- Department of Orthopaedic Surgery, Cooper Medical School of Rowan University, Camden, New Jersey, United States
- Department of Plastic Surgery, Cooper Medical School of Rowan University, Camden, New Jersey, United States
- Department of Biomedical Engineering, Rowan University, Glassboro, New Jersey, United States
- Department of Physical Medicine and Rehabilitation, Cooper Medical School of Rowan University, Camden, New Jersey, United States.
Correspondence Address:
David A. Fuller, Department of Orthopaedic Surgery, Cooper Medical School of Rowan University, Camden, New Jersey, United States.
DOI:10.25259/SNI_901_2023
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Andrew Simon1, Grace Victoria Gilbert1, Alec Hayes Fisher2, Parker Hunt Johnsen1, Brandon Herb3, Sebastián L. Vega1,3, Elliot Bodofsky4, David A. Fuller1. A comparison of two versus five epineural sutures to achieve successful polyethylene glycol (PEG) nerve fusion in a rat sciatic nerve repair model. 03-May-2024;15:152
How to cite this URL: Andrew Simon1, Grace Victoria Gilbert1, Alec Hayes Fisher2, Parker Hunt Johnsen1, Brandon Herb3, Sebastián L. Vega1,3, Elliot Bodofsky4, David A. Fuller1. A comparison of two versus five epineural sutures to achieve successful polyethylene glycol (PEG) nerve fusion in a rat sciatic nerve repair model. 03-May-2024;15:152. Available from: https://surgicalneurologyint.com/surgicalint-articles/12878/
Abstract
Background: We compared rates of successful polyethylene glycol (PEG) nerve fusion between two epineural suture repairs (2SR) and five epineural suture repairs (5SR) in a rat sciatic nerve transection neurorrhaphy model. We hypothesise that the two and five epineural neural suture repair groups will achieve a similar rate of PEG fusion.
Methods: Twenty-five Lewis rats underwent bilateral sciatic nerve transection. Primary neurorrhaphy (PN) consisting of 2SR in one hind limb and 5SR in the contralateral hind limb was performed utilizing PEG fusion. Successful PEG fusion was confirmed by a distal muscle twitch after nerve stimulation proximal to the nerve fusion site. Sciatic nerve conduction velocity (SNCV) across the repair site and the force generated by tibialis anterior muscle (TAM) contraction were also compared between the 2SR and 5SR groups.
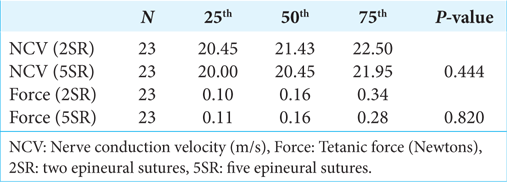
Results: Success rates were 100% for the 2SR and the 5SR groups. No statistically significant differences in SNCV (P = 0.444) or isometric tetanic TAM contractile force (P = 0.820) were observed between 2SR and 5SR in the setting of PEG fusion.
Conclusion: These findings demonstrate no significant difference in successful PEG fusion between the 2SR and 5SR groups. In addition, the findings demonstrate no statistically significant differences in SNCV or isometric tetanic TAM contractile force following sciatic nerve transection when performing a 2SR or 5SR PN in the setting of PEG fusion. Successful PEG fusion can be achieved acutely with either a two or five-epineural suture repair in a rat model.
Keywords: Electrophysiology, Epineural suture, Polyethylene glycol, Rat models
INTRODUCTION
Peripheral nerve injuries (PNIs) most commonly arise in the setting of trauma and have the potential to result in functionally devastating long-term limitations. Despite advances in primary repair techniques, only 51.6% of patients report satisfactory motor outcomes, with even fewer patients (42.6%) reporting acceptable sensory outcomes.[
PEG fusion can be achieved following a 3-step process at the time of primary neurorrhaphy, which includes (1) lavage of nerve endings with calcium-free solution, (2) an antioxidant application, and (3) a PEG application associated with suture neurorhaphy. PEG fusion is not assured even following this process. Typically, intraoperative nerve stimulation with a physiologic stimulus proximal to the nerve fusion is performed by observing for distal muscle twitch to confirm successful fusion. The optimal number of sutures for nerve coaptation in the setting of PEG fusion has not yet been clearly elucidated despite extensive investigation pertaining to nonPEG peripheral nerve repair techniques.[
This study aims to compare the success rate between the 2SR and the 5SR in a rat model of traumatic sciatic nerve transection in the setting of PEG fusion. Successful PEG fusion is confirmed with intraoperative physiologic nerve stimulation above the PN site with confirmed muscle twitch distal to the repair. For the repairs in which successful PEG fusion is confirmed, electrophysiologic data are recorded to discern immediate nerve and muscle function. The immediate sciatic nerve conduction velocity (SNCV) and isometric tetanic tibialis anterior muscle (TAM) contractile force are compared between the 2SR and the 5SR groups after the PEG fusions. Understanding the success rates and the immediate intraoperative functional outcomes based on the number of sutures used may provide better guidance for PN in the setting of PEG fusion following PNI.
MATERIALS AND METHODS
Surgical technique
Subjects consisted of 25 male Lewis rats, all of whom were retired breeders. Aseptic surgery was performed on three separate calendar days using standard microsurgical techniques under loupe magnification and microscope magnification (Leica WILD M690). Alternating sides and groups (2 or 5 sutures), the sciatic nerve was exposed through a 3 cm posterolateral longitudinal straight skin incision from the hip to the knee joint. The trifurcation of the sciatic nerve into the tibial, peroneal, and sural branches was exposed through the interval between the gluteus superficialis and biceps femoris muscles. Baseline nerve conduction velocities were measured for all nerves before transection. A transverse nerve laceration was then created 1 cm proximal to the sciatic trifurcation above the popliteal fossa using microsurgical scissors. Microsurgical suture material used with 10-0 nylon (Ethicon, Somerville, NJ). As a first step in the PN, a single epineural suture was initially placed to create a single point of epineural nerve coaptation. This helped prevent the nerve endings from retracting and kept the ends in proximity for the application of the first two solutions as part of the PEG repair. The exposed nerve endings within the surgical field were first irrigated with 0.5 mL of calcium-free solution (Plasma-Lyte A, Baxter Healthcare Corporation, Deerfield, IL), followed by dripping methylene blue antioxidant solution (Spectrum Chemical Manufacturing Corporation, Gardena, CA) until both nerve ends were impregnated with the second solution.
After the application of these first two solutions, the final epineural sutures were inserted. To complete the 2-suture (2SR), a second suture was placed 180° from the initial suture into the epineurium. To complete the 5-suture (5SR), four additional equally spaced sutures were placed around the initial suture into the epineurium. Immediately following both 2SR and 5SR, the coaptation was treated with 0.5 mL PEG solution comprising 50% w/w 3.35 kDa PEG (Sigma Life Science, St. Louis, MO) in deionized distilled water. The PEG solution bathed the PN for 90 seconds to facilitate axolemma fusion. The PEG solution was then washed off with Lactated Ringer’s solution (B. Braun Medical, Bethlehem, PA). PEG fusion was confirmed by stimulating the sciatic nerve above the PN site with a physiologic impulse (0.1 mA) and observing a contraction of the TAM. Larger, supra-physiologic impulses (1.0 mA and higher) were found to stimulate muscle contraction even without PEG fusion after PN, indicating that larger stimulation can override a PN without PEG application. Such high stimulation can produce false positives wherein a muscle contraction can occur due to the high stimulation’s ability to override the PN site even without PEG application.
The suture repairs were performed by surgical residents and faculty experienced in PEG neurorrhaphy and microsurgical techniques. Our method to confirm a successful PEG fusion has been to stimulate the nerve proximal to the PEG fusion with a physiologic stimulation (0.1 mA) and observe for muscle activation distally (ankle plantar or dorsiflexion). 0.1 mA was chosen based on work with this model studying both PEG and nonPEG neurorrhaphy. In confirming our experimental setup, we found that higher nerve stimulation can cause muscle activation distally, even across a suture neurorrhaphy without a PEG fusion. We observed that 0.3 mA was a threshold for us above which a signal could potentially transmit even across standard suture neurorrhaphy. The surgical technique in our laboratory has been previously described and published.[
Statistical analysis was performed with the assistance of a biostatistician. The data were assessed for its normality of distribution. A Wilcoxon signed-rank test was used based on distribution.
This study was conducted with the approval and supervision of our Institutional Animal Care and Use Committee (IACUC). Because it was an animal study and no humans were involved, the IACUC was the necessary approval panel. All institutional and national guidelines for the care and use of laboratory animals were followed. An Institutional Review Board approval was not necessary as no humans were involved in the research.
Electrophysiology: Nerve conduction velocity (NCV)
Electrophysiologic testing under anesthesia measuring SNCV was performed through a commercially available nerve conduction study device (NeuroMax 1002, Excel Tech LTD). A single pulse of 0.1 mA was applied to the sciatic nerve for a duration of 0.05 ms to induce nerve conduction. These measurements were obtained pre and postsciatic nerve transection, following coaptation of nerve segments, and on PEG fusion. The stimulating and recording electrodes were bonded with epoxy at a fixed distance (14 mm) apart to try to reduce variability.
Electrophysiology: Tetanic force generation
After suture repair and successful PEG fusion, the TAM tendon was first released from its insertion in the foot and freed from the surrounding tissue while preserving its neurovascular pedicle. The tibia of the hind limb was secured to a testing block with K-wires (Stryker Orthopaedics, Mahwah, NJ). The distal tibialis anterior tendon was secured with a custom clamp fashioned from a modified surgical hemostat attached to a force transducer (MDB-2.5, Transducer Techniques, Temecula, CA). The force transducer signal provided a digital readout of both active force and peak force produced. The clamp was placed such that the TAM was near its anatomical length and consistent preload. A bipolar stimulator and micro-hook electrode were used to deliver the 0.1 mA electrical impulse at 5-s intervals. Peak isometric tetanic TAM contractile force was recorded over five impulses, both proximally and distally, to the nerve repair. The greatest force measured over these five impulses was recorded.
RESULTS
Successful PEG for fusion 2SR and 5SR
We had two intraoperative deaths due to anesthetic complications, such that 23 of the 25 animals were able to successfully complete the study, resulting in 23 2SR and 23 5SR PN with PEG fusion for comparison. All 23 nerves in both the 2SR and 5SR groups achieved successful PEG fusion based on observed downstream muscle contracture at 0.1 mA stimulation above the PN. No PEG neurorrhaphy had to be redone. The 100% success rate (23 successes out of 23 attempts) for both the 2SR and the 5SR obviates the need for any statistical analysis to conclude no difference.
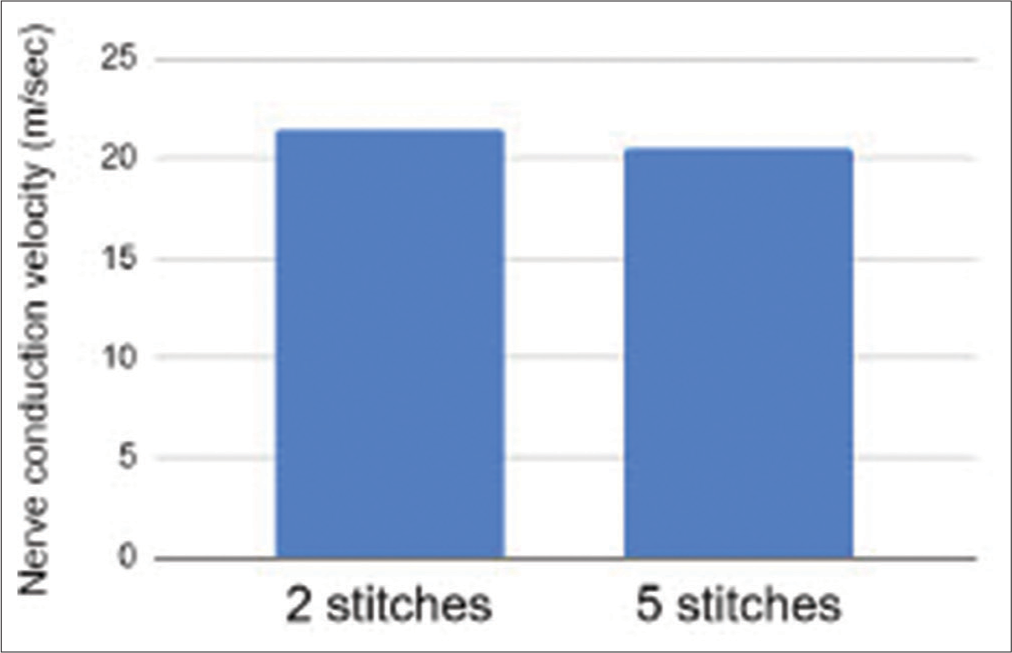
NCV between 2SR and 5SR
No difference is observed in the NCV between the 2SR and the 5SR groups [
As a positive control, the NCV was measured before transection in both experimental groups, with an average of 23.31 m/s. We expected that the pre transection NCV would be greater than the postPEG fusion NCV in both the 2SR and 5SR groups, and, indeed, this was the case.
Tetanic force between 2SR and 5SR
No difference was observed in the tetanic force between the 2SR and the 5SR groups [
As a positive control, the tetanic force was measured by stimulating distal to the repair site for both experimental groups. The average positive control was 0.42 Newtons for the two groups.
DISCUSSION
The primary purpose of our study was to determine whether the number of sutures applied to the epineurium influenced PEG fusion success rates. Since we achieved 100% successful fusion in both groups, we feel confident that successful PEG fusion can be achieved immediately with either fewer or greater number of sutures (2 vs. 5). What we did not investigate in this study were the long-term functional implications of more or less sutures in the epineurium which would require survival surgery and additional work. Given that PEG fusion is known to facilitate immediate nerve function, we investigated immediate nerve and muscle activity as a surrogate for functional outcomes. The data conclusively demonstrate minimal differences in SNCV and isometric tetanic TAM contractile force following sciatic nerve transection and PEG fusion, regardless of whether the surgeon implements two sutures or five sutures for the repair. How this translates into longer-term functional outcomes remains an open question and may be a source for additional investigation.
In our present study, although no immediate significant electrophysiological functional differences were shown between two and five sutures, other factors related to functional nerve outcome may need to be considered. There remain practical benefits to utilizing fewer sutures, including lower cost, easier procedure, less trauma to the nerve, and finally, less foreign material. Excess sutures could theoretically induce increased inflammation and scar tissue, which could challenge the long-term outcomes. Beyond successful PEG fusion considerations, a repair also needs to provide adequate tensile strength to protect the repair and simultaneously promote optimal nerve recovery over time. We already have an understanding in epineural neurorrhaphy that increasing suture numbers at the coaptation site increases load to failure. Goldberg et al. used cadavers to show that both the increased number and caliper of suture increased maximum failure load. In addition, independent caliper size and the number of sutures significantly increased the max failure load in all groups.[
Our primary research intent was to aid the peripheral nerve surgeon with an intraoperative procedural decision related to how the number of sutures applied may promote successful PEG fusion. Intraoperative muscle contracture is not always possible to achieve downstream from a PN, particularly when repairing a sensory nerve. This data supports the use of either a 2SR or a 5SR as a consideration for successful PEG fusion. Based on the results of this in vivo study, surgeons may consider other factors beyond these immediate electrophysiological outcome measures when choosing between a looser or tighter epineural neurorrhaphy augmented with PEG fusion. Our recommendation based on this work and other considerations remains to utilize fewer rather than greater numbers of epineural sutures to approximate the nerve endings when performing PN with or without PEG fusion. Because our primary interest was in immediate nerve function after PEG fusion, we did not perform any survival surgeries. Such future studies may yield relevant outcome measures beyond the data that we present in this study.
This study was limited by several factors. Primarily, due to the study design and immediate euthanasia of the subjects following transection and repair, no long-term functional outcomes could be measured nor survival data generated. We intended to study successful immediate PEG fusion as part of understanding better mechanisms to achieve better longitudinal functional outcomes. Without immediate successful PEG fusion confirmation, longer-term gains would not be possible. As such, future studies may wish to investigate whether subjects eventually approach baseline functioning postoperatively, as well as the timeline required to do so. It remains possible that increasing the number of subjects studied could have potentially yielded differences in our successful PEG fusion rate or our immediate electrophysiology data. Based on our intended aims, our statistical and power analyses, and stewardship of our resources, we believe that this series reveals that immediate PEG fusion can be achieved predictably with either a 2SR or a 5 SR.
CONCLUSION
A successful PEG fusion can be achieved similarly with either two or five epineural sutures in a rat sciatic neurorrhaphy model.
Ethical approval
An Institutional Animal Care and Use Committee Approval (IACUC #2020-004) was obtained for this project on 4/20/2020.
Declaration of patient consent
Patient’s consent was not required as there are no patients in this study.
Financial support and sponsorship
Rowan University Camden Health Research Initiative Grant.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Babovic N, Klaus D, Schessler MJ, Schimoler PJ, Kharlamov A, Miller MC. Assessment of conduit-assisted primary nerve repair strength with varying suture size, number, and location. Hand (N Y). 2019. 14: 735-40
2. Fisher AH, Johnsen PH, Simon A, Burns CJ, Romiyo V, Bodofsky EB. Fibrin glue acutely blocks distal muscle contraction after confirmed polyethylene glycol nerve fusion: An animal study. Plast Reconstr Surg Glob Open. 2024. 12: e5535
3. Ghergherehchi CL, Mikesh M, Sengelaub DR, Jackson DM, Smith T, Nguyen J. Polyethylene glycol (PEG) and other bioactive solutions with neurorrhaphy for rapid and dramatic repair of peripheral nerve lesions by PEG-fusion. J Neurosci Methods. 2019. 314: 1-12
4. Goldberg SH, Jobin CM, Hayes AG, Gardner T, Rosenwasser MP, Strauch RJ. Biomechanics and histology of intact and repaired digital nerves: An in vitro study. J Hand Surg Am. 2007. 32: 474-82
5. Gordon T. Peripheral nerve regeneration and muscle reinnervation. Int J Mol Sci. 2020. 21: 8652
6. Martins RS, Siqueira MG, Da Silva CF, Plese JP. Overall assessment of regeneration in peripheral nerve lesion repair using fibrin glue, suture, or a combination of the 2 techniques in a rat model. Which is the ideal choice? Surg Neurol. 2005. 64: 10-6
7. Paprottka FJ, Wolf P, Harder Y, Kern Y, Paprottka PM, Machens HG. Sensory recovery outcome after digital nerve repair in relation to different reconstructive techniques: Meta-analysis and systematic review. Plast Surg Int. 2013. 2013: 704589
8. Riley DC, Bittner GD, Mikesh M, Cardwell NL, Pollins AC, Ghergherehchi CL. Polyethylene glycol-fused allografts produce rapid behavioral recovery after ablation of sciatic nerve segments. J Neurosci Res. 2015. 93: 572-83
9. Ruijs AC, Jaquet JB, Kalmijn S, Giele H, Hovius SE. Median and ulnar nerve injuries: A meta-analysis of predictors of motor and sensory recovery after modern microsurgical nerve repair. Plast Reconstr Surg. 2005. 116: 484-94 discussion 495-6
10. Tang P, Schimoler P, Kim H, Gillman B, Kharlamov A, Miller M. The optimal number and location of sutures in conduit-assisted primary digital nerve repair. J Hand Surg Eur Vol. 2018. 43: 621-5