- Department of Neurosurgery, Faculty of Medicine, Universitas Airlangga – Dr. Soetomo General Academic Hospital,
- Faculty of Veterinary Medicine, Universitas Airlangga,
- Departments of Orthopaedics and Traumatology, Faculty of Medicine, Universitas Airlangga – Dr. Soetomo General Academic Hospital, Surabaya,
- Public Health and Preventive Medicine, Faculty of Medicine, Universitas Airlangga – Dr. Soetomo General Academic Hospital, Surabaya,
- Department of Neurosurgery, Faculty of Medicine, Universitas Hasanuddin – Dr. Wahidin Sudirohusodo Central General Hospital, Makassar, South Celebes,
- Department of Anatomical Pathology, Faculty of Medicine, Universitas Airlangga – Dr. Soetomo General Academic Hospital, Surabaya, East Java, Indonesia.
Correspondence Address:
I Ketut Sudiana, Department of Anatomical Pathology, Faculty of Medicine, Universitas Airlangga – Dr. Soetomo General Academic Hospital, Surabaya, East Java, Indonesia.
DOI:10.25259/SNI_62_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Tedy Apriawan1, Widjiati Widjiati2, Dwikora Novembri Utomo3, Asra Al Fauzi1, Eko Agus Subagio1, Budi Utomo4, Andi Asadul Islam5, Abdul Hafid Bajamal1, I. Ketut Sudiana6. Periosteum-induced ossification effect in skull defect through interleukin-8 and NF-κB pathway: An experimental study with Oryctolagus cuniculus rabbits. 15-Apr-2022;13:140
How to cite this URL: Tedy Apriawan1, Widjiati Widjiati2, Dwikora Novembri Utomo3, Asra Al Fauzi1, Eko Agus Subagio1, Budi Utomo4, Andi Asadul Islam5, Abdul Hafid Bajamal1, I. Ketut Sudiana6. Periosteum-induced ossification effect in skull defect through interleukin-8 and NF-κB pathway: An experimental study with Oryctolagus cuniculus rabbits. 15-Apr-2022;13:140. Available from: https://surgicalneurologyint.com/surgicalint-articles/11543/
Abstract
Background: The purpose of this study was to analyze the response of inflammatory cytokines interleukin-8 (IL-8) and NF-κB to the closure of skull defect with periosteum as a scaffolding material in bone healing used after surgery.
Methods: Thirty Oryctolagus cuniculus rabbits underwent a craniotomy to create a 20 mm diameter round defect in the parietal bones. The parietal bones were returned to its place and stabilized by an internal plate fixation. The defects were either left empty or implanted with periosteum. At 6 weeks, the specimens were euthanized and examined.
Results: Histological examination showed a more well-developed formation of woven bone in the periosteum group. Immunohistochemical examinations showed that the use of periosteum in the closure of skull defects reduced the NF-κB and IL-8 response which affected the ossification process.
Conclusion: The experiment showed that the use of periosteum was linked with IL-8 and NF-κB downregulation toward ossification effects at any point throughout the trial. Periosteum usage might be beneficial as a scaffolding material in bone healing for autograft cranioplasty in animal model and could be applied to clinical practice.
Keywords: Cranioplasty, Interleukin-8, NF-κB, Periosteum flap
INTRODUCTION
Decompressive craniectomy is a surgical procedure that involves the removal of the skull bone to protect the patient from increasing intracranial pressure.[
Numerous cranioplasty methods have been developed to address the problems of bone resorption. Duhamel and John Hunter performed one of them utilizing the periosteum membrane, which has osteogenic characteristics. The periosteal membrane is made up of two layers: an exterior (fibrous and smooth) layer and an interior layer (cambium). The outer layer is densely packed with collagen and the reticular layer is densely packed with osteoblasts and osteoprogenitor cells, while the inner layer is densely packed with osteoblasts and osteoprogenitor cells.[
MATERIALS AND METHODS
Study design
The research protocol was authorized by the authorities of Universitas Airlangga, Department of Veterinary (Experimental Animal Studies, study number 2.KE.087.10.2020). The experimental study used 10 adults Oryctolagus cuniculus rabbits that were at least 5 months old and weighed between 4 and 5 kg. Animals were randomly divided into two groups (Group: with periosteum and without periosteum), each containing five rabbits. After 6 weeks, all animals were sacrificed and immunohistochemistry examination was performed.
Medication of animals
All operations were performed under general anesthesia, 65 mg kg–1 ketamine and 4 mg kg–1 xylazine, administered intramuscularly into the hind leg. Each animal received an intramuscular injection of 100,000 IU prophylactic benzylpenicillin. Following surgery, the animals received analgesics for 3 days, once daily, muscularly using phenylbutazone.
Craniotomy protocol
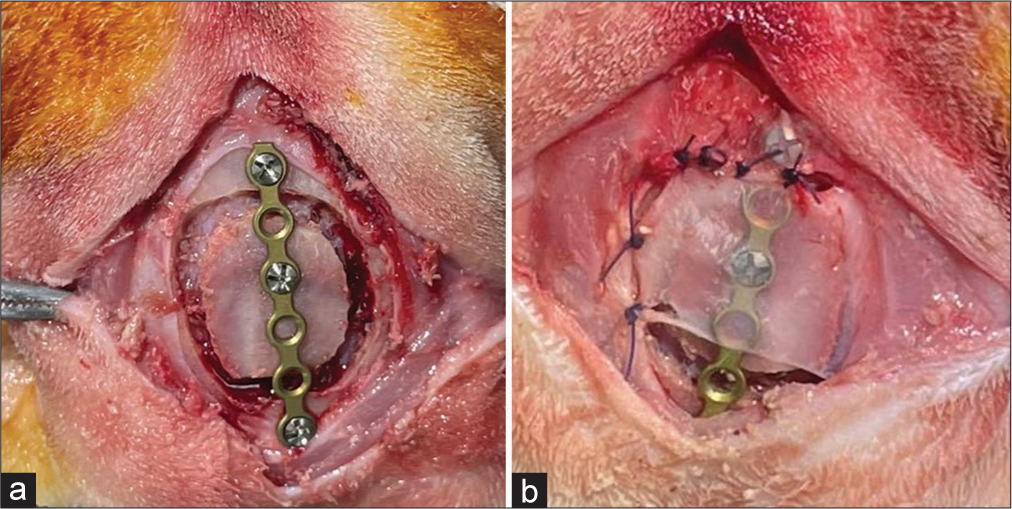
The rabbits’ head was shaved between the eyes and the ears for surgical site preparation. The skin was sterilized with a solution of 10% iodine. After administering a local anesthetic (1 mL lidocaine and epinephrine solution); subcutaneously, a midline incision was created, and the skin and periosteum were reflected to reveal the skull. Round bone defects (diameter 20 mm) were bored into the inner cortex using a bone trephine with dural preservation. Following bone plate removal, the bone is returned and fixed using a long plate [
Histopathological examination
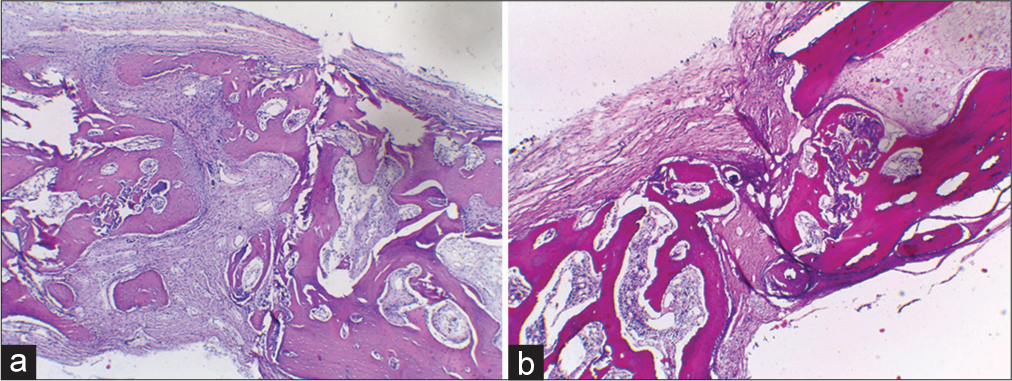
For 4 h, the specimens were preserved in cold 4% formaldehyde. The tissues were frozen in optimum cutting temperature compound (Sakura Finetek, Torrance, CA). Sections were cut to a thickness of 5 mm, attached on charged slides, and allowed to air dry for 5 min before staining. Staining with hematoxylin and eosin was carried out according to the procedure. The samples were analyzed using a light microscope (Fluorescence Microscope Olympus IX71).
Immunohistochemistry examination
Immunohistochemical responses were performed on rabbit skull tissue obtained 6 weeks after periosteum implantation. A monoclonal mouse anti-human NF-κB antibody was used to determine the expression of the NF-κB marker (ready-to-use antibody; 2J10D7; Novus Biologicals, CO, USA). Interleukin-8 expression was quantified using antibodies, anti-interleukin-8 (IL-8) antibody (monoclonal mouse, ready to use; MBS2025703; MybioSource, San Diego, USA). At room temperature, all sections were treated with the primary antibodies for 1 h. In addition, antigen determinants of all proteins were exposed by boiling sections for 15 min at 98°C in 10 mM citrate buffer (pH 6.5). Light microscope was used to examine the materials using a Fluorescence Microscope Olympus IX71 (Olympus, Tokyo, Japan).
RESULTS
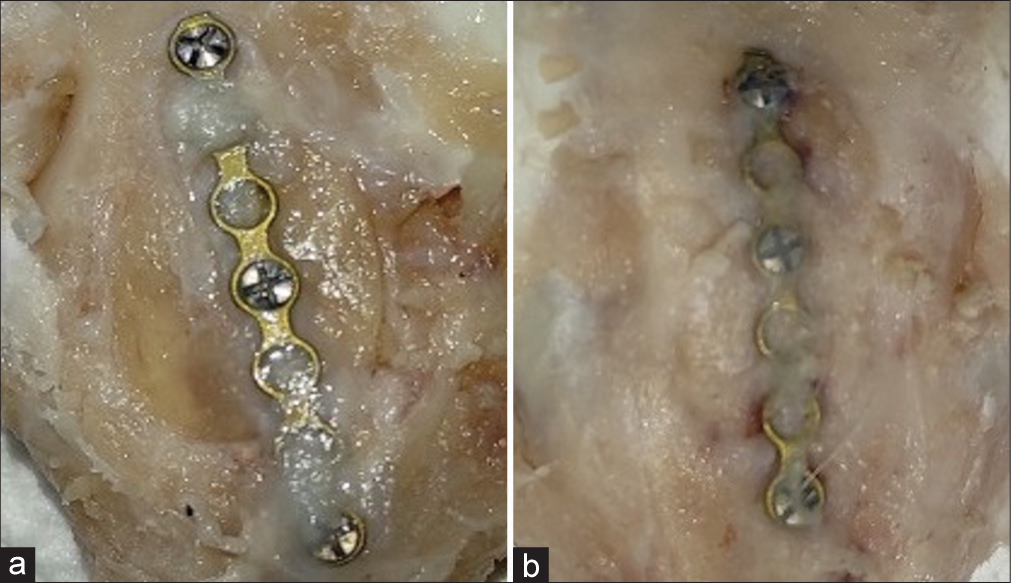
Thirty animals were utilized in the research. All animals recovered normally and were terminated as scheduled. The two groups, rabbit skull following placement of periosteum and without periosteum, were compared from all skull fragments. The final study comprised 30 animals, and [
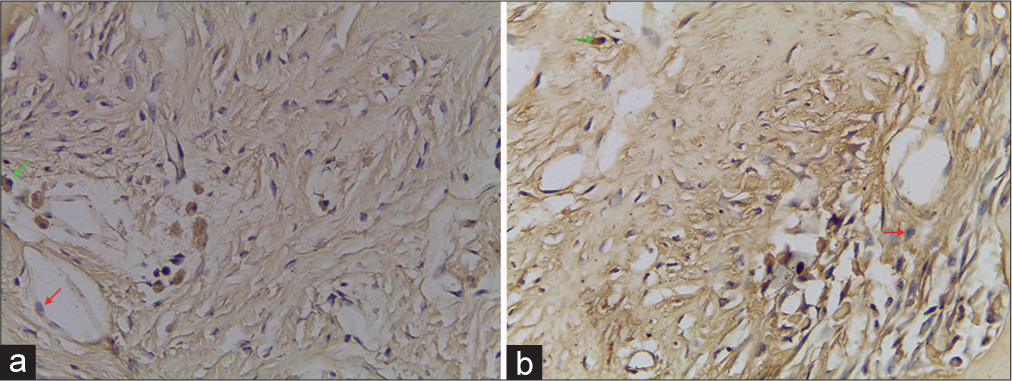
In those with periosteum, the woven bone formation could be observed on the defect walls without bridging the defects. The defect with the periosteum showed woven bone formation to the level of the original cortex [
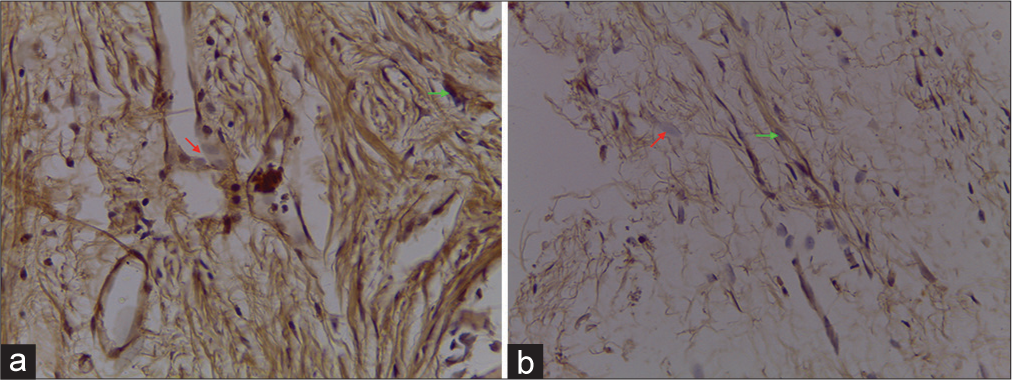
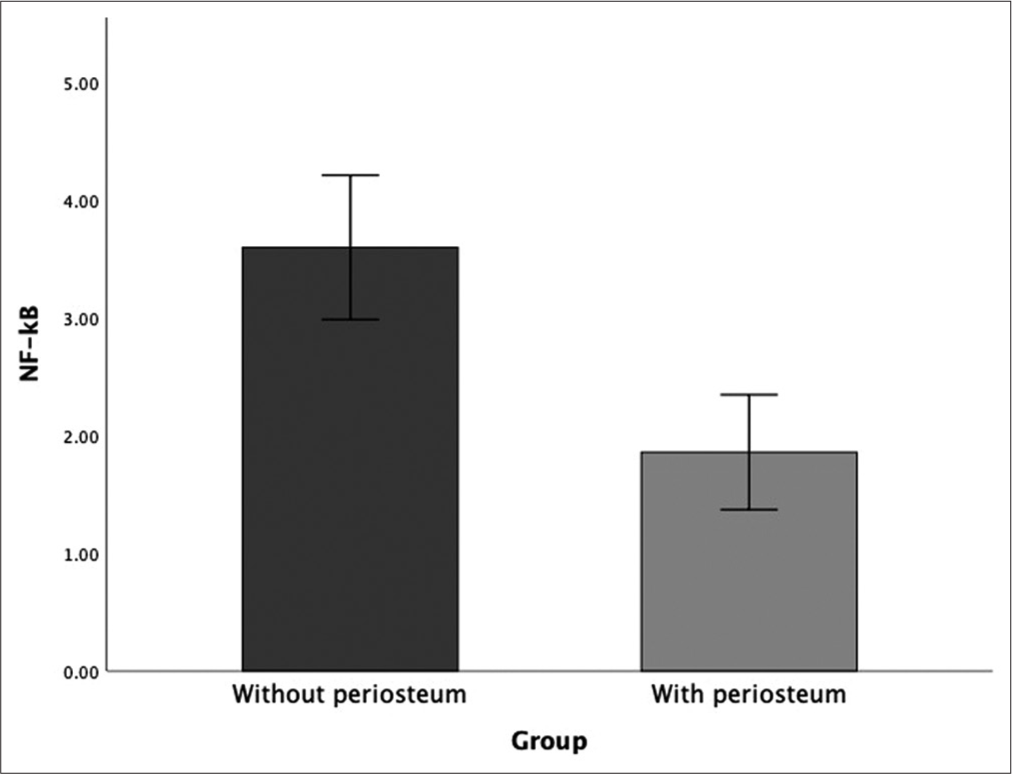
NF-κB expression/6 weeks
Statistical analysis showed a significant difference between each group at week 6 posttreatment (P < 0.05). Bone defects without periosteum showed the highest number of NF-B compared to cranioplasty with pericranium grafts [
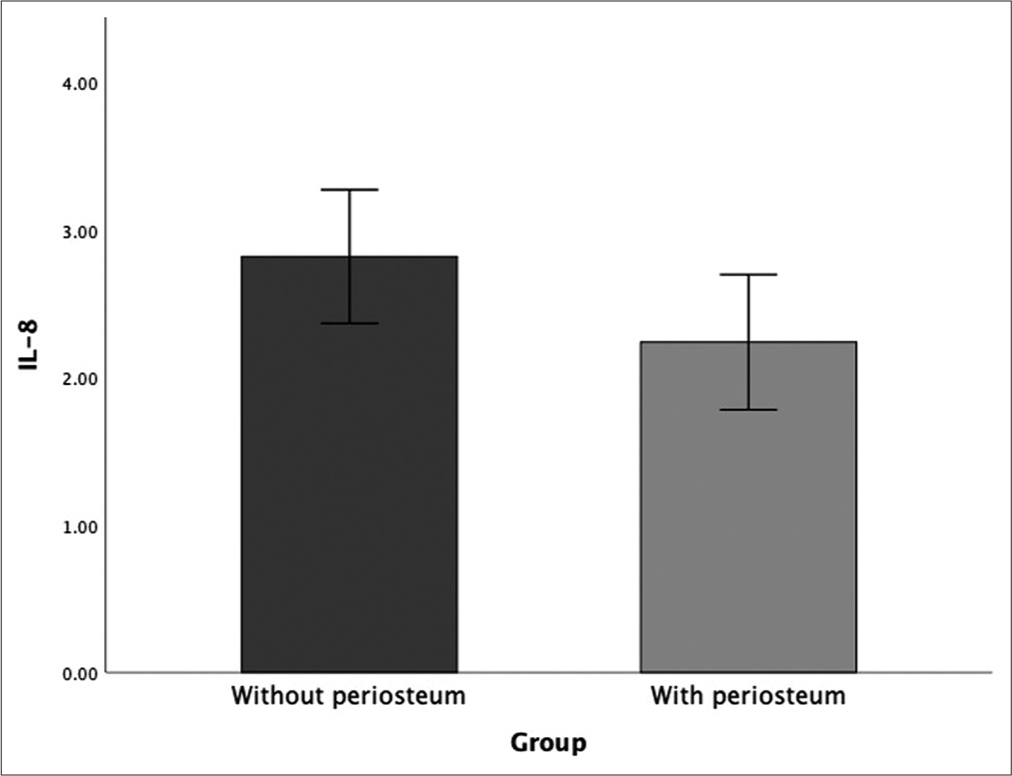
IL-8 expression/6 weeks
Statistical analysis showed no significant difference (P > 0.05) between the use of periosteum and without periosteum at week 6 posttreatment. Bone defects without periosteum showed the highest IL-8 expression compared to the use of periosteum [
DISCUSSION
Bone regeneration is a multistep process that begins with the mitotic expansion of progenitor cells at the sites of bone formation and continues with differentiation of these progenitor cells into functional osteoblasts. Our research sought to investigate if periosteum might be utilized as a scaffold for bone grafts, assisting in the multistep process of bone regeneration. In addition, we evaluated the osteogenic capacity of periosteum through the use of different osteoblastic phenotypic markers. Any bone graft material or scaffolding must possess the characteristics required to promote the growth of bone-forming cells in an optimum microenvironment. It was shown in the histologic examination that a more well-formed woven bone formation was observed in the periosteum group, indicating a faster bone healing compared to no periosteum flap.
The osteogenic capabilities of the osteoblasts were investigated for 6 weeks in our study. For assessing the osteogenic potential, Declercq, 2004, study and Aronow, 1990, study utilized time points ranging from 2 to 6 weeks. Investigators chose these different time points based on the component of osteoblasts they were studying. The 6-week endpoint appeared acceptable because we were evaluating the total response of the osteoblasts from all sources. Periosteum itself does not provide structural support, but it is ideal for covering faults and voids in the bone. It could be employed as a stand-alone substance to boost the osteogenic process. Periosteum can reduce the amount of artificial compound required to fill a defect, resulting in smaller donor sites and lower morbidity. The periosteum produced the least amount of IL-8 and NF-κB in our investigation. As a result of this discovery, we can deduce that the healing process will accelerate if bone defects are covered with periosteum. Our research demonstrated that when the graft is exposed to a sufficient supply of cytokines, nutrients, and hormones, osteoprogenitor cells become activated and stain positive for osteogenic potential indicators.
NF-κB (activated B-cell core factor) is a part of the transcription factor, which was first identified as a regulator of B-cell differentiation by its ability to bind to the B site of the kappa light chain gene in B cells. They were later shown to be involved in innate and adaptive immunity in response to pathogens and autoimmune stimuli and many aspects of normal cellular function.[
NF-κB itself is one of the cytokines that have been known to play an essential role in the tissue healing process.[
IL-8 has been reported by a previous study by Edderkaoui[
This research has several limitations, two of which are the considerably low number of samples and short period of follow-up for the new bone formation. This experiment also does not evaluate the critical bone defect size, which may play an important role in the practice of autograft osteoplasty procedure.
CONCLUSION
This study demonstrates that the periosteum has the elements required for osteogenesis. Periosteum also exhibits lower IL-8 production and NF-κB laying down properties, according to the study. Because of its porous nature and the fact that it is autogenous, it causes no immunologic reaction, making it easy to assimilate into the defect. The periosteum is ideal for covering bone defects because it is readily available and has a layer-like consistency. Periosteum proved to be effective treatment evaluated in terms of promoting ossification and inhibiting bone resorption inside bony defects produced in a rabbit calvarium model.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
The authors would like to thank all who have contributed to the process and completion of this report, including the teaching staffs and fellow residents of the Department of Neurosurgery of Faculty of Medicine, Universitas Airlangga, Dr. Soetomo General Academic Hospital, Surabaya, Indonesia. We also want to thank the Faculty of Veterinary Medicine, the Department of Orthopaedics and Traumatology and Department of Public Health and Preventive Medicine, and the Department of Anatomical Pathology Faculty of Medicine Universitas Airlangga – Dr. Soetomo General Academic Hospital. We would also like to extend our gratitude toward the Department of Neurosurgery, Faculty of Medicine Universitas Hasanuddin – Dr. Wahidin Sudirohusodo Central General Hospital.
References
1. Abu-Amer Y. NF-κB signaling and bone resorption. Osteoporos Int. 2013. 24: 2377-86
2. Augustin G, Antabak A, Davila S. The periosteum. Part 1: Anatomy, histology and molecular biology. Injury. 2007. 38: 1115-30
3. Boyce BF. Advances in osteoclast biology reveal potential new drug targets and new roles for osteoclasts. J Bone Miner Res. 2013. 28: 711-22
4. Edderkaoui B. Potential role of chemokines in fracture repair. Front Endocrinol (Lausanne). 2017. 8: 1-8
5. Heng MC. Wound healing in adult skin: Aiming for perfect regeneration. Int J Dermatol. 2011. 50: 1058-66
6. Hoff P, Gaber T, Strehl C, Schmidt-Bleek K, Lang A, Huscher D. Immunological characterization of the early human fracture hematoma. Immunol Res. 2016. 64: 1195-206
7. Jiang WG, Sanders AJ, Ruge F, Harding KG. Influence of interleukin-8 (IL-8) and IL-8 receptors on the migration of human keratinocytes, the role of PLC-γ and potential clinical implications. Exp Ther Med. 2012. 3: 231-6
8. Joaquim AF, Mattos JP, Neto FC, Lopes A, de Oliveira E. Bone flap management in neurosurgery. Rev Neurociencias. 2009. 17: 133-7
9. Zanaty M, Chalouhi N, Starke RM, Clark SW, Bovenzi CD, Saigh M. Complications following cranioplasty: Incidence and predictors in 348 cases. J Neurosurg. 2015. 123: 182-8