- Department of Neurosurgery, Government Medical College, Kota, Rajasthan, India
- Department of Anesthesiology and Intensive Care, Government Medical College, Kota, Rajasthan, India
- Department of Obstetrics and Gynecology, Government Medical College, Kota, Rajasthan, India
Correspondence Address:
Sachidanand Gautam
Department of Obstetrics and Gynecology, Government Medical College, Kota, Rajasthan, India
DOI:10.4103/2152-7806.195229
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Sachidanand Gautam, Rajesh K. Meena, Shyam C. Meena, Bhawana Gautam. Retrospective analysis of prognostic factors in dengue infected patients with intracranial bleed. 05-Dec-2016;7:
How to cite this URL: Sachidanand Gautam, Rajesh K. Meena, Shyam C. Meena, Bhawana Gautam. Retrospective analysis of prognostic factors in dengue infected patients with intracranial bleed. 05-Dec-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/retrospective-analysis-of-prognostic-factors-in-dengue-infected-patients-with-intracranial-bleed/
Abstract
Background:Dengue is one of the most common mosquito-transmitted arboviral disease of tropical and a few subtropical areas in the world. It is estimated that approximately 100 million cases occur per year and approximately 2.5 billion people are at risk of developing dengue infection. Hemorrhagic complications causing encephalopathy are quite rare but fatal consequences of this deadly disease. This study was conducted to discuss the prognostic factors in the management of intracranial hemorrhage in dengue infected patients.
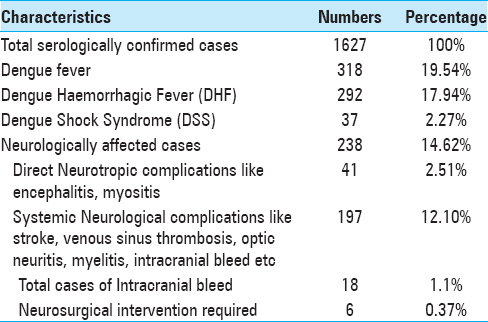
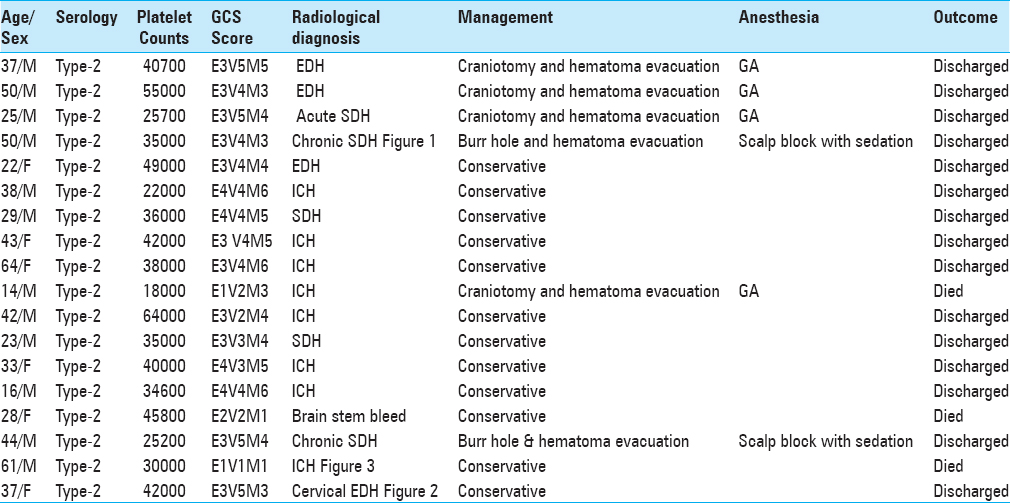
Methods:This retrospective study was conducted in the neurosurgery department of our tertiary healthcare centre. Duration of the study was 1 year, and 18 patients who presented with intracranial bleed and required neurosurgical care were included in this study. All patients had deranged coagulation profile and were thrombocytopenic. All the patients were given platelet concentrates for correction of thrombocytopenia. Eight of these patients had deterioration in their neurological status, and 6 of them underwent surgery.
Results:Out of 18 patients, 12 (66.66%) were managed conservatively, including one case of cervical extradural hemorrhage. Five patients who were conservatively managed died because they had deep-seated bleed and rapid deterioration. The remaining 7 patients who were managed conservatively improved well with few residual deficits. Six (33.33%) patients who underwent surgery had excellent outcome with one case of mortality.
Conclusion:Very high index of suspicion is required in dengue infected patients for neurological complications, especially during the convalescence period. Special attention should be given to those patients who have altered sensorium, and should not be misinterpreted as fever delirium or toxic encephalopathy. It requires immediate attention and further neurological investigation (including thorough clinical examination). Timely diagnosis using a computed tomography scan and early neurosurgical intervention after rapid correction of thrombocytopenia can save many lives.
Keywords: Dengue hemorrhagic fever, extradural hematoma, intracranial bleed, subdural hematoma, thrombocytopenia
INTRODUCTION
Dengue is a mosquito-borne infection transmitted to humans by Aedes aegypti and caused by one of the four closely related virus serotypes of the genus Flavivirus, family Flaviviridae.[
MATERIALS AND METHODS
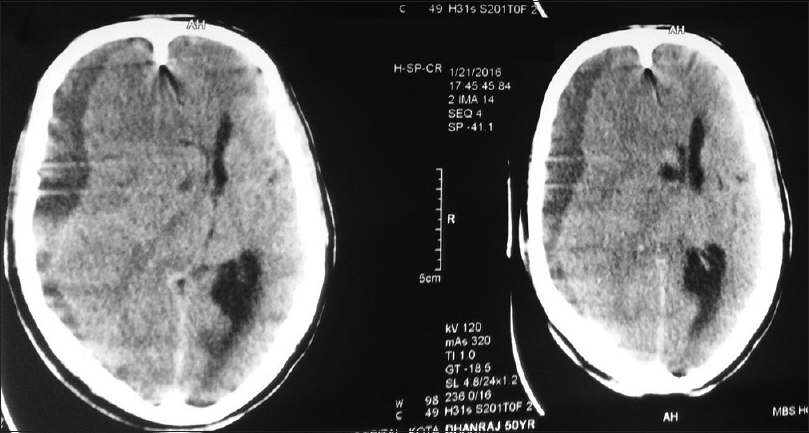
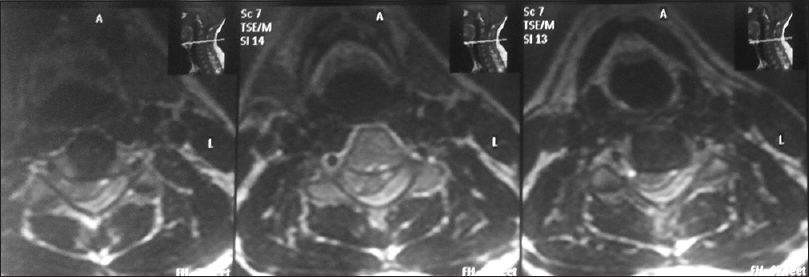
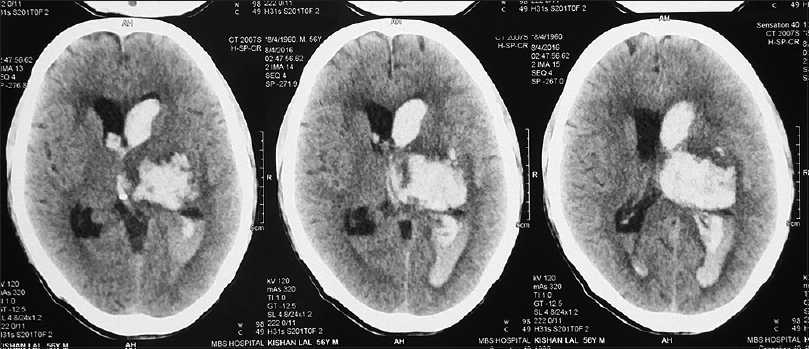
We collected retrospective data from dengue infected patients with neurological manifestations in our tertiary health centre, i.e., Government medical college, Kota (Rajasthan), India from 1st January 2015 to 31st December 2015 [Figures
RESULTS
All of them had typical neurological clinical presentations such as headache, altered consciousness, and limb weakness with deranged coagulation profile and thrombocytopenia. All were confirmed serologically for dengue viral infection. All patients were young adults and had intracranial bleed around convalescence (1 week after the onset of fever). All patients had moderate-to-severe headache before neurological deterioration. Only two of them had a history of nasal bleed and petechiae. All the patients were given platelet concentrate transfusions to keep platelet count above 100000/mL and blood components were made available during the perioperative period. Adequate hydration was maintained according to urine output and clinical assessment perioperatively in all patients, and cerebral decongestants were used according to the need. Scalp block with intravenous dexmedetomidine infusion was used in two patients for chronic subdural hemorrhage surgery while rest of them were managed with balanced general anaesthesia with mechanical ventilation. Short acting anaesthetic drugs were used and general anaesthesia was maintained with oxygen, isoflurane and vecuronium. All operated patients were extubated immediately in the postoperative period and shifted to neurosurgery ICU with stable hemodynamics.
After detailed observation of all these cases, we conclude that Glasgow coma scale (GOS) scores of >9, early admission, early serological and radiological diagnosis, early effective intravenous fluid therapy, anti-edema agents and platelet transfusion, and early neurosurgical interventions in needy patients under balanced anaesthesia are good prognostic indicators. In contrast, deep seated intracerebral massive hemorrhage, delay in effective management, associated severe meningoencephalitis, and old age of patients with associated comorbidities are bad prognostic criteria.
DISCUSSION
It is estimated that every year there are 100 million cases of dengue fever and 2.5 billion people at risk. Dengue is the second most important mosquito-borne disease affecting humans after malaria.[
Dengue is a mosquito-borne infection caused by an arbovirus. This disease is caused by any one of the 4 types of viruses, namely, dengue 1, 2, 3, and 4. People can be infected by one or more types during one episode of illness, but only once by the same type in their entire life time. Dengue 1 causes high fever and joint pains, dengue 2 causes hemorrhagic fever resulting in spontaneous bleeding from skin and gums, dengue 3 causes high fever, whereas dengue 4 causes DHF with shock. Each serotype is different from each other and there is no cross-protection. Epidemics can be caused by multiple serotypes.[
The most critical phase of this viral infection is the defervescent phase when the fever subsides but the patient develops life-threatening thrombocytopenia due to complement activation by viral antigens binding to platelets. The mainstay of treatment is supportive therapy. Platelet transfusion is rarely indicated unless the platelet level drops significantly or if there is significant bleeding. However, the transfusion is recommended on platelet count falling below 20000/ml even without hemorrhage or approximately 50000/ml with hemorrhage. Surgical intervention for ocular and intracranial surgeries can be undertaken after correction of coagulation defect with platelet count above 100000/mL.[
The exact mechanism of bleeding in DHF is not clear, however, it appears to be multifactorial. Thrombocytopenia, prolonged prothrombin time, mild degree of disseminated intravascular coagulation, and hepatic dysfunction all seem to contribute synergistically. Recent data do not advocate a delay in extubating patients when neurological impairment is the only reason for prolonged intubation. An appropriate choice of sedatives and analgesics during mechanical ventilation of neurosurgical patients allows for a narrower range of wake-up time, and weaning protocols incorporating respiratory and neurological measures may improve outcome. Shivbalan et al.[
This is the first large longitudinal study to provide incidence rates for CNS signs during dengue fever among patients seen at a tertiary health care centre in our zone. Overall, the present study suggests that neurological signs of dengue are not exceptional even in patients without the most severe features of dengue. These manifestations were spontaneously resolutive and were associated with patients’ various allergic or hemorrhagic manifestations.
CONCLUSION
GCS scores (>9), early serological and radiological diagnosis, early effective intravenous fluid supportive therapy, anti-edema agents and platelets transfusions, and obviously early neurosurgical interventions in needy patients under balanced anesthesia are good prognostic indicators in such patients. A high degree of suspicion in dengue is required, especially during convalescence in patients who are disoriented and have altered sensorium. It should not be misinterpreted as fever delirium or toxic encephalopathy. It needs immediate attention and CT scan. Timely diagnosis, adequate fluid management, and intervention can save many precious lives.
Disclosure
There is no conflict of interest for this study. Institutional ethical approval was taken for this research.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ali N, Usman M, Syed N, Khurshid M. Hemorrhagic manifestations and utility of haematological parameters in dengue fever. A tertiary care centre experience in Karachi. Scand J Infect Dis. 2007. 39: 1025-8
2. Angibaud G, Luaute J, Laille M. Brain involvement in dengue fever. J Clin Neurosci. 2001. 8: 63-5
3. Attavinijtrakarn P. Hepatic dysfunction in dengue haemorrhagic fever in Paholpolpayuhasaena Hospital. Thai J Pediatr. 2000. 39: 265-76
4. . Centers for Disease Control and Prevention. Imported dengue– Florida, 1997-1998. Can Commun Dis Rep. 2000. 26: 77-9
5. .editorsDengue hemorrhagic fever. Diagnosis, treatment, prevention, and control. Geneva: World Health Organization; 1997. p. 1-84
6. Gupta E, Dar L, Kapoor G, Broor S. The changing epidemiology of dengue in Delhi, India. Virology J. 2006. 3: 92-
7. Halstead SB, Heinz FX, Barrett AD, Roehrig JT. Dengue virus: Molecular basis of cell entry and pathogenesis, 25-27 June 2003, Vienna, Austria. Vaccine. 2005. 23: 849-56
8. Lee SH, Park KJ, Kang SH, Jung YG, Park JY, Park DH. Prognostic Factors of Clinical Outcomes in Patients with Spontaneous Thalamic Hemorrhage. Med Sci Monit. 2015. 21: 2638-46
9. Narang P, Broor S. Co-circulation of dengue serotypes 1, 2, 3 and 4 during the 2003 outbreak in Delhi, India. Emerg Infect Dis. 2006. 12: 352-3
10. Rothman AL, Ennis FA. Immunopathogenesis of dengue hemorrhagic fever. Virology. 1999. 257: 1-6
11. Shivbalan S, Anandnathan K, Balasubramanian S, Datta M, Amalraj E. Predictors of spontaneous bleeding in dengue. Indian J Pediatr. 2004. 71: 33-6
12. Shu PY, Huang JH. Current advances in dengue diagnosis. Clin Diagn Lab Immunol. 2004. 11: 642-50
13. Solomon T, Dung NM, Vaughn DW. Neurological manifestations of dengue infection. Lancet. 2000. 355: 1053-9
14. Tripathi BK, Gupta B, Sinha RS, Prasad S, Sharma DK. Experience in adult population in dengue outbreak in Delhi. J Assoc Physicians India. 1999. 47: 653-4
15. Whitehead SS, Blaney JE, Durbin AP, Murphy BR. Prospects for a dengue virus vaccine. Nat Rev Microbiol. 2007. 5: 518-28