- Department of Neurosurgery, Novosibirsk State Medical University, Novosibirsk, Russian Federation,
- Department of Neurosurgery, Peoples’ Friendship University of Russia, Moscow, Russian Federation,
- Department of Neurosurgery, European Medical Center, Moscow, Russian Federation,
- Department of Neurosurgery, Klinik Hirslanden, Zürich, Switzerland,
- Department of Neurosurgery, Meshalkin National Medical Research Center, Novosibirsk, Russian Federation.
Correspondence Address:
Aleksey Gaytan, Department of Neurosurgery, Peoples’ Friendship University of Russia, Moscow, Russian Federation.
DOI:10.25259/SNI_494_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Aleksey Krivoshapkin1, Aleksey Gaytan2, Orkhan Abdullaev3, Nidal Salim3, Gleb Sergeev3, Ilya Marmazeev3, Evaldas Cesnulis4, Tim Killeen4, Vladimir Tyuryn1, Roman Kiselev5, Pavel Syomin5, Aldo Spallone3. Prospective comparative study of intraoperative balloon electronic brachytherapy versus resection with multidisciplinary adjuvant therapy for recurrent glioblastoma. 11-Oct-2021;12:517
How to cite this URL: Aleksey Krivoshapkin1, Aleksey Gaytan2, Orkhan Abdullaev3, Nidal Salim3, Gleb Sergeev3, Ilya Marmazeev3, Evaldas Cesnulis4, Tim Killeen4, Vladimir Tyuryn1, Roman Kiselev5, Pavel Syomin5, Aldo Spallone3. Prospective comparative study of intraoperative balloon electronic brachytherapy versus resection with multidisciplinary adjuvant therapy for recurrent glioblastoma. 11-Oct-2021;12:517. Available from: https://surgicalneurologyint.com/surgicalint-articles/11166/
Abstract
Background: Intraoperative balloon electronic brachytherapy (IBEB) may provide potential benefit for local control of recurrent cerebral glioblastomas (GBMs).
Methods: This is a preliminary report of an open-label, prospective, comparative cohort study conducted in two neurosurgical centers with ongoing follow-up. At recurrence, patients at one center (n = 15) underwent reresection with IBEB while, at the second center (n = 15), control subjects underwent re-resection with various accepted second-line adjuvant chemoradiotherapy options. A comparative analysis of overall survival (OS) and local progression-free survival (LPFS) following re-resection was performed. Exploratory subgroup analysis based on postoperative residual contrast-enhanced volume status was also done.
Results: In the IBEB group, median LPFS after re-resection was significantly longer than in the control group (8.0 vs. 6.0 months; log rank χ2 = 4.93, P = 0.026, P 2 = 4.23, P = 0.04, P
Conclusion: These hypothesis-generating results from a small cohort of subjects suggest putative clinical benefit in OS and LPFS associated with maximal safe re-resection of recurrent GBM with IBEB versus re-resection and standard adjuvant therapy, a hypothesis that deserves further testing in an appropriately powered clinical trial.
Keywords: Brachytherapy, Glioblastoma, Glioma resection, Intraoperative radiation therapy, Radiation therapy
INTRODUCTION
Glioblastoma (GBM) is the most frequently occurring malignant intracerebral neoplasm in adults. It is characterized by significant infiltrative growth and an aggressive clinical course.[
Because of the obvious difficulty in conducting a randomized trial of gross total versus lesser resection in GBM, level 1 data supporting the value of gross total tumor resection for prolonging OS are rather spare,[
Level 1 data strongly support the role of radiation therapy in the management of newly diagnosed malignant gliomas; however, the use of EBRT in recurrent malignant glioma is often limited by the relatively high risk of radiation-related complications. The value of re-irradiation on OS, progression-free survival, and quality of life is still not well documented and needs to be investigated in future prospective trials.[
Attempts to use various intraoperative radiation therapy (IORT) techniques in patients with malignant gliomas have been undertaken for several decades. Several studies have shown improved survival rates in subjects treated with IORT compared with retrospective controls; however, these studies were performed in small groups of subjects, and exhibits significant limitations.[
Intraoperative balloon electronic brachytherapy (IBEB) is a new method that has been successfully applied in oncological practice over the past few years to treat breast cancer as well as other neoplasms.[
MATERIALS AND METHODS
Study subjects
This prospective cohort study was performed at two tertiary referral neurosurgical centers in the Russian Federation. The study enrolled in total 30 ≥18 years old patients with recurrent GBM (imaging-defined, using brain magnetic resonance imaging [MRI] with contrast enhancement and MR-perfusion) with Karnofsky Performance Status (KPS) ≥60%. Ineligibility criteria included contraindications to general anesthesia, decompensated chronic illness, acute infectious and non-infectious inflammatory processes, inability to undergo MRI with contrast enhancement and/or positron emission tomography-computed tomography (PETCT) with amino acid tracers, pregnancy, or breastfeeding. The final decision on subject inclusion was made by a multidisciplinary team with the mandatory participation of a neurosurgeon, a radiologist, a radiation oncologist, and a medical oncologist.
IBEB device
The electronic brachytherapy device used in this study is a miniaturized X-ray source that applies electronic brachytherapy (Xoft® Axxent® Electronic Brachytherapy (eBx®) System; Xoft®, a subsidiary of iCAD, Inc., San Jose, CA USA). The device enables highly focused therapeutic radiation to the target tissue with very rapid dose fall-off, which spares surrounding tissue. The X-ray source is complemented by a range of balloon applicators to be filled with varying volumes of saline to optimally fit the contour of the surgical cavity. This provides a well-defined geometry for the miniaturized X-ray source and allows the delivery of a more conformal radiation dose.
The Xoft System [
Ethics
This study was approved by the local ethics committees and was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its amendments.[
Study procedures
At Moscow, 15 consecutive subjects with recurrent GBM underwent maximum safe microsurgical resection of their recurrent tumor in combination with IBEB (IBEB Group). These subjects received no further adjuvant treatment. In the same period, Novosibirsk also recruited 15 consecutive subjects with recurrent GBM. These subjects underwent the same maximally safe resection, followed by routine postoperative adjuvant chemotherapy with or without radiotherapy, based on investigator preference (Control Group).
GBM recurrences were confirmed by contrast-enhanced MRI perfusion and additional PET-CT with amino acid tracers in selected cases. In case of questionable results, intraoperative pathomorphological examination (frozen section analysis) was performed. Ultimately, the presence of tumor recurrence was confirmed by pathological examination after surgery.
Preoperatively, the radiation oncologist, medical physicist, and neurosurgeon developed in all cases the IBEB plan (BrachyVision™; Varian Medical Systems, Inc., Palo Alto, CA). To provide this preoperative dosimetric estimate, reliable preoperative neuroimaging techniques were utilized to estimate the volume of the predicted post-operative surgical cavity and the appropriate balloon size to fit it. This allowed also the estimation of the IBEB X-ray source dose delivery to be applied [
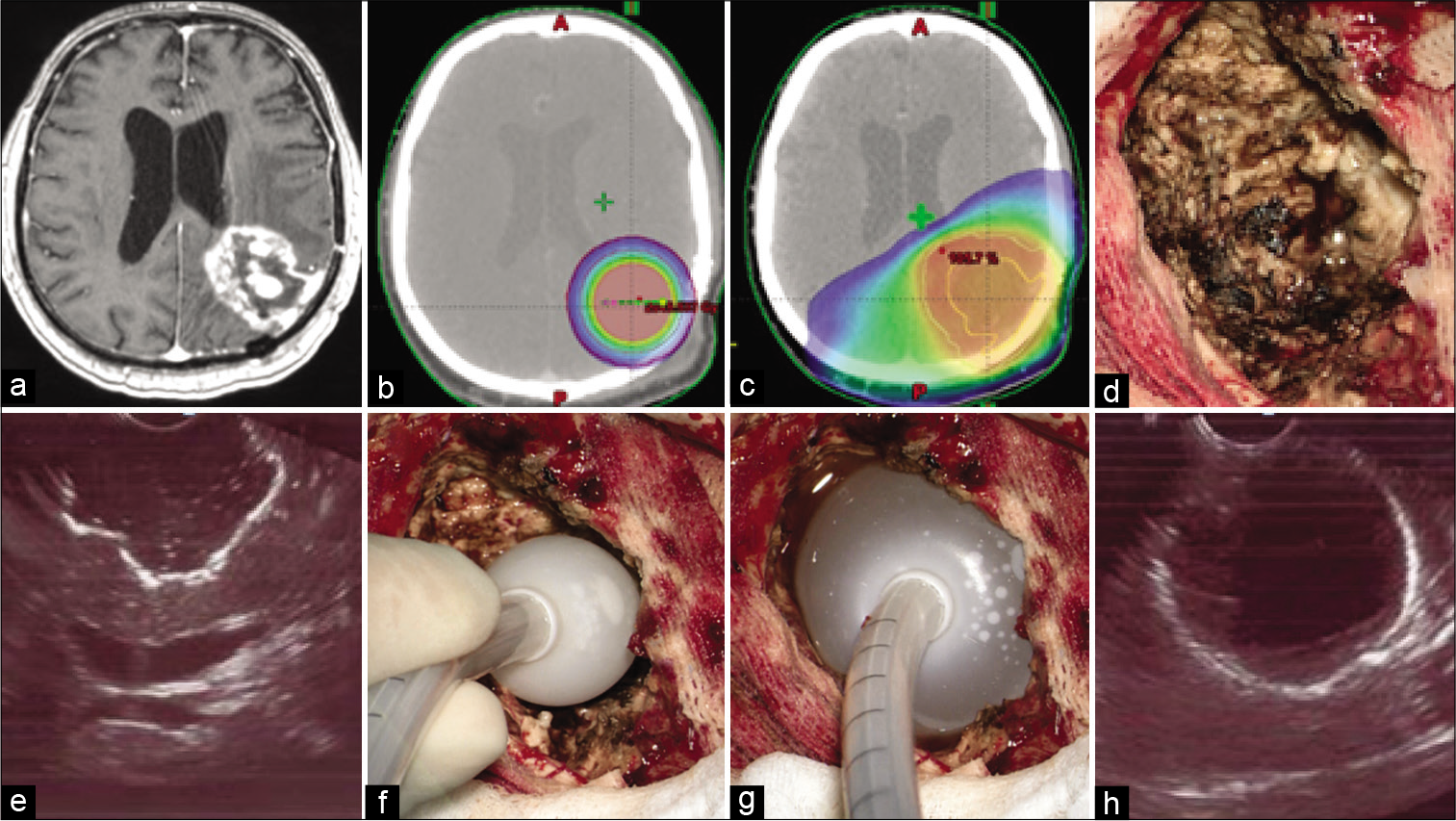
Figure 2:
(a) T1-weighted contrast-enhanced axial magnetic resonance imaging image demonstrates local glioblastoma recurrence prior to resection followed by intraoperative balloon electronic brachytherapy (IBEB). (b) IBEB isodose distribution based on computed tomography (CT) scans covers tumor bed sparing surrounding brain tissue. (c) External beam radiotherapy plan in comparison to IBEB. Isodose distribution based on the same CT scans affects extensively surrounding brain tissue which was irradiated after first surgery. (d) An overview of the post-resection cavity. (e) Intraoperative ultrasound demonstrates configuration of the postresection cavity which is free of macroscopic disease. (f) Deflated applicator balloon being introduced into the post-resection cavity. (g) Final position of the inflated applicator balloon with its dense adherence to the walls of the post-resection cavity. (h) Proper position of the balloon confirmed by intraoperative ultrasound.
Resection in both groups of subjects was conducted under general anesthesia, targeting maximal and safe resection under computed frameless MRI with contrast enhancement guidance, metabolic navigation with 5-aminolevulinic acid (5-ALA), and neurophysiological monitoring of evoked motor potentials. No “awake surgery” resection was conducted in the present patients. In the cases of multifocal tumor, only the largest neoplastic focus was resected, since it usually was responsible for the greatest mass effect. As described above IBEB was performed using the electronic brachytherapy device. After microsurgical removal of the neoplasm, the resection cavity was measured by filling it with isotonic sodium chloride solution. An empty IBEB applicator balloon was then introduced into this cavity and filled with the known volume of sodium chloride solution to give it a spherical shape, tightly adhering to the walls of the tumor bed. This was confirmed ultrasonographically [
A baseline postoperative MRI scan with and without contrast was obtained within 24 h. Following IBEB, subjects did not receive any other adjuvant treatment except for those subjects with multifocal tumors, where additional stereotactic conformal fractionated irradiation was performed to treat the remaining lesions. Two patients in the IBEB group received additional surgery and IBEB treatment to deal with multifocal tumor growth. One subject with multifocal lesions (Nr. 10) did not receive any additional radiation therapy.
In the control group, 13 subjects received adjuvant therapy by preference of the treating neurosurgeon. The systemic treatment regimens included temozolomide, bevacizumab, lomustine, and mustophoran. Eight subjects received additional EBRT with doses between 36 and 60 Gy and one subject underwent another surgery.
Follow-up and analysis
All subjects were followed monthly according to the protocol. This included clinical visits and evaluation for performance status, toxicities, and clinical evidence of progression, with imaging controls every 3 months. This included MR imaging with and without contrast, perfusion-weighted MR to assess cerebral blood volume, and PET-CT with 18-FDOPA.
Contrast-enhanced tumor volume before and after resection was evaluated with automated volumetric analysis based on DICOM MR images (NeuroSegment Software; Novosibirsk, Russia).[
Study endpoints
This is a hypothesis-generating small pilot study with two contemporaneous cohorts enrolled simultaneously at two centers, without randomization. The endpoints included OS, local progression-free survival (LPFS) and the impact of the residual postsurgical tumor volume on OS and LPFF. OS was defined as the interval from the day of the recurrent GBM surgery to death for any reason or to the last documented follow-up, whichever occurred first; LPFS was defined as the time between surgery to any Local Progression/Tumor reoccurrence within 20 mm of the cavity margin or to the last documented follow-up, whichever occurred first. Based on the suggestion of the literature regarding the impact of the extent of residual tumor volume determined by MRI within 24 h after surgery on OS,[
Statistical analysis
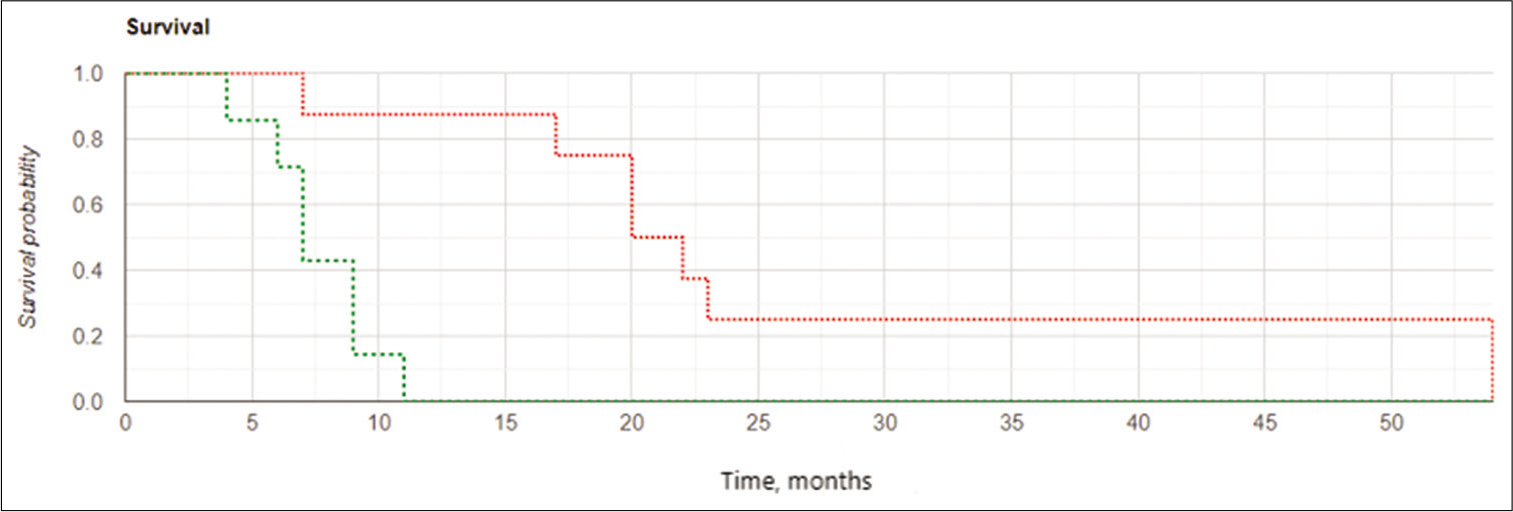
Statistical analyses of the treatment groups, including appropriate measures of central tendency and distribution, were performed using commercial software (IBM SPSS Statistics®; Armonk, NY and XLSTAT; Addinsoft, New York, NY). The effect of IBEB and the extent of tumor resection on OS and LPFS were assessed using Kaplan–Meier curves and the Log-rank test. A multivariate analysis (MANOVA) was carried out for both treatment groups. Independent variables evaluated for their impact on the efficacy endpoints included volume of residual disease, adjuvant therapy, gender, KPS, and IDH1/2 status. A univariate Cox proportional hazards model was used to calculate the hazard ratio (HR) for radical resection and IBEB therapy and a MANOVA was performed with corresponding adjustments for variables with potential confounding effects.
RESULTS
Baseline demographics and clinical characteristics
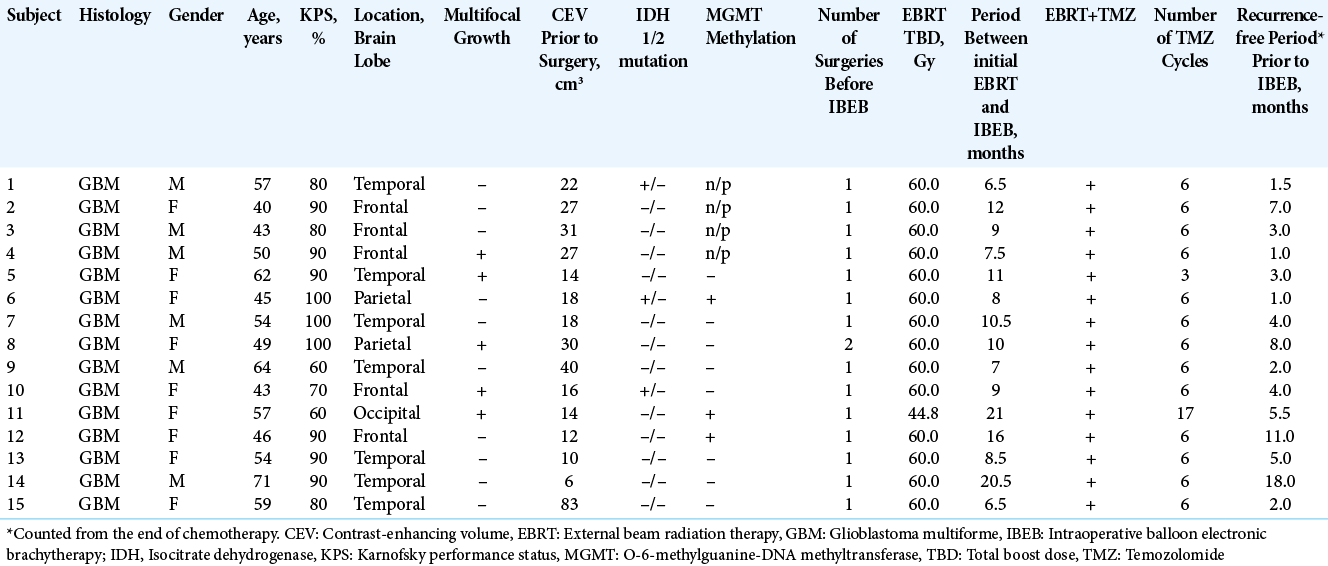
Thirty subjects were treated in the IBEB Group (Subjects 1-15) and Control Group (Subjects 16-30). Subjects in the IBEB Group had recurrent GBM (Grade IV, WHO 2016) with a mean age of 52.9 years (range, 40.0–71.0 years). Six (40%) were male. Median KPS was 90% (range, 60–100%). Other subject and tumor characteristics are summarized in [
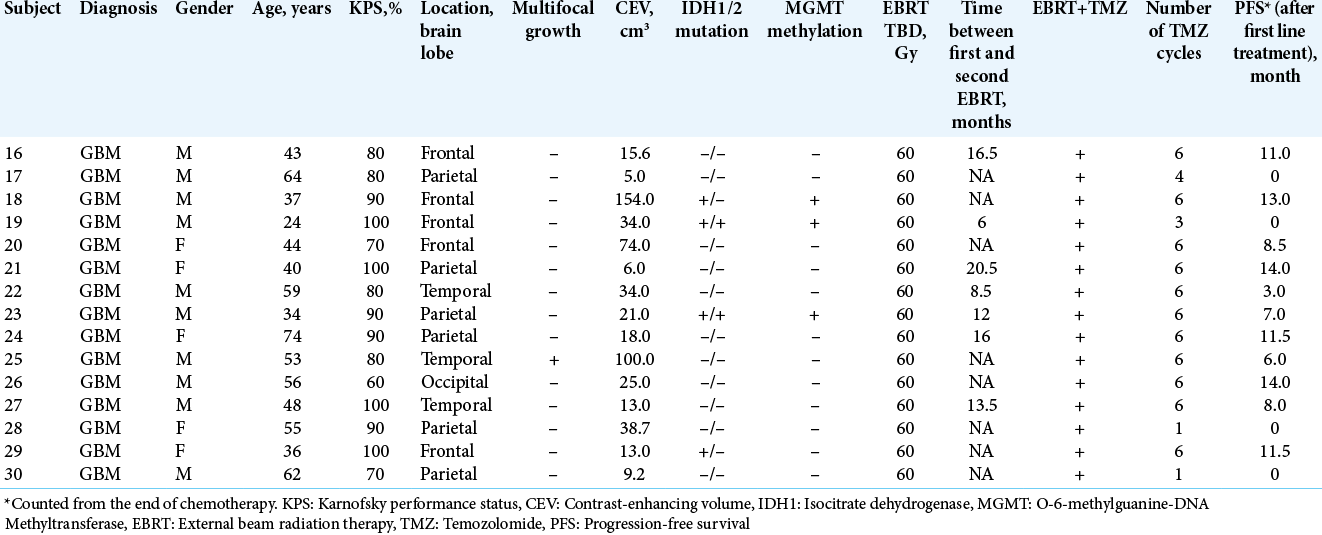
The Control Group included 15 subjects with recurrent GBM (Grade IV, WHO 2016). Two subjects did not receive any further adjuvant therapy due to postoperative complications, but they were included in the intent-to-treat analysis. The mean age of the control group subjects was slightly younger at 48.6 years (range 24.0–74.0 years). The majority, (67.7%) were male unlike what observed in the IBEB Group. Subjects in the control group had a median KPS of 85.3% (range, 60–100%). Other subject and tumor characteristics are summarized in [
Subjects in both treatment groups had a ≥6-month period between the last day of the initial radiation and any subsequent radiotherapy for the recurrent tumor [
The location of the tumor resections is presented in [
Ten IBEB Group subjects had local tumor recurrence in the immediate vicinity of the resection cavity without signs of multifocal growth. In the remaining five (Patients 4, 5, 8, 10, and 11) cases, distant tumor growth was observed in addition to the main GBM focus. Mutation in the 132nd codon of the IDH1 was detected in three subjects. No subject presented with any mutation of the IDH2 gene. Analysis of the MGMT gene promoter methylation was performed in 11 subjects. The promotor was methylated in only three cases, one of which was in combination with the mutation of the IDH1 gene.
In 14 control group subjects, local tumor recurrence occurred in the immediate vicinity of the resection cavity without signs of multifocal growth. In one case with multifocal GBM (Subject 25), there was relapse at and distant to the site of operation. The median duration of the disease-free period after the initial surgery was 8.0 months (range 0–14 months). The localization of tumor foci, subjected to resection, is presented in [
IBEB and control arm results
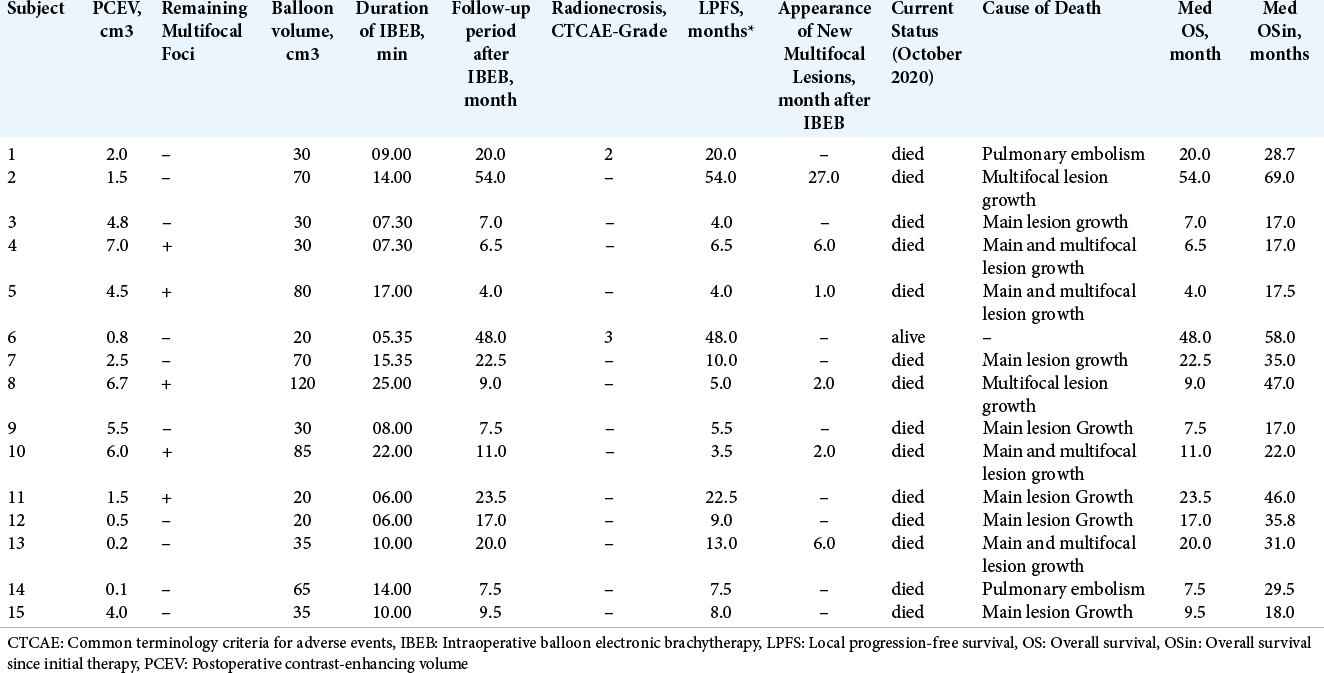
The postoperative residual contrast-enhanced volume (PCEV) values of the main lesion for each subject in the IBEB group are shown in [
In a subgroup of subjects of the IBEB group (n = 7) with PCEV >2.5 cm3, the median LPFS was 5.0 months (range 3.5– 8.0 months). It should also be noted that in this subgroup, four subjects showed multifocal growth of their GBM. In the subgroup of subjects with PCEV ≤2.5 cm3 (n = 8), the median LPFS was 16.5 months (range 7.5–54.0 months). This subgroup included one subject with multifocal GBM. Whereas five (4, 5, 8, 10, and 11) patients experienced multifocal tumor growth before IBEB treatment, two patients (2 and 13) presented multifocal lesions after IBEB, outside the radiation treatment field. Patients with multifocal lesions in the IBEB group were treated after IBEB with EBRT, or surgery followed by a second course of IBEB. Therapeutic approaches used to control the multifocal tumors in the IBEB group are given in [
The PCEV values of all subjects of the control group are shown in [
The median LPFS for the entire Control Group (n = 15) was 6.0 months (range, 2.0–10.0 months) and 33% (n = 5) of these subjects had a LPFS >6 months. In a subgroup of subjects (n = 8) with PCEV >2.5 cm3, the median LPFS was 2.0 months (range, 2.0–6.0 months). In the subgroup of subjects with PCEV ≤2.5 cm3 (n = 7), the median LPFS was 8.0 months (range, 6.0–10.0 months).
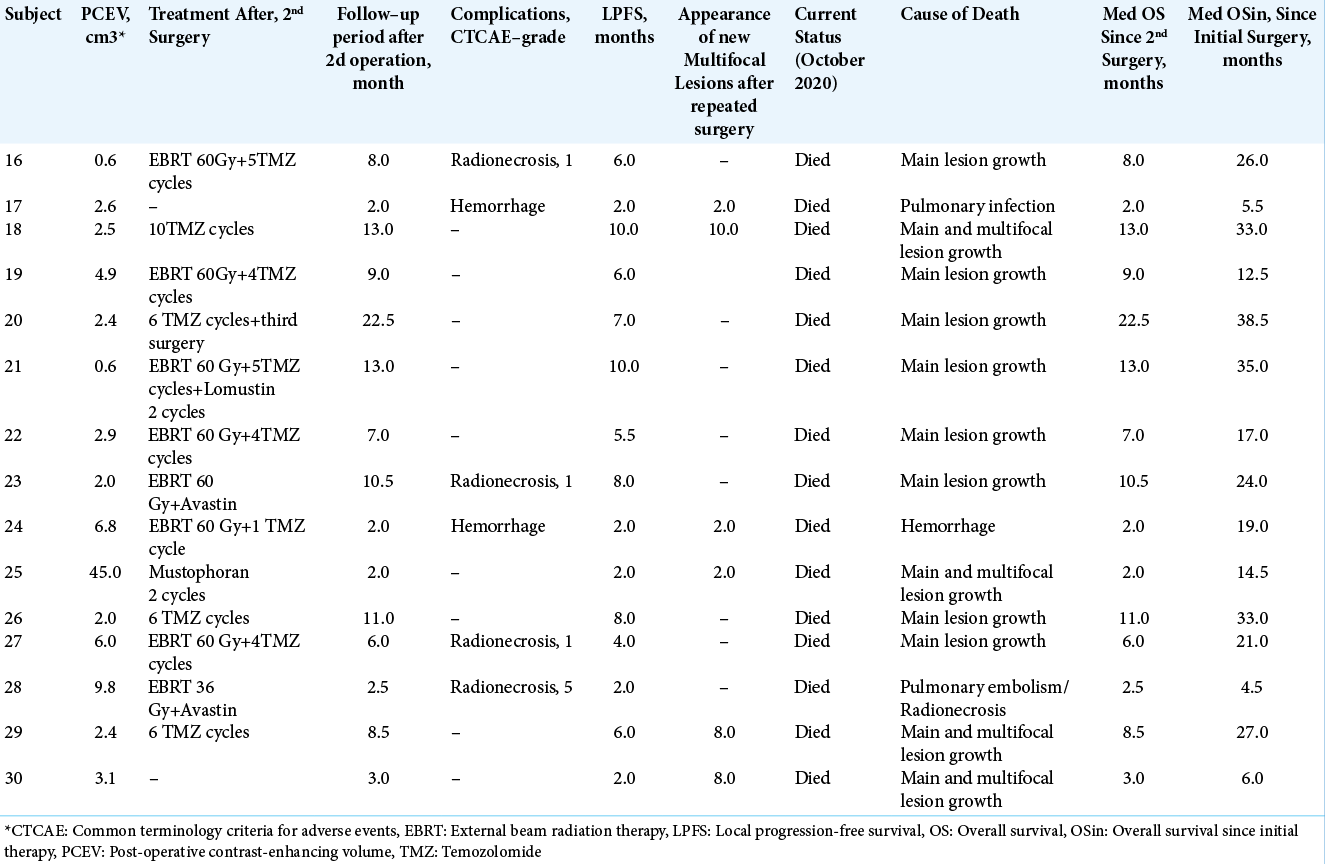
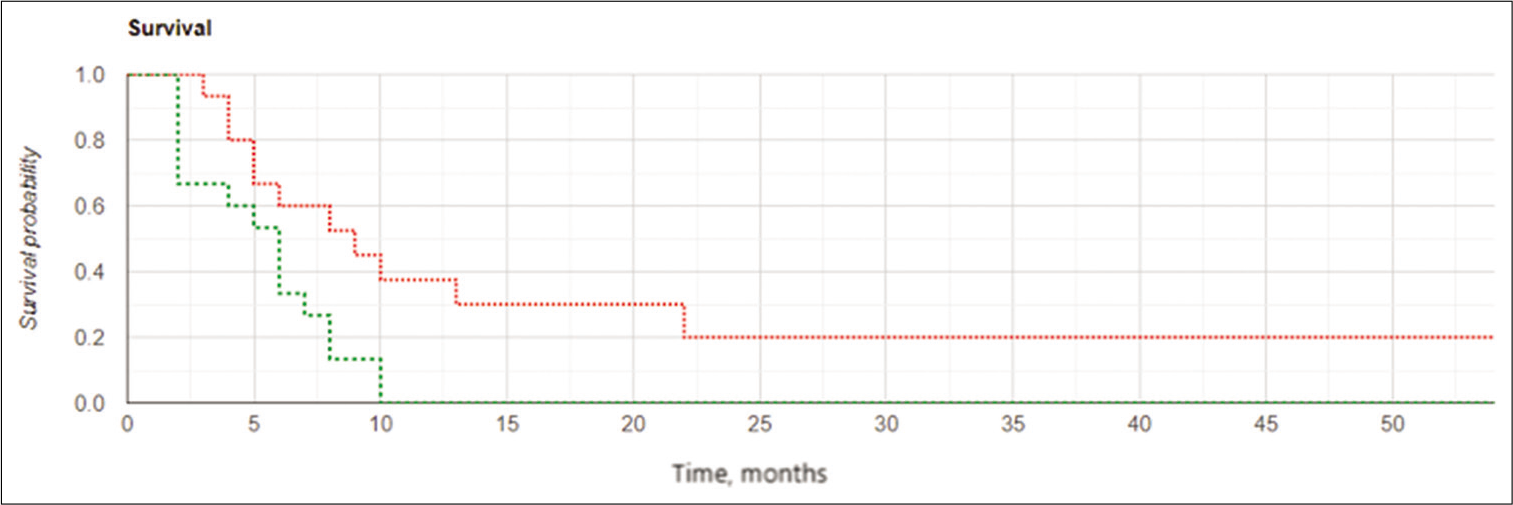
Median OS for the IBEB group was 11.0 months (range, 4.0–54.0 months). In the subgroup of subjects with PCEV >2.5 cm3 (n = 7), the median OS was 7.5 months (range, 4.0–11.0 months). In the subgroup of subjects with PCEV ≤2.5 cm3 (n = 8), median OS was 21.2 months (range, 7.5– 54.0 months). Relatively rapid progression was noted in all subjects with PCEV >2.5 cm3, irrespective of multifocal tumor growth. The Kaplan–Meier analysis of OS after reresection of recurrent GBM in combination with IBEB is shown in [
Figure 3:
Kaplan-Meier curves for overall survival in the intraoperative balloon electronic brachytherapy group stratified according to postoperative residual contrast-enhanced volume (PCEV): the subgroup of subjects with PCEV >2.5 cm3 marked in red, the subgroup of subjects with PCEV ≤2.5 cm3 marked in green; log rank χ2 = 8.03, P = 0.005, P < 0.05.
Median OS for the entire control group of subjects (n = 15) was 8.0 months (range, 2.0–22.5 months). In the subgroup of subjects with PCEV >2.5 cm3, median OS was 2.8 months (range, 2.0–9.0 months). In the subgroup of subjects with PCEV ≤2.5 cm3, median OS was 11.0 months (range, 8.0– 22.5 months). The median OSin after initial surgery for the control group was 21.0 months (range, 4.5–38.5 months) [
Treatment group comparisons
Data analysis of the CEV and PCEV, KPS, MGMT in these groups showed a normal distribution of values and the equality of variances (Livin criterion for dispersions equality, P > 0.05). Analysis of variance testing did not reveal statistically significant differences between IBEB and control groups (Pillai multivariate trace = 0.153, P = 0.390).
The results of a univariate Cox proportional hazards model to calculate the HR for PCEV and IBEB therapy are listed in [
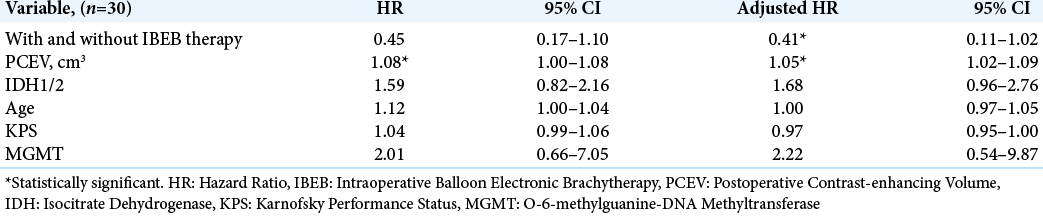
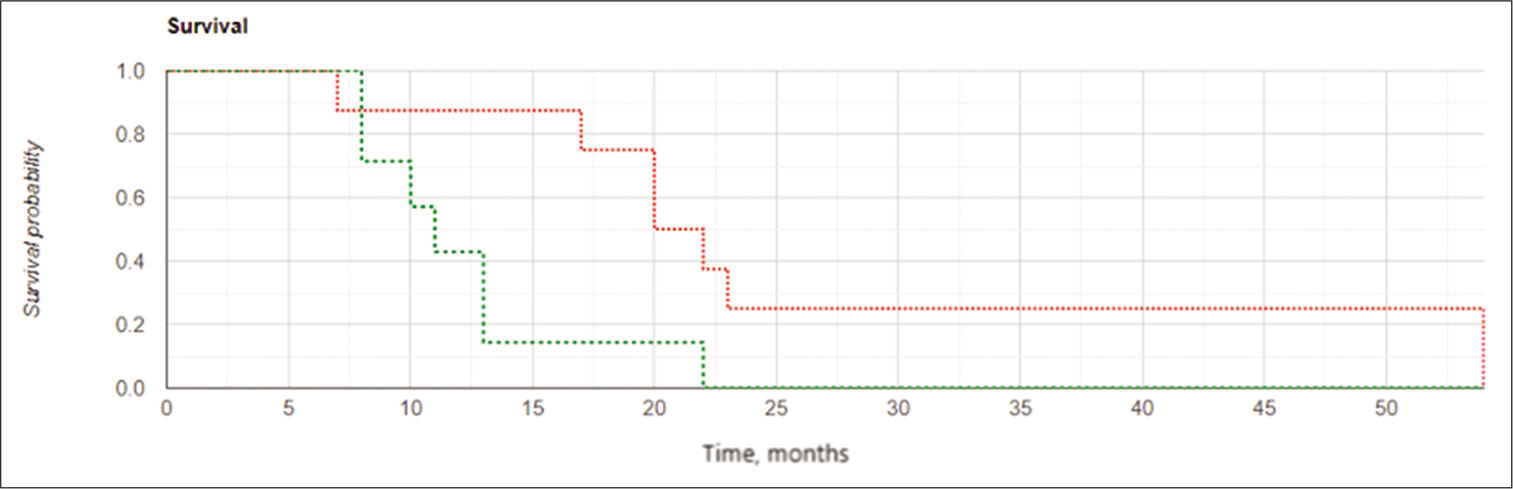
Among subjects in the IBEB, both the median LPFS and the median OS were significantly higher than those in control group (8.0 vs. 6.0 months and 11.0 vs. 8.0 months, respectively). The Kaplan–Meier curve confirmed statistically significant increased OS and LFPS for the IBEB group of subjects compared to the control group (OS: log rank χ2 = 4.23, P = 0.04, P < 0.05; LPFS: log rank χ2 = 4.93, P = 0.026, P < 0.05) [
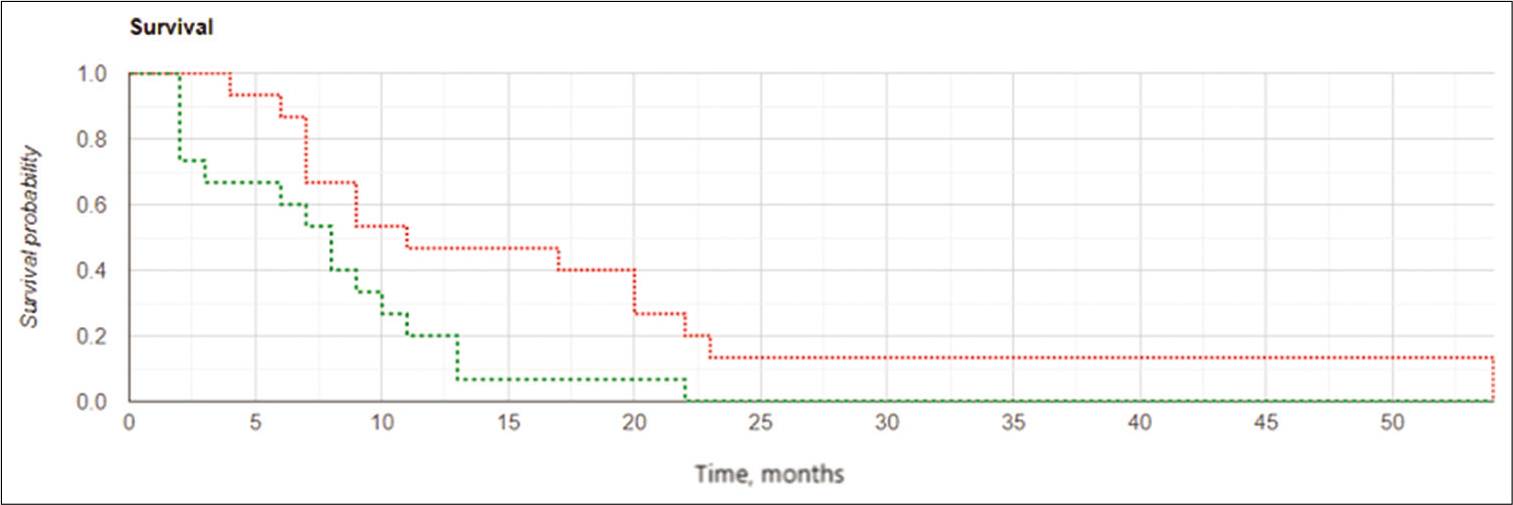
The results of the study confirmed the important role of the extent of tumor resection in cases of GBM recurrence. Kaplan– Meier curves [
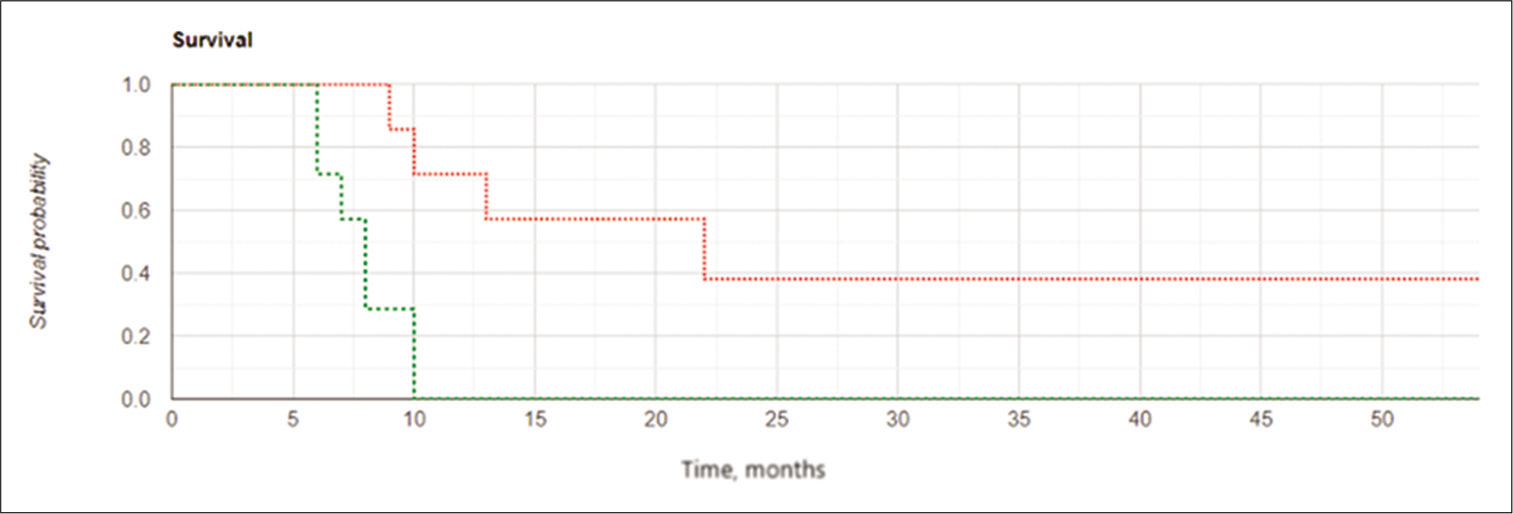
Figure 7:
Kaplan-Meier curves for local progression-free survival in the subgroups of subjects with postoperative residual contrast-enhanced volume ≤2.5 cm3: the intraoperative balloon electronic brachytherapy group marked in green, the control group marked in red; Log Rank χ2 = 7.24, P = 0.007, P < 0.05.
DISCUSSION
The treatment of recurrent GBM is still matter of debate. The role of re-resection has become widely accepted in the last decade provided some clinical parameters would seem to justify this, including age, relatively good Karnofsky status and no invasion of functionally relevant areas.[
However, others factors such as MGMT methylation, IDH 1/2 mutation, and comorbidities are known to play a role in the outcome of GBM patients. This means that evaluation of newly proposed therapeutic protocols requires unavailable extreme caution, and preliminary though encouraging results, if convincing, should be considered only a suggestive base for properly designed future studies.
In our study, we proposed for the first time the use of IBEB technique in GBM patients. This technique was recently introduced and has become a widely accepted form of local radiation therapy for malignant tumors of several organs which seems to offer the advantage of increasing the radiation dosage delivered to the malignancy while sparing almost completely the surrounding healthy tissue. We designed an open-label, not randomized study because the IBEB technology was available only in one center. For the recruitment of the control group of patients, another independent center was chosen to eliminate bias. However, the two institutions share several members of the medical staff and have strictly similar treatment protocols for GBM patients. In particular, criteria for reoperation were absolutely the same, and postoperative management in no-IBEB group of patients followed the most updated therapeutic recommendations.
The present results indicate improved, LPES in the IBEB treated group which reached statistical significance (8.0 months vs. 6.0 months) which appeared also more evident in the cases in which near-total removal had been achieved (16.5 months vs. 8.0 months). Consequently also OS was significantly longer in the IBEB group.
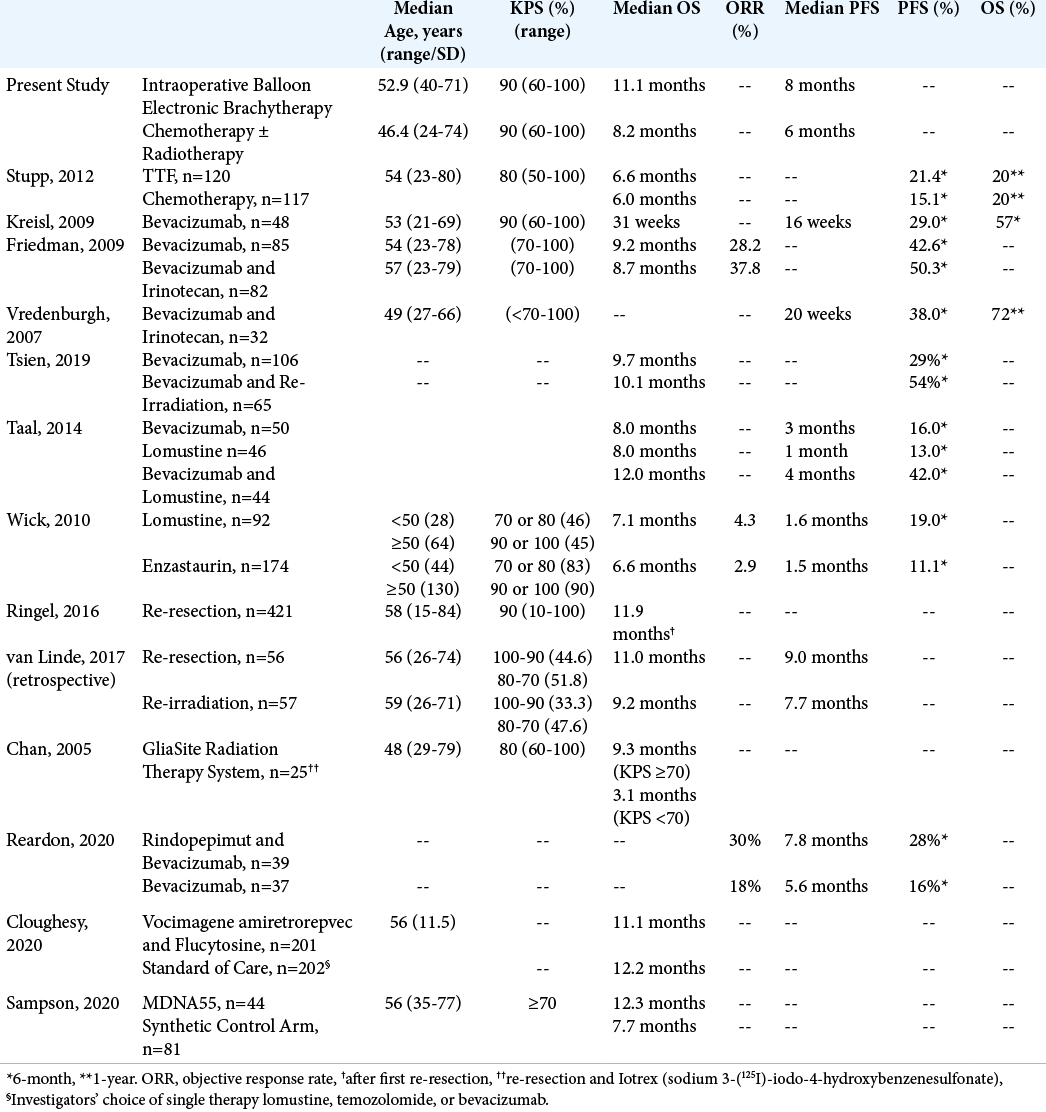
We collected from the relevant literature previously published outcomes data for the treatment of recurrent GBMs. These are summarized in [
Our study reports two cases of radionecrosis in the IBEB Group with only one CTCAE Grade 3 toxicity. No other grade ≥3 adverse events occurred in the IBEB group. This is in the same range as RTOG 1205, which reported 5% grade ≥3 events.[
In our patients the IBEB treatment took approximately 30 min, with a duration of <20 min in the vast majority of the patients, thus IBEB treatment did not significantly increase the total surgery time [
The effect of any adjuvant therapy decreases with the amount of residual tumor. GBM is an aggressively growing tumor, with the potential to increase substantially in volume within a short period.[
As mentioned previously, metabolic guidance using 5-ALA was used intraoperatively. Protoporphyrin IX (PpIX, a 5-ALA metabolite) selectively accumulates in cancer cells and was characterized as a radio-responsive compound. As in vitro studies and in vivo studies in small animals have shown, PpIX produces reactive oxygen species upon X-ray irradiation, which induces DNA double-strand breaks resulting in cell cycle arrest.[
We did not operate any of the present patients on awake surgery, a treatment protocol of which we have extensive experience particularly in recurrent GBM. Awake surgery would give immediate, functional control to the operating surgeon, and might encourage more aggressive, thought safe, surgical conduct. However, highly developed technology such as sophisticate neuromonitoring and metabolic navigation in anesthetized patients can offer equivalent safety standards to the operating surgeon who is so encouraged to perform maximally aggressive though safe resection.
Study limitations include an open-label study design, inability to control for all the factors contributing to outcome, inclusion of subjects with multifocal disease, sample size, and limited follow-up period. In addition, patients were not allocated randomly to either treatment regiments as they were treated in two different Institutions. However, we believe that the present results are appealing and should encourage further investigations by properly designed clinical trials in order determine if the innovative IBEB protocol which we used in this study might be helpful to other subjects with similar characteristics.
CONCLUSION
The results of this prospective, two-center, and comparative cohort pilot study suggest that a significant improvement in median LPFS and OS may occur in subjects undergoing IBEB following repeated resection of recurrent GBM in comparison with a control group who received standard adjuvant chemo-radiotherapy following re-resection. IBEB was associated with manageable toxicity. Subjects with a PCEV <2.5 cm3 and free of concurrent multifocal disease showed particular benefit from IBEB. This first comparative study, while limited by small sample size and its open-label nature, provides hypothesis-generating data that may warrant further investigation on the potential use of IBEB in malignant gliomas and its risk/benefit/ratio.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgment
The authors acknowledge the editorial assistance of Dr. Carl S. Hornfeldt, Apothekon, Inc., during the preparation of this manuscript.
References
1. Adkison JB, Tomé W, Seo S, Richards GM, Robins HI, Rassmussen K. Reirradiation of large-volume recurrent glioma with pulsed reduced-dose-rate radiotherapy. Int J Radiat Oncol Biol Phys. 2011. 79: 835-41
2. Barney C, Shukla G, Bhamidipati D, Palmer JD. Re-irradiation for recurrent glioblastoma multiforme. Chin Clin Oncol. 2017. 6: 36
3. Cabrera AR, Cuneo KC, Desjardins A, Sampson JH, McSherry F, Herndon JE. Concurrent stereotactic radiosurgery and bevacizumab in recurrent malignant gliomas: A prospective trial. Int J Radiat Oncol Biol Phys. 2013. 86: 873-9
4. Chan TA, Weingart JD, Parisi M, Hughes MA, Olivi A, Borzillary S. Treatment of recurrent glioblastoma multiforme with GliaSite brachytherapy. Int J Radiat Oncol Biol Phys. 2005. 62: 1133-9
5. Clarke J, Neil E, Terziev R, Gutin P, Barani I, Kaley T. Multicenter, phase 1, dose escalation study of hypofractionated stereotactic radiation therapy with bevacizumab for recurrent glioblastoma and anaplastic astrocytoma. Int J Radiat Oncol Biol Phys. 2017. 99: 797-804
6. Cloughesy TF, Petrecca K, Walbert T, Butowski N, Salacz M, Perry J. Effect of vocimagene amiretrorepvec in combination with flucytosine vs standard of care on survival following tumor resection in patients with recurrent high-grade glioma: A randomized clinical trial. JAMA Oncol. 2020. 12: e203161
7. D’Amico RS, Englander ZK, Canoll P, Bruce JN. Extent of resection in glioma-a review of the cutting edge. World Neurosurg. 2017. 103: 538-49
8. Eljamel S. 5-ALA fluorescence image guided resection of glioblastoma multiforme: A meta-analysis of the literature. Int J Mol Sci. 2015. 16: 10443-56
9. Epstein MS, Silverstein MJ, Lin K, Kim B, de Leon C, Khan S. Acute and chronic complications in patients with ductal carcinoma in situ treated with intraoperative radiation therapy. Breast J. 2016. 22: 630-6
10. Fogh SE, Andrews DW, Glass J, Curran W, Glass C, Champ C. Hypofractionated stereotactic radiation therapy: An effective therapy for recurrent high-grade gliomas. J Clin Oncol. 2010. 28: 3048-53
11. Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009. 27: 4733-40
12. Gabayan AJ, Green SB, Sanan A, Jenrette J, Schultz C, Papagikos M. GliaSite brachytherapy for treatment of recurrent malignant gliomas: A retrospective multi-institutional analysis. Neurosurgery. 2006. 58: 701-9
13. Gallia GL, Brem S, Brem H. Local treatment of malignant brain tumors using implantable chemotherapeutic polymers. J Natl Compr Canc Netw. 2005. 3: 721-8
14. Goyal U, Pan J, Cui H, Stea B. Does ultrasound measurement improve the accuracy of electronic brachytherapy in the treatment of superficial non-melanomatous skin cancer?. J Contemp Brachytherapy. 2017. 9: 14-9
15. Grabowski MM, Recinos PF, Nowacki AS, Schroeder JL, Angelov L, Barnett GH. Residual tumor volume versus extent of resection: Predictors of survival after surgery for glioblastoma. J Neurosurg. 2014. 121: 1115-23
16. Gutin PH, Iwamoto FM, Beal K, Mohile NA, Karimi S, Hou BL. Safety and efficacy of bevacizumab with hypofractionated stereotactic irradiation for recurrent malignant gliomas. Int J Radiat Oncol Biol Phys. 2009. 75: 156-63
17. Hervey-Jumper SL, Berger MS. Maximizing safe resection of low-and high-grade glioma. J Neurooncol. 2016. 130: 269-82
18. Kazmi F, Soon YY, Leong YH, Koh WY, Vellayappan B. Reirradiation for recurrent glioblastoma (GBM): A systematic review and meta-analysis. J Neurooncol. 2019. 142: 79-90
19. Kim ES, Choi YE, Hwang SJ, Han YH, Park MJ, Bae IH. IL-4, a direct target of miR-340/429, is involved in radiation-induced aggressive tumor behavior in human carcinoma cells. Oncotarget. 2016. 7: 86836-56
20. Kreisl TN, Kim L, Moore K, Duic P, Royce C, Stroud I. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol. 2009. 27: 740-5
21. Krivoshapkin A, Gaytan A, Salim N, Abdullaev O, Sergeev G, Marmazeev I. Repeat resection and intraoperative radiotherapy for malignant gliomas of the brain: A history and review of current techniques. World Neurosurg. 2019. 132: 356-62
22. Krivoshapkin AL, Sergeev GS, Gaytan AS, Kalneus LE, Kurbatov VP, Abdullaev OA. Automated volumetric analysis of postoperative magnetic resonance imaging predicts survival in patients with glioblastoma. World Neurosurg. 2019. 126: e1510-7
23. Krivoshapkin AL, Sergeev GS, Gaytan AS, Kurbatov VP, Kiselev RS, Kalneus LE. Impact of pre-and postoperative tumor volumetric data on survival of patients with glioblastomas. Bull J Neurol Psychiatr Neurosurg. 2016. 6: 59-67
24. Krivosheya D, Prabhu SS, Weinberg JS, Sawaya R. Technical principles in glioma surgery and preoperative considerations. J Neurooncol. 2016. 130: 243-52
25. Li YM, Suki D, Hess K, Sawaya R. The influence of maximum safe resection of glioblastoma on survival in 1229 patients: Can we do better than gross-total resection?. J Neurosurg. 2016. 124: 977-88
26. Lloyd SA, Rahn DA, Hoisak JD, Dragojević I. Evaluation of effective treatment depth in skin cancer treatments with electronic brachytherapy. Brachytherapy. 2018. 17: 990-4
27. Louis DN, Perry A, Reifenberger G, Deimling A, FigarellaBranger D, Cavenee WK. The 2016 World Health Organization classification of tumors of the central nervous system: A summary. Acta Neuropathol. 2016. 131: 803-20
28. Lozares-Cordero S, Font-Gómez JA, Gandía-Martínez A, Miranda-Burgos A, Méndez-Villamón A, Villa-Gazulla D. Treatment of cervical cancer with electronic brachytherapy. J Appl Clin Med Phys. 2019. 2: 78-86
29. Molinaro AM, Hervey-Jumper S, Morshed RA, Young J, Han SJ, Chunduru P. Association of maximal extent of resection of contrast-enhanced and non-contrast-enhanced tumor with survival within molecular subgroups of patients with newly diagnosed glioblastoma. JAMA Oncol. 2020. 6: 495-503
30. National Cancer Institute US National Institutes of Health. Common Terminology Criteria for Adverse Events (CTCAE Version 4.03). Available from: https://www.ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm [Last accessed on 2020 Oct 30].
31. NCCN Guidelines Version 2. 2020 Anaplastic Gliomas, Glioblastoma, Recurrence. Available from: https://www.nccn.org/professionals/physician_gls/default.aspx [Last accessed on 2020 Nov 25].
32. Rapp M, Baernreuther J, Turowski B, Steiger HJ, Sabel M, Kamp MA. Recurrence pattern analysis of primary glioblastoma. World Neurosurg. 2017. 103: 733-40
33. Ringel F, Pape H, Sabel M, Krex D, Bock HC, Misch M. Clinical benefit from resection of recurrent glioblastomas: Results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro Oncol. 2016. 18: 96-104
34. Salim N, Krivoshapkin AL, Gaytan AS, Marmazeev IV, Abdullaev O, Sergeev GS. The use of intraoperative radiation therapy in patients with recurrent gliomas of high malignancy. J Malignant Tumors. 2017. 3: 110
35. Sampson JH, Achrol A, Aghi MK, Bankiewicz KS, Bexon M, Brem S. MDNA55 survival in recurrent glioblastoma (rGBM) patients expressing the interleukin-4 receptor (IL4R) as compared to a matched synthetic control. J Clin Oncol. 2020. 38: 2513-3
36. Sanger F, Nicklen S, Coulson AR. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA. 1977. 74: 5463-7
37. Scharfen CO, Sneed PK, Wara WM, Larson DA, Phillips TL, Prados MD. High activity iodine-125 interstitial implant for gliomas. Int J Radiat Oncol Biol Phys. 1992. 24: 583-91
38. Silverstein MJ, Epstein MS, Lin K, Chen P, Khan S, Snyder L. Intraoperative radiation using low-kilovoltage X-rays for early breast cancer: A single site trial. Ann Surg Oncol. 2017. 24: 3082-7
39. Stummer W, Novotny A, Stepp H, Goetz C, Bise K, Reulen HJ. Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: A prospective study in 52 consecutive patients. J Neurosurg. 2000. 93: 1003-13
40. Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005. 352: 987-96
41. Stupp R, Wong ET, Kanner AA, Steinberg D, Engelhard H, Heidecke V. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: A randomised phase III trial of a novel treatment modality. Eur J Cancer. 2012. 48: 2192-202
42. Taal W, Oosterkamp HM, Walenkamp AM, Dubbink HJ, Beerepoot LV, Hanse MC. Single-agent bevacizumab or lomustine versus a combination of bevacizumab plus lomustine in patients with recurrent glioblastoma (BELOB trial): A randomised controlled phase 2 trial. Lancet Oncol. 2014. 15: 943-53
43. Takahashi J, Nagasawa S, Ikemoto MJ, Sato C, Sato M, Iwahashi H. Verification of 5-aminolevurinic radiodynamic therapy using a murine melanoma murine metastasis model. Int J Mol Sci. 2019. 20: 5155
44. Tatter SB, Shaw EG, Rosenblum ML, Karvelis KC, Kleinberg L, Weingart J. An inflatable balloon catheter and liquid 125I radiation source (GliaSite radiation therapy system) for treatment of recurrent malignant glioma: Multicenter safety and feasibility trial. J Neurosurg. 2003. 99: 297-303
45. Tsien C, Pugh S, Dicker AP, Raizer JJ, Matuszak MM, Lallana E. Randomized Phase II trial of re-irradiation and concurrent bevacizumab versus bevacizumab alone as treatment for recurrent glioblastoma (NRG Oncology/RTOG 1205): Initial outcomes and RT plan quality report. Int J Radiat Oncol Biol Phys. 2019. 105: S78
46. van Linde ME, Brahm CG, de Witt Hamer PC, Reijneveld JC, Bruynzeel AM, Vandertop WP. Treatment outcome of patients with recurrent glioblastoma multiforme: A retrospective multicenter analysis. J Neurooncol. 2017. 135: 183-92
47. Vuorinen V, Hinkka S, Färkkilä M, Jääskeläinen J. Debulking or biopsy of malignant glioma in elderly people-a randomised study. Acta Neurochir (Wien). 2003. 145: 5-10
48. Westphal M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC. A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro Oncol. 2003. 5: 79-88
49. Wick W, Puduvalli VK, Chamberlain MC, van den Bent MJ, Carpentier AF, Cher LM. Phase III study of enzastaurin compared with lomustine in the treatment of recurrent intracranial glioblastoma. J Clin Oncol. 2010. 28: 1168-74
50. 51. Yachi K, Watanabe T, Ohta T, Fukushima T, Yoshino A, Ogino A. Relevance of MSP assay for the detection of MGMT promoter hypermethylation in glioblastomas. Int J Oncol. 2008. 33: 469-75 52. Yamada K, Murayama Y, Kamada Y, Arita T, Kosuga T, Konishi H. Radiosensitizing effect of 5-aminolevulinic acid in colorectal cancer in vitro and in vivo. Oncol Lett. 2019. 17: 5132-8 53. Zhang Q, Xiao G, Sun Q, Zeng J, Mu X, Chen L. Radio-dynamic therapy (RDT) which combines cerenkov-induced PDT and RT. Int J Radiat Oncol Biol Phys. 2018. 102: E506