- Department of Neurosurgery, Neurozone 3D Research Center, Lima, Peru
Correspondence Address:
Cristian Eugenio SalazarCampos, Department of Neurosurgery, Neurozone 3D Research Center, Lima, Peru.
DOI:10.25259/SNI_767_2024
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Christian Alexander Yataco-Wilcas, Luis Alberto Lengua-Vega, Yosimar Salomon Coasaca-Tito, Bruno Eduardo Diaz-Llanes, Cristian Eugenio Salazar-Campos. Neurocircle microsurgery model: Description of simulation-based training and exoscope. 01-Nov-2024;15:392
How to cite this URL: Christian Alexander Yataco-Wilcas, Luis Alberto Lengua-Vega, Yosimar Salomon Coasaca-Tito, Bruno Eduardo Diaz-Llanes, Cristian Eugenio Salazar-Campos. Neurocircle microsurgery model: Description of simulation-based training and exoscope. 01-Nov-2024;15:392. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=13200
Abstract
Background: Microsurgery is crucial in neurosurgery, requiring precise skills for interventions on delicate structures. Effective training is essential for developing these skills. In Peru and Latin America, however, there is a notable shortage of specialized training centers and high costs associated with foreign simulators, hindering the development of neurosurgical skills. To address this issue, the NeuroZone3D Research Center has initiated a project to create a national dataset on Peruvian craniometry and develop locally adapted training models.
Methods: The “NeuroCircle Microsurgery Model” was created through a multi-phase process. Phase 1 involved designing a simulation platform using a 3D printer. Phase 2 focused on creating a realistic biomodel with polyurethane and advanced modeling techniques. In Phase 3, the biomodel was assembled and integrated into the platform. Phase 4 included using a wooden module, a cost-effective exoscope simulation, and surgical instruments to provide a realistic training environment.
Results: The “NeuroCircle Microsurgery Model” provided a stable and effective training environment. Feedback from training sessions with neurosurgery residents and medical students indicated significant improvements in microsurgical skills. Participants valued the model’s realism and its role in enhancing their surgical proficiency.
Conclusion: The “NeuroCircle Microsurgery Model” is a significant advancement in microsurgery training for Peru and Latin America. Its development provides a valuable, locally adaptable tool for improving surgical skills and addresses the critical training gaps in the region.
Keywords: Microsurgery, Neuroanatomy, Neurosurgery, Skills, Training
INTRODUCTION
Microsurgery plays a fundamental role in the field of neurosurgery, enabling precise and highly delicate interventions on neuroanatomical structures, including the brain, blood vessels, and cranial nerves.[
However, in the Peruvian context, there is a notable shortage of appropriate models for microsurgery training. This lack restricts the development of skills among local professionals and, in turn, may affect the quality of medical care provided in the country.
In an effort to address this issue, the NeuroZone3D Research Center is leading an initiative to create a national dataset on Peruvian craniometry.[
In this context, we introduce the first simulation model developed in Peru, the “NeuroCircle Microsurgery Model,” which stands out for its pioneering role in training neurosurgery residents and medical students. Through two successful training sessions facilitated by simulation and a cost-effective exoscope, participants significantly improved their microsurgical skills. This article outlines the development process of the model, from its conception to the formal training provided at a high-reliability simulation center, representing a significant advancement in Peruvian neurosurgery by offering solutions tailored to local needs in medical training.
MATERIALS AND METHODS
Phase 1: Platform creation
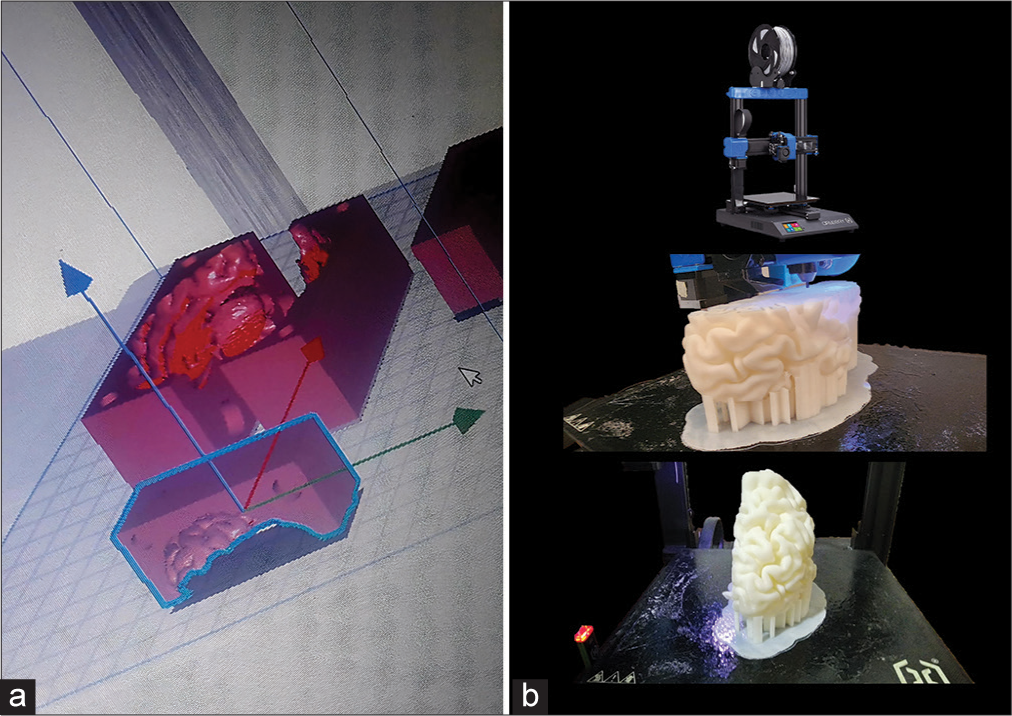
The “NeuroCircle Microsurgery Model” simulation platform was designed to provide a realistic and effective environment for microsurgery training [
Phase 2: Creation of the biomodel
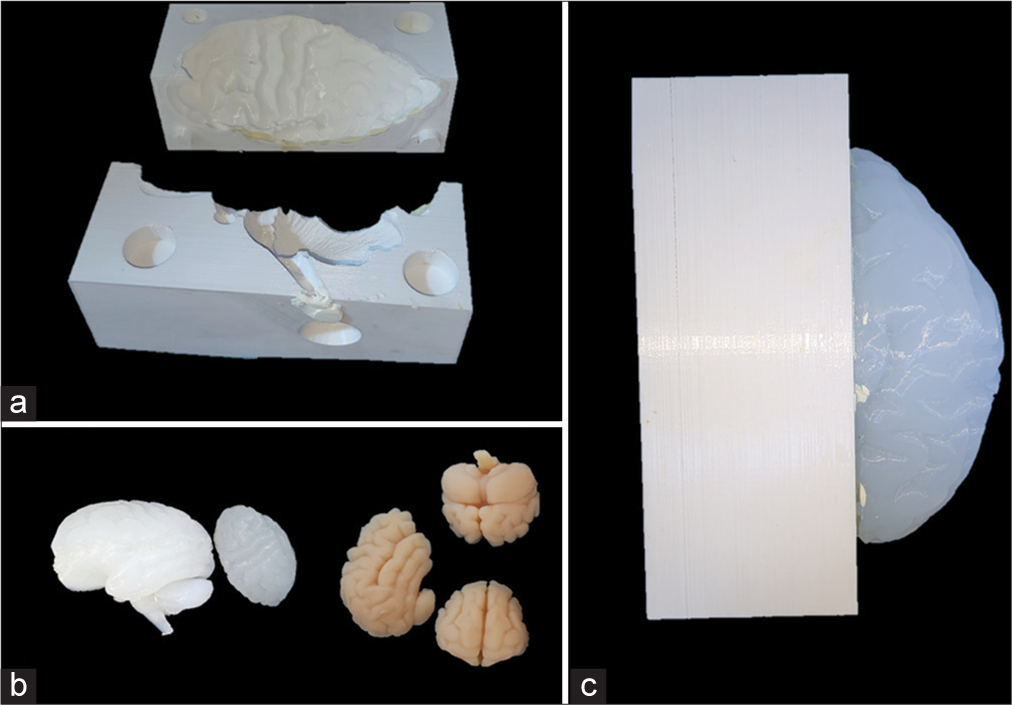
For the creation of the biomodel, advanced modeling techniques were employed using the open-source software Ultimaker Cura (Ultimaker Company, Netherlands). The process began with the design of a negative filament model to serve as a mold for the brain mass [
Once molded, the models underwent a curing process using a special mixture of solid starch and cellulose components, which removed residues from the filament-negative mold and prepared the biomodels for assembly.
Phase 3: Assembly of the biomodel
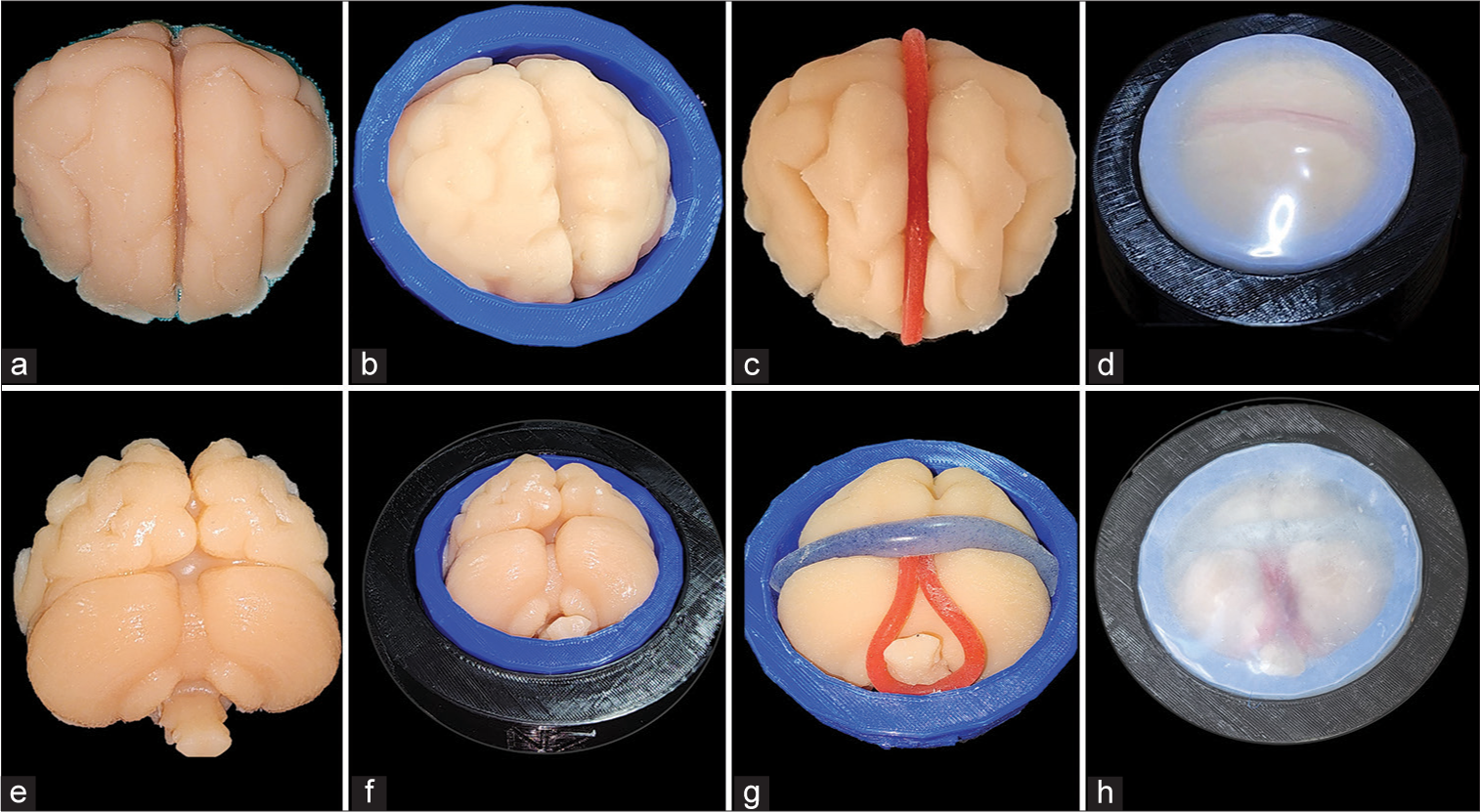
The assembly of the biomodel was carried out with precision to ensure its integrity and functionality during the training sessions in the neurosurgery laboratory. An acrylic adhesive in aerosol form (3M Company, Minnesota, USA) was applied to the polyurethane brain mass models to simulate the arachnoid layer in the biomodel. The polyurethane models were then integrated into the circular platform according to a carefully planned sequence. The cured brain mass model was placed first on the smaller diameter platform, followed by the blood vessels and the dura mater. By overlaying the larger diameter circular platform, the ideal tension of the dura mater was achieved to keep the biomodels in place and simulate real operating room conditions [
Phase 4: Use of the biomodel in the laboratory
To facilitate training in a simulated neurosurgery environment, a wooden cube module with an opening on one side was designed. The module measured 24 cm × 20 cm × 23 cm, with a thickness of 2 cm, providing a stable and adequate space for the placement of the biomodel during training sessions [
To simulate a cost-effective exoscope, a webcam (Halion, China) with an adjustable lens and image capture focus was used. In addition, a circular LED light (LightTech, China) with 18 W power and white light was implemented to ensure optimal visibility during the neurosurgery training sessions [
For microsurgery practice, various surgical instruments were employed, including microsurgical scissors, Castroviejo micro needle holders, Prolene 5–0 sutures, filament clips, and malleable retractors. Furthermore, 35 mm × 40 mm filament aneurysm clips were created to complement the available tools for microsurgical practice.
This module design, along with the careful selection of tools and equipment, provided a realistic and effective training environment where participants could acquire and enhance their microsurgical skills in a controlled and safe setting.
RESULTS
Workstation
The designed workstation effectively met the goal of providing a stable environment for microsurgery practice. The wooden training module provided a stable and accessible structure, allowing participants to perform microsurgical practices with increased comfort and confidence. The opening on one side of the module facilitated access to the biomodel, enabling effective and precise manipulation of the simulated neuroanatomical structures. This setup promoted an immersive and detailed learning experience, essential for the development of advanced microsurgical skills, and allowed each participant to take an active role in their training.
Laboratory instruments
The surgical instruments selected for training were crucial in creating a realistic and effective practice environment. The toolset included microsurgical scissors, Castroviejo micro needle holders, Prolene 5–0 sutures, filament clips, malleable retractors, and 35 mm × 40 mm aneurysm clips. These instruments were carefully chosen to replicate real surgical conditions, providing participants with the opportunity to become familiar with handling high-precision surgical tools. The availability of these instruments enabled participants to develop and refine their microsurgical skills in a controlled and supervised environment, promoting safe and effective learning.
Implementation of laboratory training
The training in the laboratory was conducted using the “NeuroCircle Microsurgery Model,” with two training sessions involving neurosurgery residents and medical students. During these sessions, participants practiced various microsurgical techniques, including vascular anastomosis, dura mater dissection, and manipulation of delicate brain structures.
Each training session began with a theoretical introduction to the techniques to be practiced, followed by practical demonstrations conducted by experienced instructors. Participants then engaged in individual practice, applying what they had learned under the supervision and guidance of the instructors.
The participants’ feedback on the training was positive. Residents and medical students appreciated the opportunity to practice in a controlled and realistic environment, which allowed them to become familiar with microsurgical techniques in a safe setting. They expressed satisfaction with the quality of the biomodel and the workstation, highlighting the effectiveness of the simulator in replicating real surgical conditions.
Future publications will present detailed results of the training, including quantitative and qualitative analyses of the effectiveness of the “NeuroCircle Microsurgery Model” in developing microsurgical skills in neurosurgery.
DISCUSSION
In Latin America, and specifically in Peru, there is a notable lack of specialized microsurgery training centers. This deficiency significantly limits continuous training opportunities for local neurosurgeons, adversely affecting the quality of neurosurgical care in the region. Unlike other parts of the world where simulation and training centers are well-established and accessible, such resources are scarce and often unavailable in our region.[
One of the main obstacles to developing microsurgical skills in our region is the high cost associated with acquiring foreign models and simulators. Although effective, these tools are financially out of reach for many institutions and health-care professionals in Latin America.[
To overcome economic barriers and improve accessibility to microsurgery training, it is imperative to develop low-cost simulation models that can be replicated locally. These models should be designed to allow personalized training, adapting to the individual progress of each neurosurgeon. The ability to manufacture these models in-house or at local research and development centers would significantly contribute to the continuous education of neurosurgeons, enhancing their skills and, consequently, the quality of medical care. In addition, these cost-effective models would facilitate the implementation of broader and more frequent training programs, allowing more neurosurgeons to access high-quality education.
Creating simulation models that closely resemble real microsurgical conditions is crucial for developing neuromuscular memory in neurosurgeons. Neuromuscular memory refers to the ability to replicate an action or procedure with greater ease and precision as it is practiced repeatedly.[
A fundamental aspect of microsurgery training is the coordination of the eye, brain, and hand (EBH). This coordination is essential for performing precise and delicate procedures within milliseconds. Circular models, such as the “NeuroCircle Microsurgery Model,” are particularly useful for developing this coordination. Practicing with these models helps neurosurgeons learn to see, process, and execute movements with precision and speed. This capability is crucial in microsurgery, where every movement must be exact and calculated to avoid damaging delicate neuroanatomical structures.[
CONCLUSION
The implementation of low-cost, high-fidelity simulation models, such as the “NeuroCircle Microsurgery Model,” represents an innovative and necessary solution for enhancing microsurgery training in Peru and Latin America. The shortage of specialized training centers and the high costs of foreign models have been significant obstacles to developing advanced neurosurgical skills in our region. However, by creating replicable and affordable models that enable personalized and continuous training, we can overcome these barriers and provide our neurosurgeons with the essential tools to develop and refine their skills.
The “NeuroCircle Microsurgery Model” not only offers a realistic environment that facilitates the development of neuromuscular memory and eye-mind-hand coordination, but it has also been positively received by participants in initial training trials. The ability to practice on a model that accurately simulates real surgical conditions is crucial for training competent neurosurgeons and building their confidence.
The local adoption and ongoing development of simulation models like the “NeuroCircle Microsurgery Model” represent a significant advancement for medical education in our country. Continued efforts and future publication of results will contribute to validating and refining these models, ensuring their effectiveness and adaptability to the specific needs of our health-care professionals.
Ethical approval
The Institutional Review Board approval is not required as it does not involve patients or confidential data.
Declaration of patient consent
Patient’s consent was not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Byvaltsev VA, Akshulakov SK, Polkin RA, Ochkal SV, Stepanov IA, Makhambetov YT. Microvascular anastomosis training in neurosurgery: A review. Minim Invasive Surg. 2018. 2018: 6130286
2. Cherian I, Burhan H, Efe IE, Jacquesson T, Maldonado IL, Uhl JF, Jorge J, Lopes DS, Campos PF, editors. Innovations in microscopic neurosurgery. Digital anatomy : Applications of virtual, mixed and augmented reality. Cham: Springer International Publishing; 2021. p. 243-56
3. CIENHUS - Centro de investigación. Available from: https://cienhus.com/en [Last accessed on: 2024 Jul 23].
4. De Luca P, Di Stadio A, Scarpa A, Ricciardiello F, Viola P, Radici M. 3-D virtual reality surgery training to improve muscle memory and surgical skills in head and neck residents/young surgeons. Eur Arch Otorhinolaryngol. 2024. 281: 2767-70
5. Efe IE, Çinkaya E, Kuhrt LD, Bruesseler MM, MührerOsmanagic A. Neurosurgical education using cadaver-free brain models and augmented reality: First experiences from a hands-on simulation course for medical students. Medicina (Kaunas). 2023. 59: 1791
6. Evgeniou E, Walker H, Gujral S. The role of simulation in microsurgical training. J Surg Educ. 2018. 75: 171-81
7. Goiri MA, de Amorim BL, Sarti TH, da Costa MD, ChaddadNeto F. Building a microneurosurgical laboratory in Latin America: Challenges and possibilities. Surg Neurol Int. 2021. 12: 573
8. Haase J, Boisen E. Neurosurgical training: More hours needed or a new learning culture?. Surg Neurol. 2009. 72: 89-95
9. Hafez A, Raj R, Lawton MT, Niemelä M. Simple training tricks for mastering and taming bypass procedures in neurosurgery. Surg Neurol Int. 2017. 8: 295
10. Petrone S, Cofano F, Nicolosi F, Spena G, Moschino M, Di Perna G. Virtual-augmented reality and life-like neurosurgical simulator for training: First evaluation of a hands-on experience for residents. Front Surg. 2022. 9: 862948
11. Pujari V. Muscle memory and the brain: How physical skills are stored and retrieved. J Adv Med Dent Scie Res. 2019. 7: 273-9
12. Spetzger U, Etingold J, Schilling A, editors. Training models for skull-base and vascular micro-neurosurgery. London: Intechopen; 2022. p.
13. Torres CS, Mora AE, Campero A, Cherian I, Sufianov A, Sanchez EF. Enhancing microsurgical skills in neurosurgery residents of low-income countries: A comprehensive guide. Surg Neurol Int. 2023. 14: 437
14. Wang T, Li H, Pu T, Yang L. Microsurgery robots: Applications, design, and development. Sensors (Basel). 2023. 23: 8503
15. Yataco-Wilcas CA, Diaz-Llanes BE, Coasaca-Tito YS, Lengua-Vega LA, Salazar-Campos CE. Craniometric analysis in modern Peruvian population. Neurosurg Cases Rev. 2023. 6: 144
16. Yataco-Wilcas CA, Diaz-Llanes BE, Coasaca-Tito YS, Lengua-Vega LA, Salazar-Campos CE. Morphometric analysis of transsphenoidal surgery in Peruvian population. Surg Neurol Int. 2024. 15: 156
17. Yataco-Wilcas CA, Salazar-Ascurra A, Diaz-Llanes BE, Coasaca-Tito YS, Lengua-Vega LA, Salazar-Campos CE. Morphometric analysis of the foramen magnum in the Peruvian population. Surg Neurol Int. 2024. 15: 9