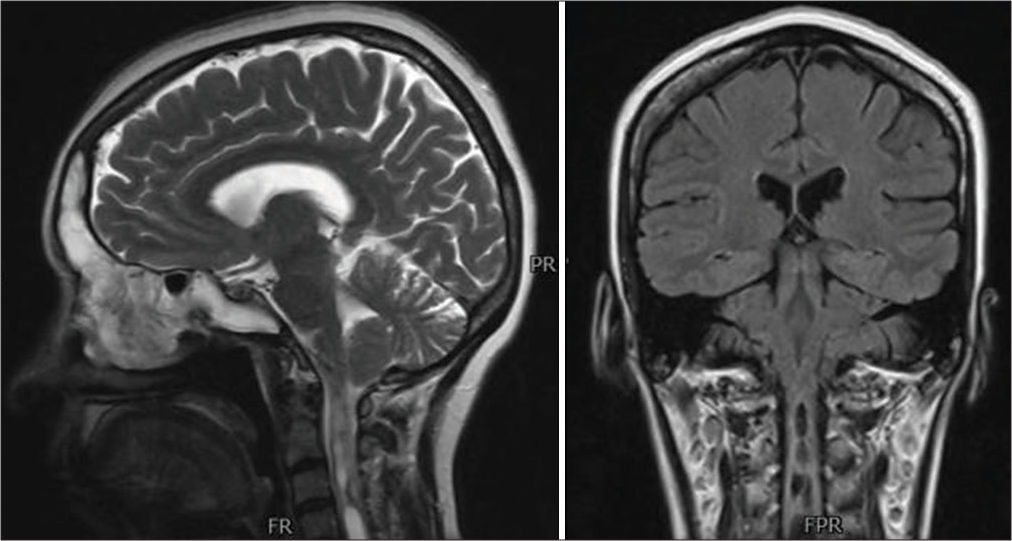
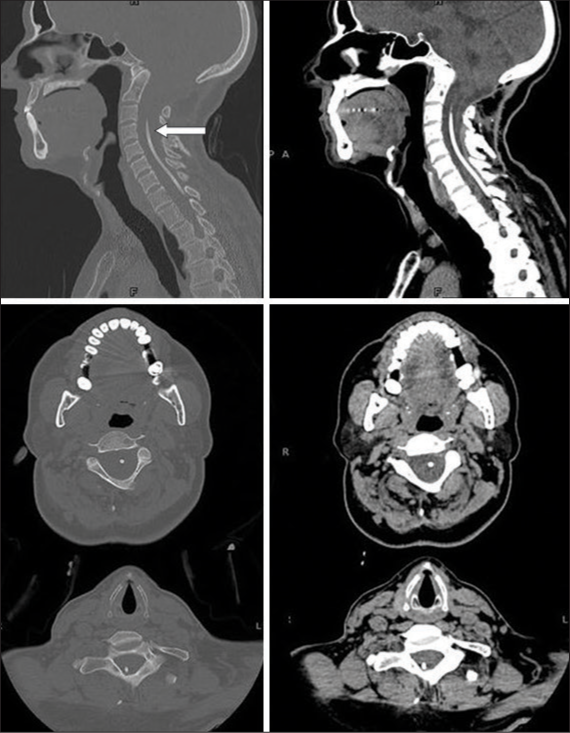
- Department of Neurosurgery, Medical College, The Aga Khan University, Karachi, Sindh, Pakistan
- Department of Neurosurgery, Aga Khan University Hospital, Karachi, Sindh, Pakistan.
Correspondence Address:
Muhammad Shahzad Shamim, Department of Neurosurgery, Aga Khan University Hospital, Karachi, Sindh, Pakistan.
DOI:10.25259/SNI_383_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Syed Faisal Nadeem1, Ahmer Nasir Baig2, Qurat ul Ain Tariq2, Muhammad Shahzad Shamim2. Spinal arachnoiditis and syringomyelia: Review of literature with emphasis on postinfectious inflammation and treatment. 15-Jul-2022;13:299
How to cite this URL: Syed Faisal Nadeem1, Ahmer Nasir Baig2, Qurat ul Ain Tariq2, Muhammad Shahzad Shamim2. Spinal arachnoiditis and syringomyelia: Review of literature with emphasis on postinfectious inflammation and treatment. 15-Jul-2022;13:299. Available from: https://surgicalneurologyint.com/surgicalint-articles/11724/
Abstract
Background: Arachnoiditis refers to chronic inflammation of the arachnoid mater and subarachnoid space due to three major etiologies: chemical, mechanical, and postinfectious. As a rare disease with variable symptomatology and severity at presentation, arachnoiditis can be extremely debilitating with many complications, prominent among which is syringomyelia.
Methods: We reviewed current literature concerning postinfectious spinal arachnoiditis and associated syringomyelia with emphasis on the treatment options that have been used to date and discuss their respective benefits and drawbacks.
Results: It is critical to understand the natural history and potential complications of patient with postinfectious arachnoiditis. Surgical and medical treatments both have their own merits and demerits. Different surgical approaches have been employed with variable success rates.
Conclusion: At present, no consensus exists regarding management of these patients due to the variable nature of the disease that affects treatment efficacy; however, surgical intervention in selected cases may be beneficial.
Keywords: Arachnoiditis, Meninges, Scarring, Shunt, Syringomyelia
INTRODUCTION
Arachnoiditis is a persistent inflammation of the arachnoid mater and the subarachnoid space due to a diverse range of etiologies that can be grouped into three major categories: chemical, mechanical, and postinfectious.[
REVIEW OF LITERATURE
Inflammation in the arachnoid mater results in the deposition of collagen and generation of scar tissue that encapsulates nerve roots of the spinal cord, tethering them together and depriving them of adequate blood supply. This leaves the nerve roots undernourished, hypoxic, and exposed to waste metabolites, eventually culminating in spinal nerve atrophy and its associated neurologic deficits.[
Arachnoiditis as a condition has variable severity and clinical manifestations with many of the cases being mild or subclinical and hence going undiagnosed. These factors, together with the absence of a unified nomenclature to describe the condition, make it difficult to estimate epidemiological parameters (most notably the incidence) for the disease.[
A review of literature suggests a number of different infectious agents to be involved in causing arachnoiditis including Mycobacterium tuberculosis, Cryptococcus, Coccidioides, Taenia solium (neurocysticercosis), and Histoplasma to name a few.[
Clinical arachnoiditis presents very similarly to other diseases involving spinal nerve compression with a range of symptoms including burning lower back pain that radiates down the legs – urinary urgency, frequency, and incontinence – muscle spasms in the back and legs – sensory deficits below the lesion site – and paraplegia or paraparesis. Due to the insidious development of the disease, finding an underlying cause can often be difficult.[
Syringomyelia has been frequently associated with arachnoiditis [ The meningeal scarring produces pathologic changes in the meningeal vessels and in extension those supplying the spinal cord, causing focal ischemia and resulting necrosis which causes syrinx formation, The scarring itself impedes communication between the subdural and subarachnoid spaces and impedes cerebrospinal fluid flow through the spinal cord, causing it to move out of these spaces and into the central canal, producing a syrinx.
Another theory for syringomyelia formation secondary to a tethered spinal cord (a spinal cord adhered to the tissues surrounding it, often a sequela of arachnoiditis) is proposed by Tsitouras and Sgouros.[
Complete alleviation of the pain and debility associated with spinal arachnoiditis is often not possible and the target of intervention is to reduce the pain experienced by the patient to the greatest extent.[
Surgical management of syringomyelia in patients with preexisting arachnoiditis also appears to have mixed success rates in the current literature. Davidoff et al. review the treatment received by eight patients who had syringomyelia secondary to craniocervical junction arachnoiditis.[
Zuev et al. discussed their experience on surgically treating 34 patients with syringomyelia secondary to adhesive arachnoiditis.[
Kannapadi et al.[
Naito et al. provided a retrospective analysis of the safety and efficacy of syringoperitoneal shunting in patients with syringomyelia secondary to spinal adhesive arachnoiditis. Average functionality (measured using the modified McCormick functional schema) was preserved at 4.8 at the most recent follow-up visit (compared to 4.7 preoperatively) while there was significant improvement in pain (measured using the sensory pain scale), which reduced from 2.9 preoperatively to 2.5 at the most recent follow-up visit. About 93.3% of patients reviewed showed radiographic improvement in their syringomyelia and there were no reported recurrences postoperatively. Comparing it to microsurgical arachnoidolysis, Naito et al. champion CSF shunt placement as a safer treatment option, since arachnoidolysis is both more technically demanding and associated with a high risk of postoperative neurologic deterioration due to iatrogenic meningeal scarring.[
CONCLUSION
The literature concerning management of patients with infectious arachnoiditis and concomitant syringomyelia lacks consensus on whether surgical management is better than conservative management or vice versa and also provides different levels of efficacy for each based on the extent and severity of the underlying disease. A number of different surgical techniques have also been elucidated with their own benefits and drawbacks in individual conditions, they were applied in. It would thus be prudent to conclude that management of the patient population in question should be closely tailored to the individual patients’ needs, taking into account their preoperative level of functionality and extent of disease. Imaging studies may prove helpful for selecting treatment options for these patients.
Declaration of patient consent
Patients’ consent not required as patients’ identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Alatorre-Fernández CP, Venzor-Castellanos JP, ContrerasCabrera JA, Fuentes-venegas A, Castro-Martínez E. Arachnoiditis and tuberculous cortical encephalitis in an HIV+ patient. Gaceta Medica Mexico. 2009. 145: 239-40
2. Anderson TL, Morris JM, Wald JT, Kotsenas AL. Imaging appearance of advanced chronic adhesive arachnoiditis: A retrospective review. AJR Am J Roentgenol. 2017. 209: 648-55
3. Caplan LR, Norohna AB, Amico LL. Syringomyelia and arachnoiditis. J Neurol Neurosurg Psychiatry. 1990. 53: 106-13
4. Cárdenas G, Aristizábal S, Salinas C, Delgado-Hernández R, Angeles-Morales V, Soto-Hernández J. Coccidioidal meningitis in non-AIDS patients. A case series at a Mexican neurological referral center. Clin Neurol Neurosurg. 2020. 196: 106011
5. Cherian A, Ajitha KC, Iype T, Divya KP. Neurotuberculosis: An update. Acta Neurol Belg. 2021. 121: 11-21
6. Conermann T.editors. Arachnoiditis. Treasure Island, FL: Stat Pearls Publishing; 2021. p.
7. Davidoff CL, Liu S, Wong JHY, Koustais S, Rogers JM, Stoodley MA. Treatment of syringomyelia in patients with arachnoiditis at the craniocervical junction. World Neurosurg. 2017. 107: 565-73
8. Demetriades AK. Arachnoiditis and Chiari I malformation. Acta Neurochir (Wien). 2021. 163: 227-8
9. Di Ieva A, Barolat G, Tschabitscher M, Rognone E, Aimar E, Gaetani P. Lumbar arachnoiditis and thecaloscopy: Brief review and proposed treatment algorithm. Cent Eur Neurosurg. 2010. 71: 207-12
10. Esses SI, Morley TP. Spinal arachnoiditis. Can J Neurol Sci. 1983. 10: 2-10
11. Fehlings MG, Bernstein M. Syringomyelia as a complication of tuberculous meningitis. Can J Neurol Sci. 1992. 19: 84-7
12. Garg RK, Malhotra HS, Gupta R. Spinal cord involvement in tuberculous meningitis. Spinal Cord. 2015. 53: 649-57
13. Guyer DW, Wiltse LL, Eskay ML, Guyer BH. The long-range prognosis of arachnoiditis. Spine (Phila Pa 1976). 1989. 14: 1332-41
14. Hui AC, Chan YL, Kay R. Syrinx and tuberculoma formation in tuberculous arachnoiditis. Can J Neurol Sci. 2001. 28: 148-9
15. Jesrani G, Kaur J, Gupta M, Sawal N. Syringomyelia as a delayed complication of lumbar-sacral adhesive arachnoiditis in Pott’s disease. BMJ Case Rep CP. 2020. 13: e234032
16. Kalina J. Arachnoiditis. J Pain Palliat Care Pharmacother. 2012. 26: 176-7
17. Kannapadi NV, Alomari SO, Caturegli G, Bydon A, Cho SM. Management of syringomyelia associated with tuberculous meningitis: A case report and systematic review of the literature. J Clin Neurosci. 2021. 87: 20-5
18. Kaynar MY, Koçer N, Gençosmanoğlu BE, Hanci M. Syringomyelia as a late complication of tuberculous meningitis. Acta Neurochir (Wien). 2000. 142: 935-8
19. Klekamp J, Batzdorf U, Samii M, Bothe HW. Treatment of syringomyelia associated with arachnoid scarring caused by arachnoiditis or trauma. J Neurosurg. 1997. 86: 233-40
20. Klekamp J, Iaconetta G, Batzdorf U, Samii M. Syringomyelia associated with foramen magnum arachnoiditis. J Neurosurg. 2002. 97: 317-22
21. Morisako H, Takami T, Yamagata T, Chokyu I, Tsuyuguchi N, Ohata K. Focal adhesive arachnoiditis of the spinal cord: Imaging diagnosis and surgical resolution. J Craniovertebr Junction Spine. 2010. 1: 100-6
22. Naito K, Yamagata T, Ohata K, Takami T. Safety and efficacy of syringoperitoneal shunting with a programmable shunt valve for syringomyelia associated with extensive spinal adhesive arachnoiditis: Technical note. World Neurosurg. 2019. 132: 14-20
23. Nardone R, Alessandrini F, Tezzon F. Syringomyelia following Listeria meningoencephalitis: Report of a case. Neurol Sci. 2003. 24: 40-3
24. Nash TE, O’Connell EM. Subarachnoid neurocysticercosis: Emerging concepts and treatment. Curr Opin Infect Dis. 2020. 33: 339-46
25. Panackal AA, Komori M, Kosa P, Khan O, Hammoud DA, Rosen LB. Spinal arachnoiditis as a complication of cryptococcal meningoencephalitis in Non-HIV previously healthy adults. Clin Infect Dis. 2017. 64: 275-83
26. Phanthumchinda K, Kaoropthum S. Syringomyelia associated with post-meningitic spinal arachnoiditis due to Candida tropicalis. Postgrad Med J. 1991. 67: 767-9
27. Rangel-Castilla L, Hwang SW, White AC, Zhang YJ. Neuroendoscopic diagnosis of central nervous system histoplasmosis with basilar arachnoiditis. World Neurosurg. 2012. 77: 399.E9-13
28. Rodriguez ED. Central nervous system tuberculosis following delayed and initially missed lung miliary tuberculosis: A case report. Infez Med. 2018. 26: 270-5
29. Snyder P. Chiari malformation and syringomyelia. Radiol Technol. 2008. 79: 555-8
30. Todeschi J, Chibbaro S, Gubian A, Pop R, Proust F, Cebula H. Spinal adhesive arachnoiditis following the rupture of an Adamkiewicz aneurysm: Literature review and a case illustration. Neurochirurgie. 2018. 64: 177-82
31. Tsitouras V, Sgouros S. Syringomyelia and tethered cord in children. Childs Nerv Syst. 2013. 29: 1625-34
32. Turnbull IM, Shulman R, Woodhurst WB. Thalamic stimulation for neuropathic pain. J Neurosurg. 1980. 52: 486-93
33. Wright MH, Denney LC. A comprehensive review of spinal arachnoiditis. Orthop Nurs. 2003. 22: 215-9
34. Zuev AA, Lebedev VB, Pedyash NV, Epifanov DS, Levin RS. Treatment of syringomyelia associated with adhesive arachnoiditis. Zh Vopr Neirokhir Im N N Burdenko. 2017. 81: 39-47