multiple corridors through a step-by-step dissection of the middle fossa triangles, providing a roadmap for surgeons.
Methods: A step-by-step dissection of the cavernous sinus was performed in two fresh-frozen cadavers aiming to describe the anatomy of ten different middle fossa triangles, demonstrating the feasibility of the use of their spaces while surgically approaching this area.
Results: The intradural opening of the roof of the cavernous sinus was obtained by dissection of clinoidal, carotid-oculomotor, supratrochlear, optic-carotideal, and oculomotor triangles, allowing an expanded superior view. On the counterpart, the extradural exploration of the lateral wall through the middle fossa floor peeling exposed the infratrochlear, anteromedial, and anterolateral triangles. The middle fossa floor itself was the door to approaching posterior fossa through anterior petrosectomy. The dissection of each individual triangle can be amplified exponentially with exploration of its adjacents, providing broader surgical corridors.
Conclusion: The cavernous sinus still remains far from an “every man’s land,” but its systematic study based on direct approaches can ease the challenges of its surgical exploration, allowing surgeons to feel more comfortable with its navigation, with consequently benefit in the treatment of patients.
Keywords: Cavernous sinus, Microneuroanatomy, Middle fossa, Skull base, Triangles
INTRODUCTION
The groundbreaking works of Dolenc[
The description of the middle fossa and cavernous sinus triangles by the groups of Parkinson,[
As these triangles are carefully dissected, one can note that their initially rigid limits and narrow corridors, seen with skepticism, become progressively more flexible, and their content become significantly more extensive. Furthermore, since these structures are adjacent to one another, the exploration of one triangle can exponentially increase the avenue of the next, creating an unforeseen working space for the surgeon.
MATERIALS AND METHODS
The study of the middle fossa was performed in a fresh-frozen injected cadaver specimen and was approved by the institutional research board.
RESULTS
The dissection was photographically registered step-by-step to demonstrate the approach to ten different middle fossa triangles, with documentation of the limits that compose them and their exploration regarding its contents and navigation. Different angles were included aiming to ease the anatomical study of the structures and the surgical corridors allowed by them. Initially, anterior clinoidectomy was performed intradurally and the triangles of the roof of cavernous sinus were dissected in a sequential manner. The middle fossa floor was then peeled away extradurally to demonstrate the lateral wall of cavernous sinus and progress with the corresponding triangles. Afterward, the middle fossa floor was explored and drilled in a way to address the structures present in the posterolateral and the posteromedial triangles.
The approaches to the middle fossa triangles were divided in those related to the roof of the cavernous sinus, its lateral wall and the middle fossa floor itself, aiming to allow a systematic review of the exploration, contents, and corridors they provide.
DISCUSSION
Roof of the cavernous sinus
The following triangles [
Figure 1:
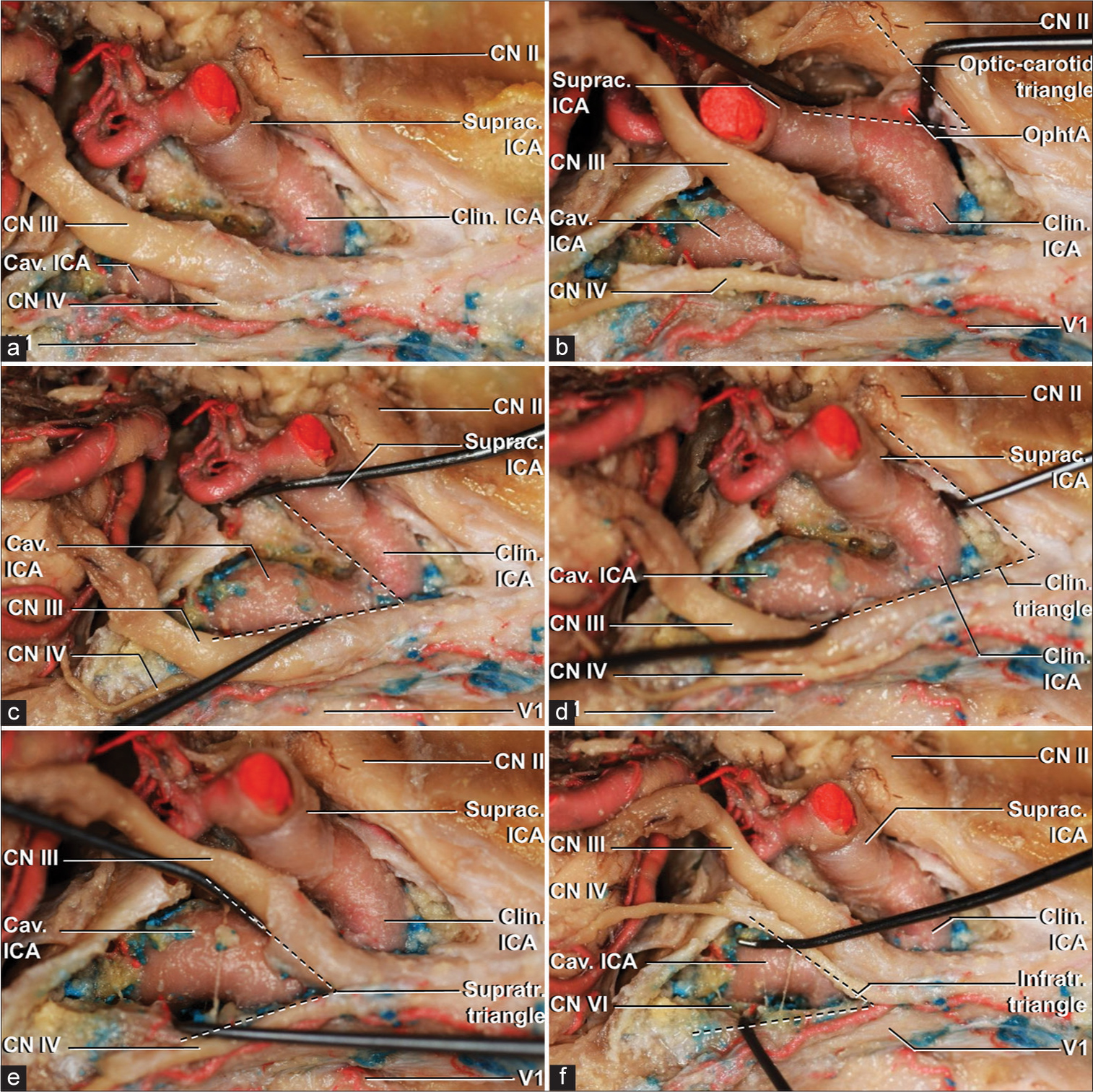
A superior view of a right cavernous sinus with a 30° clockwise rotation. (a) An overview of the roof of the cavernous sinus after complete clinoidectomy. (b) optic-carotid triangle, limited medially by the lateral border of CN II and laterally by the medial border of supraclinoid ICA. The emergence of the ophthalmic branch of ICA can be noted within the limits of the triangle. (c) the carotid-oculomotor triangle, formed by the junction of the lateral margin of supraclinoid ICA and medial margin of CN III. Within its limits, after opening the roof of the cavernous sinus, cavernous ICA can be demonstrated. (d) clinoidal triangle, marked by the lateral border of CN II and medial border of CN III, demonstrates the three segments composing the paraclinoid internal carotid artery – cavernous, clinoidal and ophthalmic segment of supraclinoid ICA. (e) the supratrochlear triangle is a narrow corridor located between the infero-lateral border of CN III and superomedial border of CN IV and can be used for exposition of the proximal horizontal portion of the cavernous segment of ICA. (f) infratrochlear or Parkinson’s triangle, composed by the junction of the inferior margin of CN IV and superior margin of V1 was dissected, demonstrating the posterior loop of cavernous ICA and the abducens nerve. Cav: Cavernous, Clin: Clinoidal, CN II: Optic nerve, CN III: Oculomotor nerve, CN IV: Trochlear nerve, ICA: Internal carotid artery, Infratr: Infratrochlear, OphtA: Ophthalmic artery, Suprac: Supraclinoid, Supratr: Supratrochlear, V1: Ophthalmic division of trigeminal nerve.
Figure 2:
A superolateral view of a right cavernous sinus with a 45º clockwise rotation. (a) an overview of the roof of the cavernous sinus after complete clinoidectomy. (b) optic-carotid triangle, limited medially by the lateral border of CN II and laterally by the medial border of supraclinoid ICA. The emergence of the ophthalmic branch of ICA can be noted within the limits of the triangle. (c) the carotid-oculomotor triangle, formed by the junction of the lateral margin of supraclinoid ICA and medial margin of CN III. Within its limits, after opening the roof of the cavernous sinus, cavernous ICA can be demonstrated. (d) clinoidal triangle, marked by the lateral border of CN II and medial border of CN III, demonstrates the three segments composing the paraclinoid internal carotid artery – cavernous, clinoidal, and ophthalmic segment of supraclinoid ICA. (e) the supratrochlear triangle is located between the infero-lateral border of CN III and superomedial border of CN IV and can be used for exposition of the proximal horizontal portion of the cavernous segment of ICA. (f), infratrochlear or Parkinson’s triangle, composed by the junction of the inferior margin of CN IV and superior margin of V1 was dissected, demonstrating the posterior loop of cavernous ICA and the abducens nerve. Cav: Cavernous, Clin: Clinoidal, CN II: Optic nerve, CN III: Oculomotor nerve, CN IV: Trochlear nerve, ICA: Internal carotid artery, Infratr: Infratrochlear, OphtA: Ophthalmic artery, Suprac: Supraclinoid, Supratr: Supratrochlear, V1: Ophthalmic division of trigeminal nerve.
Dolenc’s or clinoidal triangle
Described as “the anteromedial triangle” by Dolenc in 1989,[
The opening of cavernous sinus can then be performed by dissection of the carotid-oculomotor membrane, medial to CN III, and the cavernous segment of carotid artery in its distal horizontal and anterior loop is made evident. Therefore, the approach to this triangle is the only that allows exposure of three different segments of ICA – distal cavernous, clinoidal and ophthalmic segment of supraclinoid –, or, in other words, the entire paraclinoid carotid artery [
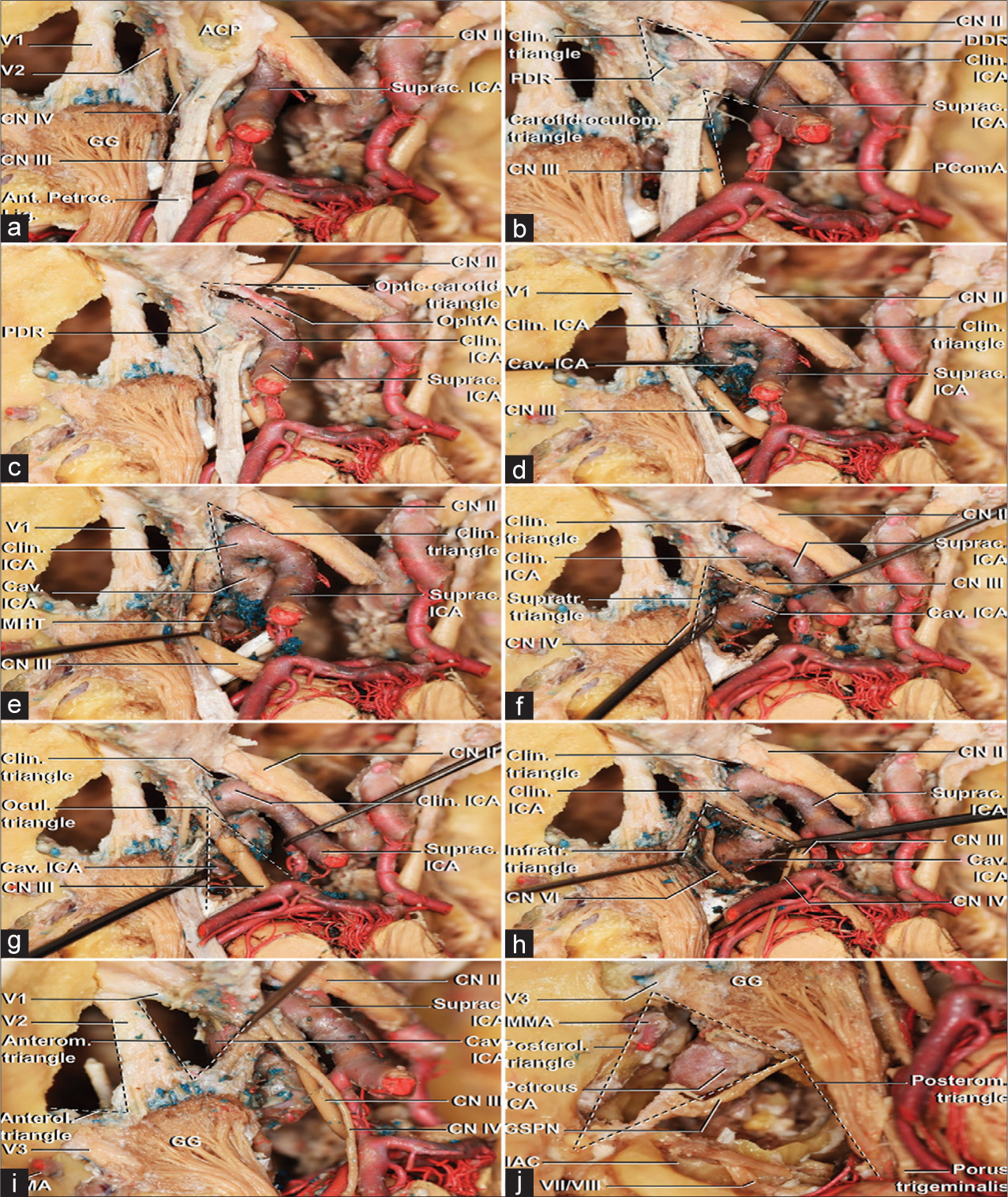
Figure 3:
A supero-lateral view of a left cavernous sinus with a 15° counter clockwise rotation through a step-by-step dissection. (a) An overview of the roof and lateral wall of the cavernous sinus after the middle fossa peeling was conducted and an anterior clinoidectomy was partially performed. (b) The anterior clinoid process removal was completed along with the opening of the dura propria covering the optic nerve. It is possible to visualize the proximal dural ring, also known as the carotid-oculomotor membrane, and the distal dural ring. Proximal to the PDR, the cavernous segment of ICA is present. Between PDR and DDR, the carotid is adjacent to the anterior clinoid process – the clinoidal segment. Distal to the DDR, the supraclinoid segment of ICA. The carotid-oculomotor triangle exposes the emergence of the posterior communicating artery. (c) Dissection of the optic-carotid triangle after section of the DDR demonstrated the initial course of the ophthalmic artery, inferior to the optic nerve. (d) With sectioning of the PDR, the cavernous sinus is opened medially to the oculomotor nerve. The anterior loop and distal horizontal portions of cavernous ICA are exposed. (e) Further dissection demonstrates the meningohypophyseal trunk emerging from cavernous ICA. (f) The supratrochlear triangle allows approaching the proximal horizontal portion of the cavernous segment of ICA. (g) The complete opening of the oculomotor or Hakuba’s triangle exposes the distal ascending and posterior loop of the cavernous ICA. (h) The dissection of the infratrochlear or Parkinson’s triangle, conversely, exposes the posterior loop of cavernous ICA, the origin of the meningohypophyseal trunk, and the abducens nerve through most of its intracavernous course. (i) The anteromedial triangle exposes the distal horizontal portion of the cavernous ICA. Bone drilling in both anteromedial or anterolateral triangles allows approaching sphenoid sinus. (j) Bone drilling adjacent to the posterolateral and posteromedial triangles expose the course of the petrous carotid artery from its entrance in the carotid canal to the passage under the petrolingual ligament and penetration of the cavernous sinus. Under the posterolateral triangle, formed by the junction of the GSPN and the lateral border of the gasserian ganglion and V3 until its entrance in the foramen ovale, Eustachian tube and tensor tympani are present. The posteromedial triangle relies adjacent to Glasscock’s area and it is limited by the gasserian ganglion and V3 anteromedially, GSPN anterolaterally and a line connecting the geniculate ganglion and petrous apex on the counterpart. Bone drilling within its limits, medial to the IAC, and lateral to the porus trigeminus, over the petrous apex, exposes the posterior fossa dura-mater. ACP: Anterior clinoid process, Ant: Anterior, Anterol: Anterolateral, Anterom: Anteromedial, Cav: Cavernous, Clin: Clinoidal, CN II: Optic nerve: CN III: Oculomotor nerve, CN IV, trochlear nerve, CN VII: Facial nerve, CN VIII: Vestibulocochlear nerve, GG: Gasserian ganglion, GSPN: Great superficial petrous nerve, IAC, Internal acoustic canal, ICA: Internal carotid artery, Infratr: Infratrochlear, Lat: Lateral, Lig: Ligament, MMA: Middle meningeal artery, OphtA: Ophthalmic artery, PComA: Posterior communicating artery, PDR: Proximal dural ring, Petroclin: Petroclinoid, Posterol: Posterolateral, Posterom: Posteromedial, Suprac: Supraclinoid, Supratr: Supratrochlear, V1: Ophthalmic division of trigeminal nerve, V2: Maxillary division of trigeminal nerve, V3: Mandibular division of trigeminal nerve.
The dissection allowed by this triangle, if associated with removal of the dura-mater and bone that constitutes the roof of optic canal, also allows medial and superior mobilization of optic nerve and removal of its dura própria on its inferolateral side, with ophthalmic artery becoming evident from its origin – regardless if it ascends from supraclinoid, clinoidal, or cavernous segment of ICA – to its entrance in the optic canal, with an expansion of the opticocarotid triangle.[
The exploration of the clinoidal triangle, therefore, constitutes a versatile and frequently useful strategy, allowing better manipulation of the carotid artery, CN, and optic nerve and the treatment of a range of pathologies involving these structures.
The removal of anterior clinoidal process also improves the surgeon’s angle of vision, facilitating other surgical maneuvers, such as posterior clinoidectomy (by ampliation of carotidoculomotor triangle) and middle fossa peeling. Hence, it constitutes a keystone in approaching the cavernous sinus by both its superior and lateral walls as far as posterior fossa.
Supratrochlear or paramedial triangle
The supratrochlear triangle, as the name suggests, is formed by the union of the superomedial border of the trochlear nerve (CN IV) and inferolateral border of the CN III, limited posteriorly by a line connecting the point of piercing of both nerves into dura-mater.[
This narrow triangle, therefore, is located within the superolateral wall of the cavernous sinus, which needs to be opened for exposure of its content. Once the CN III is dissected from its dural piercing to the inferior clinoidal pillar – which can be eased with the opening of clinoidal triangle,the cavernous segment of ICA can be visualized from the posterior to the anterior bends, including the most common point of emergence of the ILT [
Optic-carotid triangle
The optic-carotid triangle is formed by the union of the lateral border of the optic nerve, medial border of the supraclinoid segment of ICA and fist segment of anterior cerebral artery (pre-communicating segment of anterior cerebral artery).[
Exploring this triangle can be useful in order to approach sella turcica and pituitary stalk. Also, in a lateral to medial view, the contralateral CN III can be exposed. In an anterior to posterior transcavernous view, the basilar apex and both superior cerebellar artery (SCA), first segment of both posterior cerebral (pre-communicating segment of posterior cerebral artery, P1), the ipsilateral posterior communicating (PComA), and thalamoperforating arteries can be explored.[
Carotid-oculomotor triangle
Lateral to the optic-carotid triangle, the carotid-oculomotor triangle is marked by the lateral border of supraclinoid segment of ICA, medial border of CN III, and first segment of middle cerebral artery (pre-bifurcation segment of middle cerebral artery). It can also be included within the clinoidal triangle.[
The loosening of third nerve and ICA by opening of this triangle, that constituted the superior wall of cavernous sinus – which can be maximized by opening of the clinoidal triangle, – allows medial mobilization of ICA and lateralization of CN III, exposing the posterior clinoid process, which can be removed especially in the context of exposing both SCA and P1 origins, the anterior portion of the second segment of posterior cerebral artery, the basilar artery, and its apex and ipsilateral PComA, and its perforating branches,[
Hakuba’s or oculomotor triangle
The oculomotor triangle was initially described by Hakuba in 1986[
Lateral wall of the cavernous sinus
The lateral wall of the cavernous sinus is divided in two layers – an internal and an external. The external layer is continuous with the dura covering the floor of middle fossa, and can be approached intradurally. On the other hand, the dissection of its inner layer, partially constituted by the cranial nerves IV and ophthalmic (V1), maxillary (V2), and mandibular (V3) divisions of trigeminal nerve (CN V), depends on the removal of its external layer, what is more easily performed extradurally, with a peeling that detaches the dura-mater from the floor of middle fossa up until the cavernous sinus, being limited by the tentorial incisura medially, marking the end of lateral wall and beginning of superior wall.
Such as in the cases of the above-mentioned triangles, since they are adjacent to one another [
Figure 4:
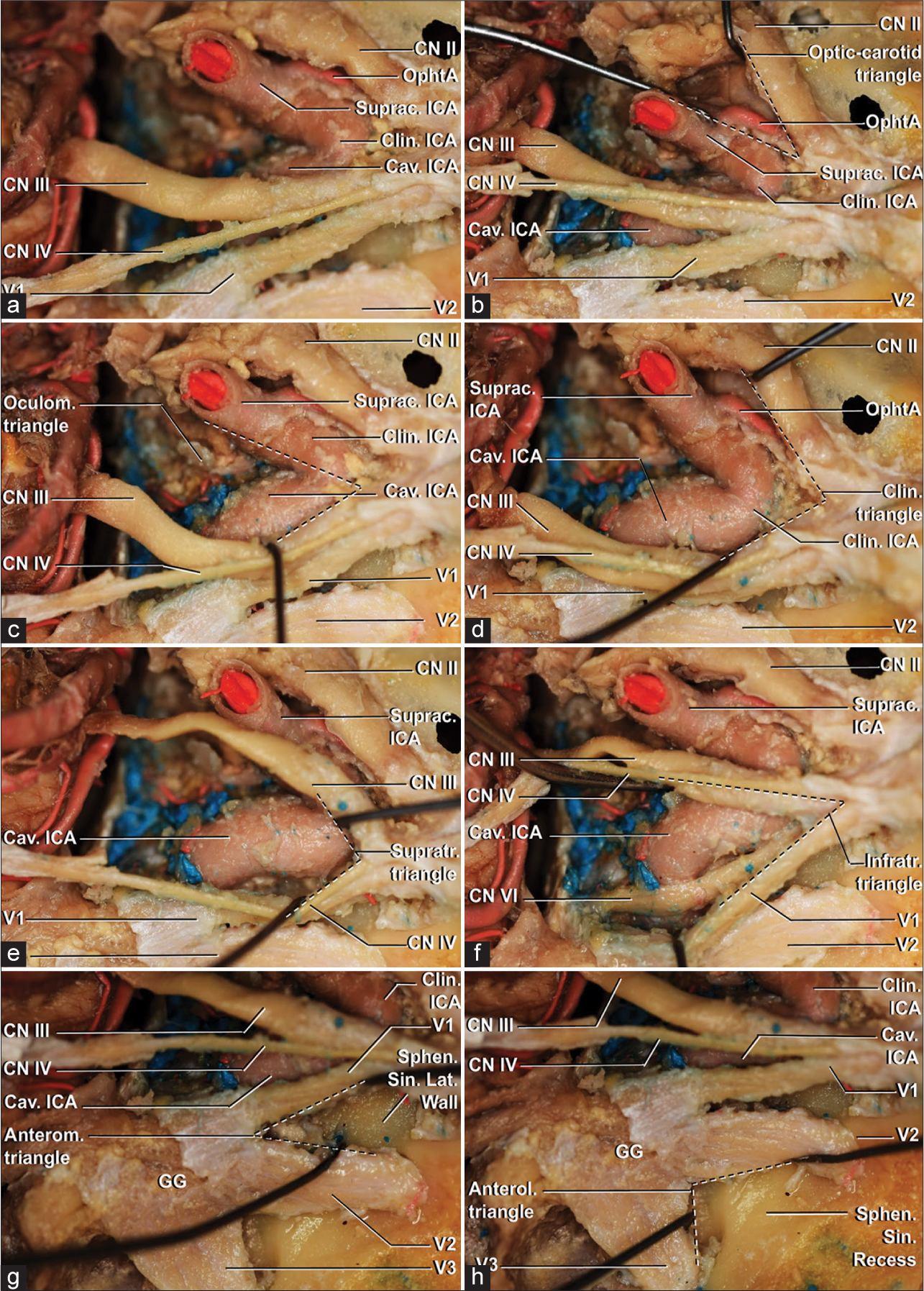
A super-lateral view of a right cavernous sinus with a 60º clockwise rotation. (a) The overview of the roof and lateral wall of the cavernous sinus. (b) Exploration of the optic-carotid triangle demonstrates the emergence of the ophthalmic artery from the supraclinoid portion of the internal carotid artery, distal from the distal dural ring. (c) Exposition of the carotid-oculomotor triangle above the roof of the cavernous sinus. D, dissection of the clinoidal triangle demonstrates the distal cavernous (horizontal and anterior loop portions), clinoidal and supraclinoid segment of the internal carotid artery. (e) The opening of the supratrochlear triangle exposes the horizontal portion of the cavernous segment. (f) Demonstration of the infratrochlear triangle exposes the posterior loop and proximal horizontal portions of cavernous ICA and the abducens nerve, lateral to the artery and medial to ophthalmic division of the trigeminal nerve. Cavernous sinus is filled with venous blood. (g) The anteromedial triangle, limited medially by the inferior margin of V1 and laterally by the superior margin of V2, allows approaching the lateral wall of the sphenoid sinus. (h) The wall to the lateral recess of the sphenoid sinus is located in the anterolateral triangle, located between the inferior border of V2 and superior border of V3. Anterol: Anterolateral, Anterom: Anteromedial, Cav: Cavernous, Clin: Clinoidal, CN II: Optic nerve, CN III: Oculomotor nerve, CN IV: Trochlear nerve, ICA: Internal carotid artery, Infratr: Infratrochlear, Lat: Lateral, OphtA: Ophthalmic artery, Sin: sinus, Sphen: sphenoid, Suprac: Supraclinoid, Supratr: Supratrochlear, V1: Ophthalmic division of trigeminal nerve, V2: Maxillary division of trigeminal nerve, V3: Mandibular division of trigeminal nerve.
Figure 5:
A lateral view of a right cavernous sinus with a 90º clockwise rotation. (a) Overview of transition between the roof and lateral wall of the cavernous sinus. (b) The carotid-oculomotor triangle can be visualized with emergence of the posterior communicating artery. (c) Oculomotor triangle, on the roof of the cavernous sinus, laterally limited by the anterior petroclinoid ligament, medially by the interclinoid ligament ang posteriorly by the posterior petroclinoid ligament. (d) The point of entrance of the trochlear nerve in the dura-mater and lateral wall of the cavernous sinus. (e) Infratrochlear triangle opening with exposure of the posterior loop and horizontal portions of cavernous internal carotid artery. Also, the emergence of the meningohypophyseal trunk is noted. ACP: Anterior clinoid process, Ant: Anterior, Anterom: Anteromedial, Cav: Cavernous, Clin: Clinoidal, CN II, optic nerve, CN III: Oculomotor nerve, CN IV: Trochlear nerve, ICA: Internal carotid artery, Infratr: Infratrochlear, Lig: Ligament, PComA, Posterior communicating artery, Petroclin: Petroclinoid, Suprac: Supraclinoid, Supratr: Supratrochlear, V1: Ophthalmic division of trigeminal nerve.
Parkinson’s or infratrochlear triangle
Described by Parkinson in 1964,[
The opening of the Parkinson triangle is used for the direct approach of the posterior bend and proximal horizontal segment of the cavernous ICA and the origin of the MHT.
Furthermore, it allows visualization of the entire intradural course of abducens nerve, which follows the carotid lateral wall, internally to the lateral wall of the cavernous sinus, which can be eased with lateral retraction of V1.[
Mullan’s or anteromedial triangle
The anteromedial triangle is located between nerves V1 and V2, from its origin in the gasserian ganglion, until the nerves follow their course, entering the superior orbital fissure and foramen rotundum, respectively. It was first described by Mullan, in 1979.[
Anterolateral or lateral triangle
This narrow triangle, described by Dolenc in 1989,[
Middle fossa floor
Glasscock’s and Kawase’s triangles [
Glasscock’s or posterolateral triangle
Glasscock’s triangle, described in 1979,[
Kawase’s or posteromedial triangle
The posteromedial triangle, first described in 1977,[
The anterior face of the triangle consists of an interface with Glasscock’s and can be approached to expose ICA similarly.
The importance of this triangle is paramount in skull base surgery, since it allows, through the anterior petrosectomy, the communication between middle and posterior fossa. By drilling the petrous apex and the triangular and quadrangular areas medial and superior to the IAC and GSPN, the clivus posterior to porus trigeminus and the inferior petrous sinus can be exposed, so as cochlea and IAC, laterally, and, in between, the dura-mater of posterior fossa. The approach then proceeds to an intradural phase, in which the superior petrosal sinus and tentorium are divided and the anterolateral portion of pons, the vertebrobasilar junction and anteroinferior cerebellar artery can be visualized.[
CONCLUSION
Although time has allowed immense evolution of the knowledge regarding the cavernous sinus, once known as a surgical “no man’s land,” it still remains an indigestible subject for a great deal of neurosurgeons, including skull base surgeons. Its intricate tridimensional anatomy, though inviting, can be seen as excessively difficult to comprehend, especially for beginners. The systematic revision of these constant anatomic references for approaching this space is, therefore, keen, for those who seek for a better understanding and management of its related pathologies and, consequently, improvement in patient care.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Dolenc VV. A combined epi-and subdural direct approach to carotid-ophthalmic artery aneurysms. J Neurosurg. 1985. 62: 667-72
2. Dolenc V, editors. Anatomy and Surgery of the Cavernous Sinus. Vienna, Austria: Springer-Verlag; 1989. p.
3. Drazin D, Wang JM, Alonso F, Patel DM, Granger A, Shoja MM. Intracranial anatomical triangles: A comprehensive illustrated review. Cureus. 2017. 9: e1741
4. Glasscock ME, Jackson GC, Dickins JR, Wiet RJ. Panel discussion: Glomus jugulare tumors of the temporal bone. The surgical management of glomus tumors. Laryngoscope. 1979. 89: 1640-54
5. Hakuba A. Surgical approaches to the cavernous sinus--repair of a C-C fistula at the C5 portion of the internal carotid artery. No Shinkei Geka. 1986. 14: 601-7
6. Hendricks BK, Benet A, Lawrence PM, Benner D, Preul MC, Lawton MT. Anatomical triangles for use in skull base surgery: A comprehensive review. World Neurosurg. 2022. 164: 79-92
7. Kanzaki J, Kawase T, Sano K, Shiobara R, Toya S. A modified extended middle cranial fossa approach for acoustic tumors. Arch Otorhinolaryngol. 1977. 217: 119-21
8. Mullan S. Treatment of carotid-cavernous fistulas by cavernous sinus occlusion. J Neurosurg. 1979. 50: 131-44
9. Parkinson D. A surgical approach to the cavernous portion of the carotid artery. Anatomical studies and case report. J Neurosurg. 1965. 23: 474-83
10. Rhoton AL. The cavernous sinus, the cavernous venous plexus, and the carotid collar. Neurosurgery. 2002. 51: 375-410
11. Rhoton AL. The supratentorial cranial space: Microsurgical anatomy and surgical approaches. Neurosurgery. 2002. 51: S1-iii