- Department of Neurosurgery, Neurological Institute of Thailand, Bangkok, Thailand
- Department of Neuroradiology, Neurological Institute of Thailand, Bangkok, Thailand
- Department of Radiology, Bumrungrad International Hospital, Bangkok, Thailand.
Correspondence Address:
Prasert Iampreechakul, Department of Neurological Surgery, Neurological Institute of Thailand, 312 Rachawithi Road, Khwaeng Thung Phaya Thai, Bangkok, Thailand.
DOI:10.25259/SNI_4_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Prasert Iampreechakul1, Songpol Chuntaroj2, Yodkhwan Wattanasen2, Sunisa Hangsapruek2, Punjama Lertbutsayanukul2, Somkiet Siriwimonmas3. Spontaneous regression of extradural high-flow vascular malformation in spinal arteriovenous metameric syndrome (SAMS): A unique case report. 05-May-2023;14:163
How to cite this URL: Prasert Iampreechakul1, Songpol Chuntaroj2, Yodkhwan Wattanasen2, Sunisa Hangsapruek2, Punjama Lertbutsayanukul2, Somkiet Siriwimonmas3. Spontaneous regression of extradural high-flow vascular malformation in spinal arteriovenous metameric syndrome (SAMS): A unique case report. 05-May-2023;14:163. Available from: https://surgicalneurologyint.com/surgicalint-articles/12314/
Abstract
Background: Spinal arteriovenous metameric syndrome (SAMS) is a rare nonhereditary genetic vascular disorder, involving multiple layers of tissues in the same metameric level. Spontaneous regression of SAMS has never been reported in the medical literature.
Case Description: A 42-year-old woman presented with intermittent low back pain for 6 months. Magnetic resonance imaging of the thoracolumbar spine incidentally found clusters of spinal vascular malformations involving spinal cord, vertebral bodies, epidural space, and paraspinal muscles. There was no sign of venous congestion. Magnetic resonance angiography and spinal angiography revealed intradural spinal cord arteriovenous malformation (SCAVM) at the level of T10-11 and extradural high-flow osseous arteriovenous fistula. Due to asymptomatic SAMS and a high risk of anterior spinal arterial compromise during treatment, conservative treatment was considered in our patient. Spinal angiography obtained 8 years after initial angiography demonstrated significant regression of extradural component of SAMS and stable intradural SCAVM.
Conclusion: We describe a unique case of SAMS with spontaneous regression of extradural component during a long-term observation period.
Keywords: Spinal arteriovenous metameric syndrome, spinal vascular malformation, spinal cord arteriovenous malformation, spinal epidural arteriovenous fistula, spontaneous resolution
INTRODUCTION
Spinal arteriovenous metameric syndrome (SAMS), a rare subtype of spinal cord arteriovenous malformations (SCAVMs), is a complex nonhereditary genetic vascular disorder, affecting spinal cord, bony structure, epidural space, and/or paraspinal soft tissue or muscles in the same metameric level.[
CASE DESCRIPTION
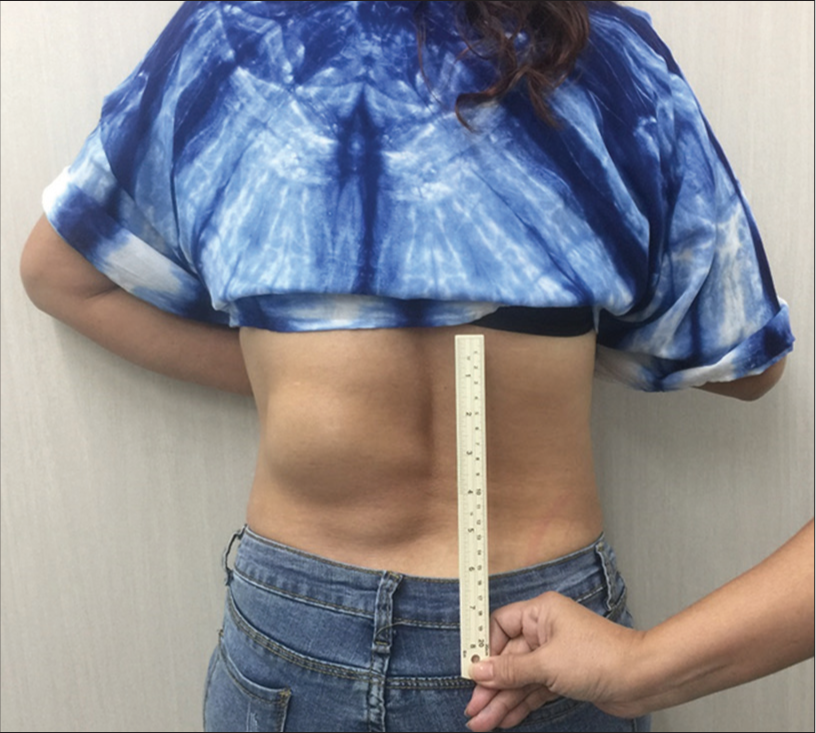
A 42-year-old woman presented with intermittent low back pain for 6 months without history of trauma. She went to the local hospital and obtained plain radiography of lumbar spine, revealing scoliosis and mixed lytic and sclerotic lesions at L1 and L2 vertebral bodies with collapse of left side of L1 vertebral body. Magnetic resonance imaging (MRI) of the thoracolumbar spine was performed and incidentally found clusters of vascular malformations. SCAVM was presumably diagnosed, and the patient was transferred to our institute for further investigation and proper treatment. Physical examination showed a large well-circumscribed mass, which is approximately 14 cm in diameter, over the left side of the lower back [
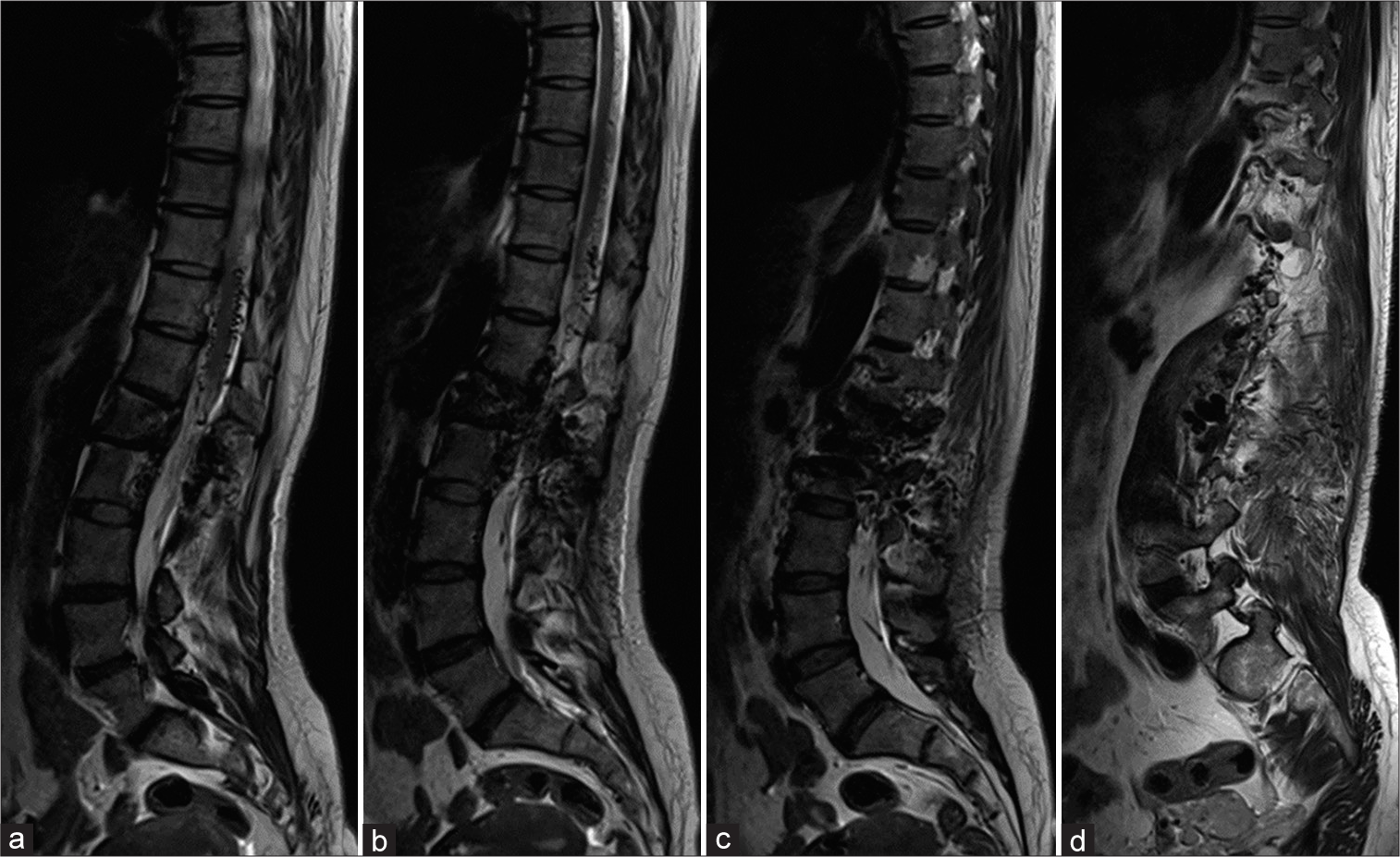
MRI of the thoracolumbar spine demonstrated abnormal tortuous and dilated flow voids surrounding the lower spinal cord and conus medullaris, particularly at posterior surface of T10-11 level, and at left-side of L1 and L2 vertebral bodies, epidural space, neural foramina, and paraspinal muscles including psoas major, multifidus, erector spinae, and quadratus lumborum. The affected muscles from T12 to L4 vertebral levels were hypertrophy and markedly enhanced after administration of contrast material with mild hyperintense T2 signal change on T2-weighted sequence. There was no signal intensity change of the spinal cord [
Figure 2:
(a-d) Sequential sagittal T2-weighted images from midline to left side of the thoracolumbar spine reveal abnormal tortuous and dilated flow voids surrounding the lower spinal cord and conus medullaris, particularly at posterior surface of T10-11 level, and at left-side of L1 and L2 vertebral bodies, neural foramina, and paraspinal muscles. There is no signal intensity change of spinal cord.
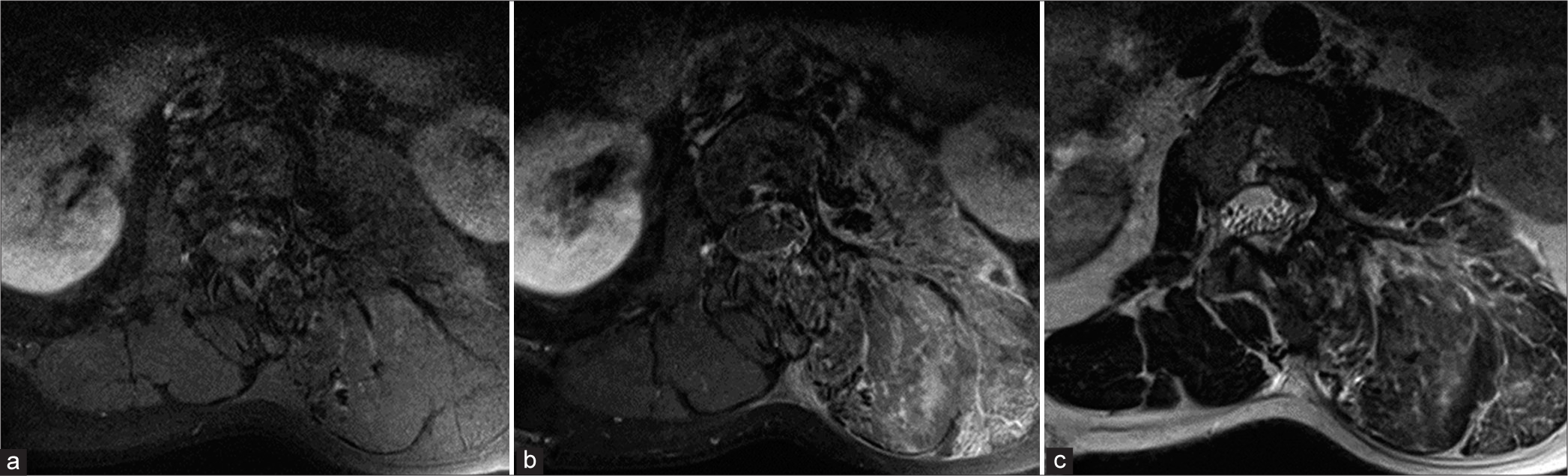
Figure 4:
Axial T1-weighted (a) without and (b) with contrast-enhanced, and (c) T2-weighted images at the level of L2 vertebral body disclose multiple abnormal dilated and tortuous flow voids at left side of vertebral body, epidural space, and nearby paraspinal muscles with associated diffuse enlargement and enhancement of affected muscles.
Figure 5:
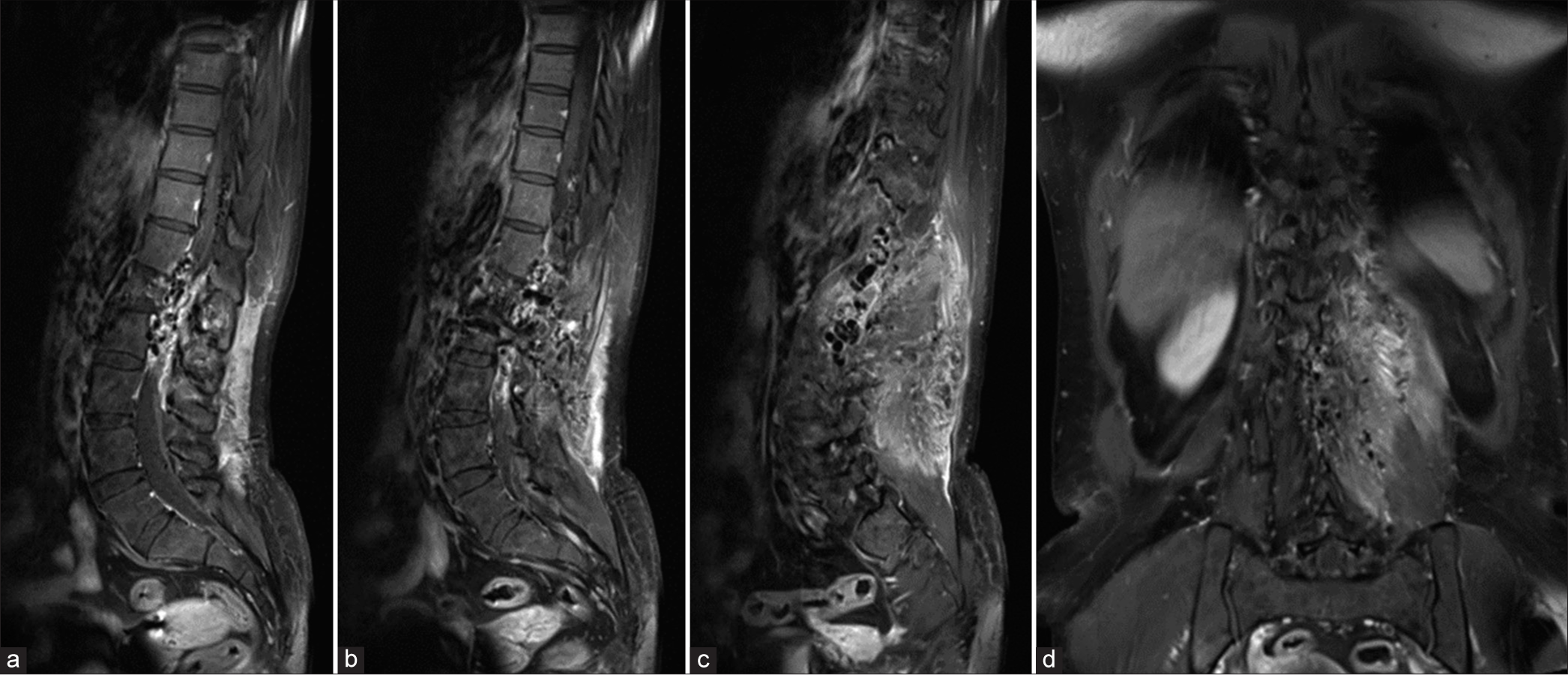
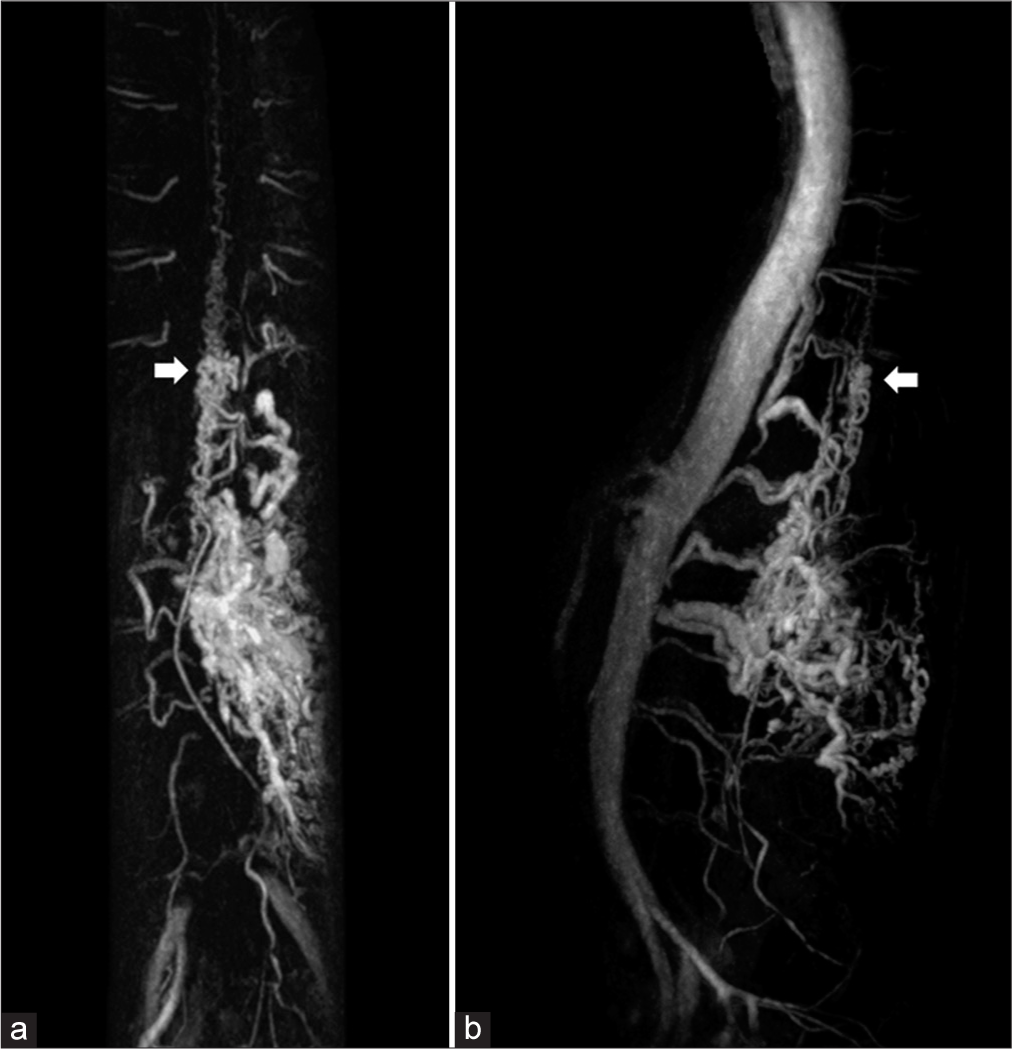
Contrast-enhanced magnetic resonance angiography of the thoracolumbar spine. (a) Coronal and (b) sagittal maximum intensity projection images reveal clusters of vascular malformations including intradural spinal cord arteriovenous malformation (arrows) at the level of T10-11 and extradural paravertebral arteriovenous malformation from T12 to L2 vertebral levels with significant enlargement of bilateral T 12 intercostal, L1, and L2 segmental arteries, particularly on the left.
Figure 6:
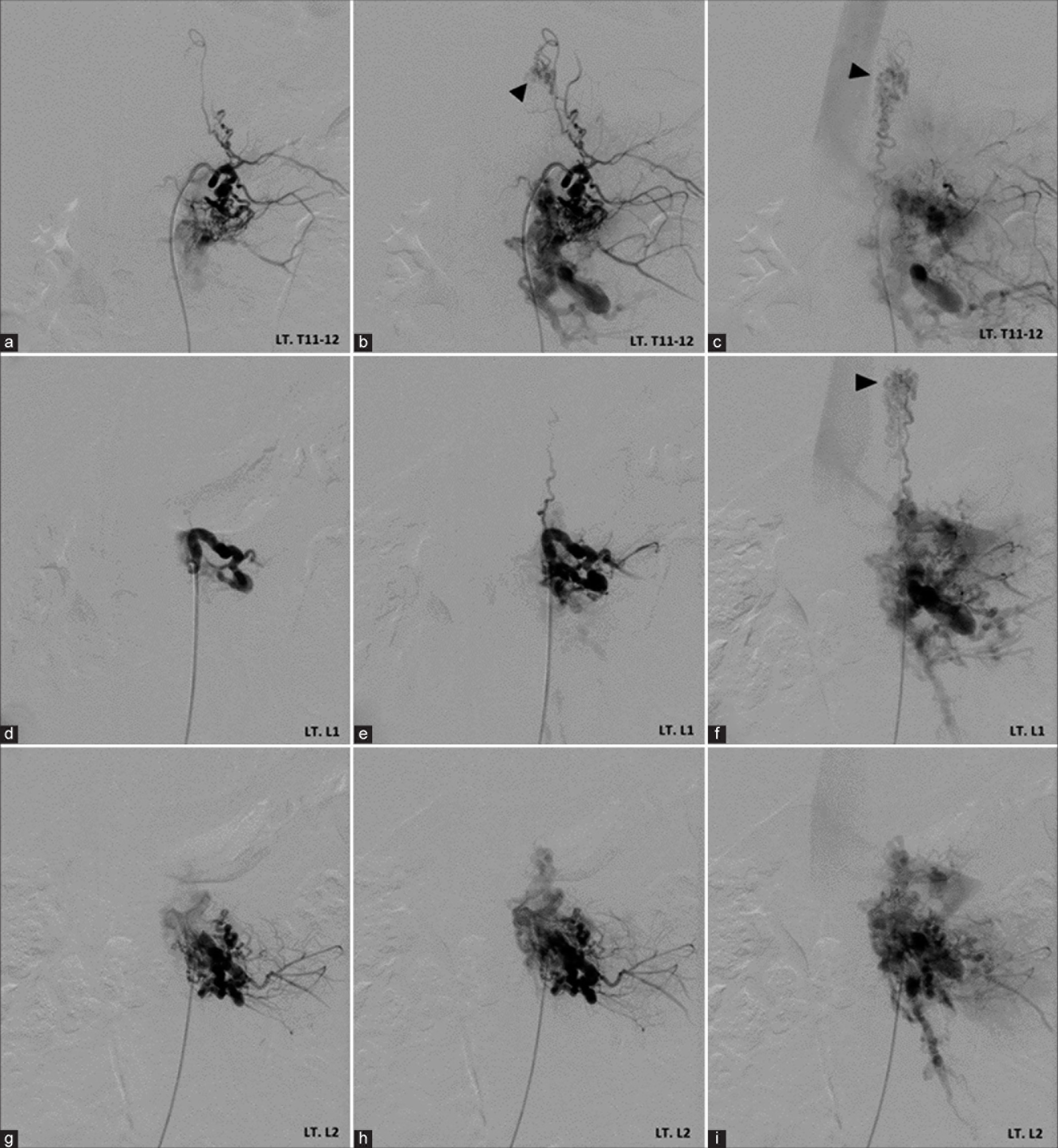
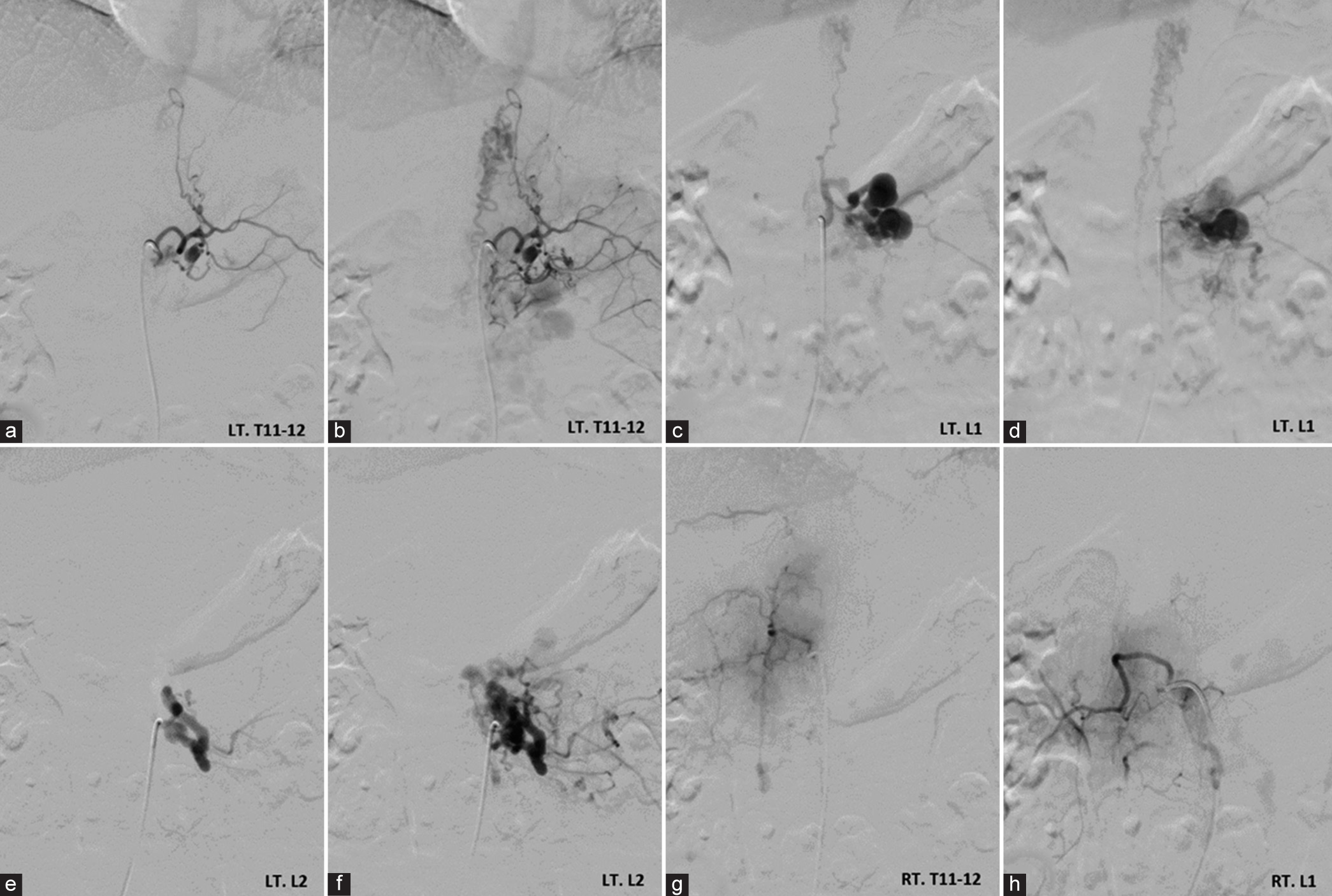
Anteroposterior views of the left (a-c) T11-12 intercostal, (d-f) L1, and (g-i) L2 segmental arteries angiography demonstrate spinal cord arteriovenous malformation (arrowheads) at the T10-11 level, supplied by left posterior spinal arteries that receives radiculopial branches from the left T11intercostal and L1 segmental arteries, and spinal osseous epidural arteriovenous fistula fed by dilated radicular arteries from left T12 intercostal, L1, and L2 segmental arteries converging to the venous lake at the level of L1 and L2 with venous drainage through enlarged epidural and paravertebral venous plexus.
Figure 8:
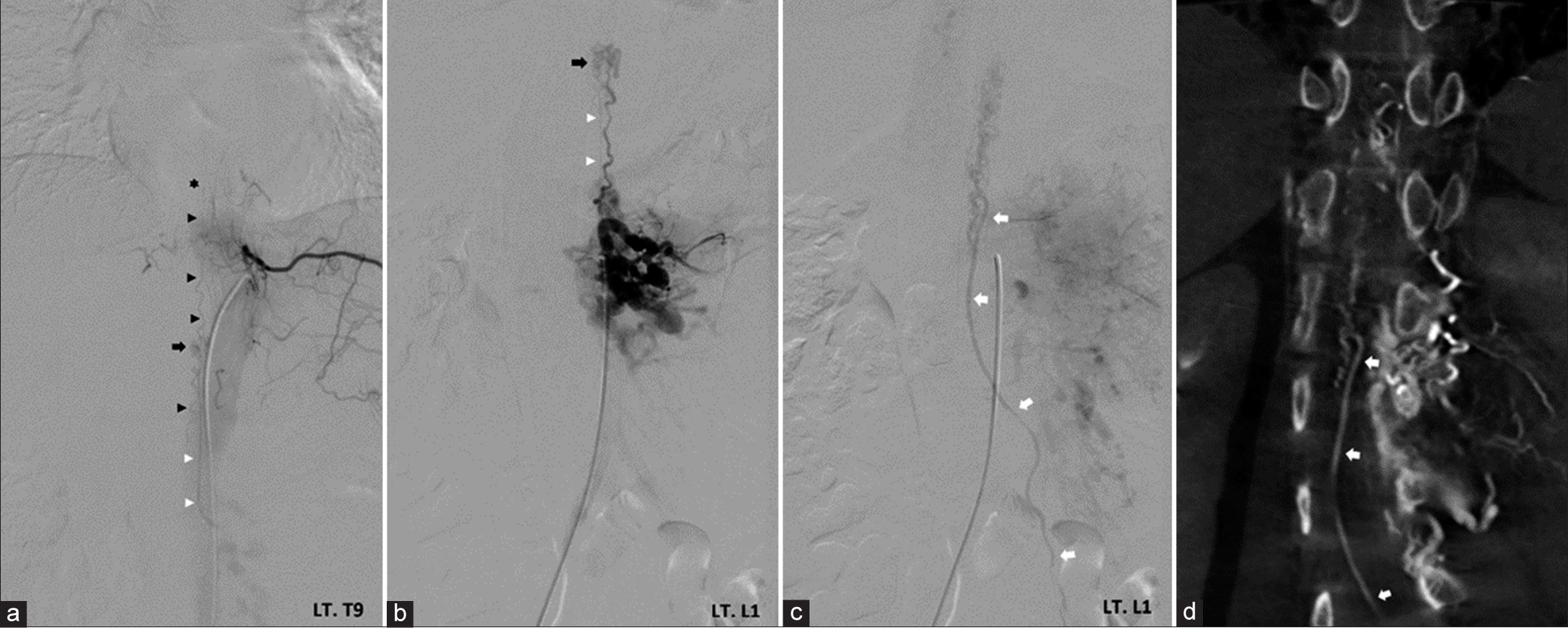
(a) Anteroposterior view of the left T9 intercostal artery angiography demonstrates spinal cord arteriovenous malformation (SCAVM) (black arrow) at the T10-11 level supplied by an anterior spinalartery (black arrowheads) with a characteristic hairpin turn (asterisk). Another feeding artery from the radiculomedullary branch (white arrowheads) from the left L1 segmental artery is identified. Anteroposterior views of the left L1 segmental artery angiography in (b) arterial and (c) venous phases reveal the same SCAVM (black arrow) fed by the radiculomedullary branch (white arrowheads) and dilated radiculopial artery. There is a high-flow spinal osseous arteriovenous fistula supplied by enlarged radicular arteries from the left L1 segmental artery with caudal drainage into perimedullary veins and subsequent drainage to the dilated left sacral radicular vein (white arrows). (d) Coronal multiplanar reconstruction image of angiographic computerized tomography clearly illustrates the enlarged left radicular vein (white arrows).
Figure 9:
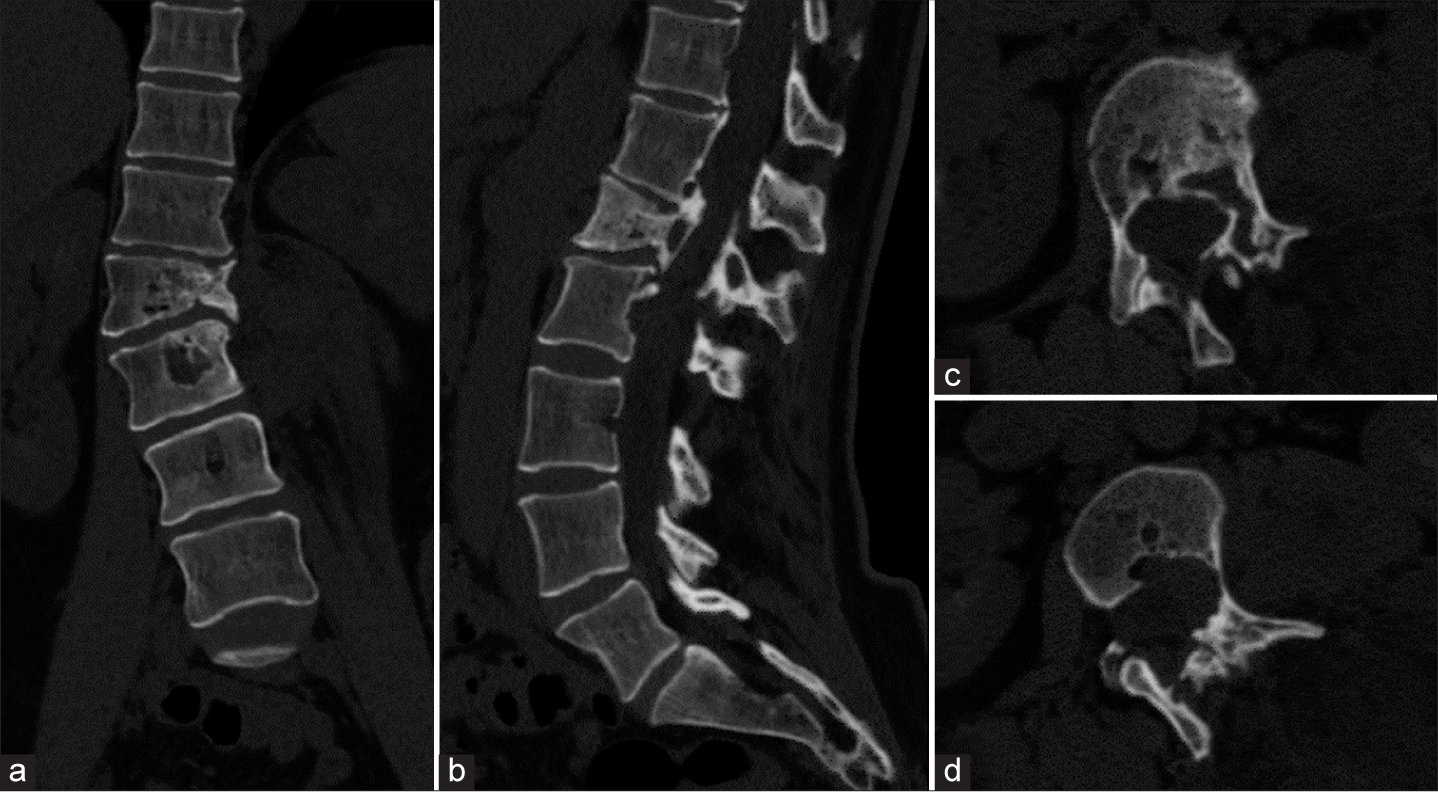
Computed tomography scan of the lumbar spine. (a) Coronal, (b) sagittal, and axial views at the level of (c) L1 and (d) L2 reveal dextroscoliosis and decrease of left sided L1 vertebral height with associated sclerotic change. There are multiple well-defined tubular and round-shaped osteolytic lesions and erosion with sclerotic rims involving particularly left-sided vertebral bodies and posterior elements of L1 and L2, corresponding with the dilated venous lake.
All above-mentioned imaging features contributed to a definite diagnosis of SAMS at T10-11–L2 levels. Surgical or endovascular treatment of these lesions was considered a high risk of anterior spinal arterial compromise. In addition, she was asymptomatic from vascular malformations. Therefore, the patient was kept for continuous observation and has attended our outpatient department every year without missing any follow-up visit. At 3rd year of the follow-up, MRI and MRA were performed and showed unchanged of the lesions. At 4th year of the follow-up, spinal angiography was scheduled to perform but the procedure was postponed due to the COVID-19 pandemic. Spinal angiography obtained 8 years after initial angiography surprisingly showed significant regression of extradural component of SAMS and stable intradural SCAVM [
Figure 10:
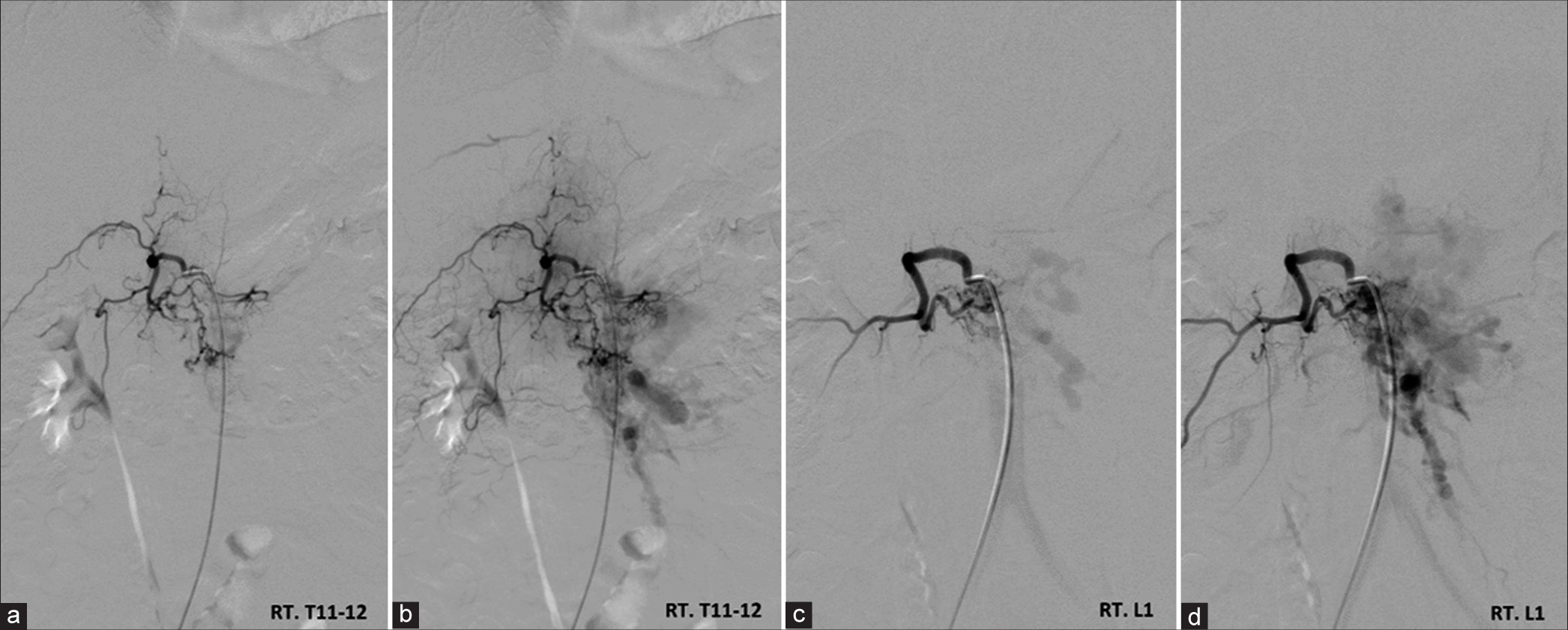
Spinal angiography obtained 8 years after initial angiography. Anteroposterior views of the (a and b) left T11-12, (c and d) left L1 segmental, (e and f) left L2 segmental, (g) right T11-12 intercostal, and (h) right L1 segmental arteries angiography demonstrate significant regression of the spinal osseous epidural arteriovenous fistula at the level of L1 and L2. Spinal cord arteriovenous malformation at the level of T 10-11 remains stable in size and angioarchitecture.
DISCUSSION
Since 1915, Cobb syndrome was characterized by the concurrent findings of a spinal vascular malformation and a vascular skin lesion affecting the corresponding dermatome.[
SAMS is classified as extradural-intradural arteriovenous malformations with two or more vascular malformations involving spinal cord, bone, paraspinal musculature, subcutaneous tissues, and/or skin in one or several metameric segments.[
Due to the rarity and poorly understood pathophysiology of SAMS, optimal management remains unclear. Treatment options include endovascular treatment, surgical resection, a combined approach, or conservative management. Complete removal of SAMS is often difficult without significant risk of neurological morbidity. It is difficult that a complex SAMS could be cured by only endovascular treatment. For this reason, the aim of endovascular treatment is symptom improvement or stabilization of a progressive disease with poor long-term functional prognosis. Spinal angiography remains the gold standard to evaluate the angioarchitecture for therapeutic decisions. Careful assessment of symptoms of patients and causative vascular pathology is required to perform treatment at the appropriate time. Long-term clinical follow-up and periodic spinal angiography are imperative to control this dynamic disease.[
In the present study, SAMS included intradural SCAVM and extradural high-flow SOEAVF. Spinal epidural arteriovenous fistulas (SEAVFs) are likely acquired lesions and usually present with congestive myelopathy secondary to intradural venous drainage and may present with compressive symptoms resulting from exclusive extradural venous drainage.[
Drake et al.[
To the best of our knowledge, spontaneous regression of SAMS has never been reported in the medical literature. Spontaneous regression of extradural high-flow vascular malformation in SAMS occurred in our patient during a long-term observation period. Based on the study of Kang et al.,[
Spontaneous regression of high-flow spinal extradural AVFs remains unclear and has rarely been reported.[
Because the natural history of SAMs remains poorly understood. Therefore, further long-term clinical assessment and radiological follow-up in our patient are required to monitor the progression of intradural SCAVM and recanalization of extradural SOEAVF.
CONCLUSION
SAMS is a rare subtype of SCAVM. This syndrome occurs from a somatic mutation developing in the region of the neural crest or adjacent mesoderm before migration and differentiation, affecting the corresponding metameric distribution. We describe a unique case of SAMS with spontaneous regression of extradural component during a long-term observation period.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Bhattacharya JJ, Luo CB, Suh DC, Alvarez H, Rodesch G, Lasjaunias P. Wyburn-mason or bonnet-dechaume-blanc as cerebrofacial arteriovenous metameric syndromes (CAMS). A new concept and a new classification. Interv Neuroradiol. 2001. 7: 5-17
2. Choi IS. Spinal arteriovenous metameric syndrome: Angioarchitecture and their prognosis. AJNR Am J Neuroradiol. 2013. 34: 464-5
3. Cobb S. Haemangioma of the spinal cord: Associated with skin naevi of the same metamere. Ann Surg. 1915. 62: 641-9
4. Drake B, Patro S, Quateen A, Cora EA, Finitsis S, Sinclair J. Metameric spinal AVM: Long-term symptomatic relief achieved by embolization of the extradural component. Interv Neuroradiol. 2019. 25: 469-73
5. Elkordy A, Endo T, Sato K, Sonoda Y, Takahashi A, Tominaga T. Exclusively epidural spinal metameric arteriovenous shunts: Case report and literature review. Spine J. 2015. 15: e15-22
6. Iampreechakul P, Liengudom A, Wangtanaphat K, Tirakotai W, Wattanasen Y, Lertbutsayanukul P. Spinal epidural arteriovenous fistula with intradural venous reflux: The possibility of acquired origin caused by spinal stenosis and/or disc herniation. Clin Neurol Neurosurg. 2021. 207: 106794
7. Iampreechakul P, Tirakotai W, Lertbutsayanukul P, Thammachantha S, Siriwimonmas S. Spontaneous spinal osseous epidural arteriovenous fistula with long segments of prominent epidural venous drainage causing severe compressive thoracic myelopathy successfully treated with combined endovascular and surgical treatments: A case report and review of the literature. Asian J Neurosurg. 2020. 15: 1041-9
8. Iampreechakul P, Tirakotai W, Tanpun A, Wattanasen Y, Lertbusayanukul P, Siriwimonmas S. Spontaneous resolution of direct carotid-cavernous fistulas: Case series and literature review. Interv Neuroradiol. 2019. 25: 71-89
9. Kang J, Gregg L, Gailloud P. Spontaneous resolution of low-flow spinal arteriovenous fistulas. Neuroradiology. 2017. 59: 1003-12
10. Kinoshita M, Asai A, Komeda S, Yoshimura K, Takeda J, Uesaka T. Spontaneous regression of a spinal extradural arteriovenous fistula after delivery by cesarean section. Neurol Med Chir (Tokyo). 2009. 49: 313-5
11. Lenck S, Nicholson P, Tymianski R, Hilditch C, Nouet A, Patel K. Spinal and paraspinal arteriovenous lesions. Stroke. 2019. 50: 2259-69
12. Lin YH, Huang YC, Tsuang FY. Spontaneous closure of an incidental high-flow paravertebral arteriovenous fistula caused by vertebral giant cell tumor curettage: Illustrative case. J Neurosurg Case Lessons. 2021. 1: CASE2116
13. Niimi Y, Uchiyama N, Elijovich L, Berenstein A. Spinal arteriovenous metameric syndrome: Clinical manifestations and endovascular management. AJNR Am J Neuroradiol. 2013. 34: 457-63
14. Nijenhuis RJ, Sluzewski M, van Rooij WJ. Contrast agent induced obliteration of a spinal arteriovenous malformation. AJNR Am J Neuroradiol. 2012. 33: E1-3
15. Panciani PP, Fontanella M, Crobeddu E, Schatlo B, Bergui M, Ducati A. Spontaneous occlusion of a spinal arteriovenous malformation: Is treatment always necessary?. J Neurosurg Spine. 2010. 12: 397-401
16. Rodesch G, Hurth M, Alvarez H, Tadié M, Lasjaunias P. Classification of spinal cord arteriovenous shunts: Proposal for a reappraisal--the Bicêtre experience with 155 consecutive patients treated between 1981 and 1999. Neurosurgery. 2002. 51: 374-80
17. Spetzler RF, Detwiler PW, Riina HA, Porter RW. Modified classification of spinal cord vascular lesions. J Neurosurg. 2002. 96: 145-56
18. Takai K, Usui M. Spontaneous thrombosis of a spinal conus perimedullary arteriovenous fistula. Case report. Neurol Med Chir (Tokyo). 2012. 52: 103-6
19. Wong IY, Batista LL, Alvarez H, Lasjaunias PL. Craniofacial arteriovenous metameric syndrome (CAMS) 3--a transitional pattern between CAM 1 and 2 and spinal arteriovenous metameric syndromes. Neuroradiology. 2003. 45: 611-5