- Department of Neuroendovascular Therapy, Instituto Nacional de Neurologia y Neurocirugia, Manuel Velasco Suarez, Mexico city, Mexico
- Department of Clinical Research, Instituto Nacional de Neurologia y Neurocirugia, Manuel Velasco Suarez, Mexico city, Mexico
- Institute of Clinical Neuroscience and Medical Psychology, University Hospital Düsseldorf, Germany
Correspondence Address:
Felipe Padilla-Vazquez
Institute of Clinical Neuroscience and Medical Psychology, University Hospital Düsseldorf, Germany
DOI:10.4103/sni.sni_273_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Felipe Padilla-Vazquez, Marco A. Zenteno, Jorge Balderrama, Victor Hugo Escobar-de la Garma, Daniel San Juan, Carlos Trenado. A proposed classification for assessing rupture risk in patients with intracranial arteriovenous malformations. 27-Dec-2017;8:303
How to cite this URL: Felipe Padilla-Vazquez, Marco A. Zenteno, Jorge Balderrama, Victor Hugo Escobar-de la Garma, Daniel San Juan, Carlos Trenado. A proposed classification for assessing rupture risk in patients with intracranial arteriovenous malformations. 27-Dec-2017;8:303. Available from: http://surgicalneurologyint.com/surgicalint-articles/a-proposed-classification-for-assessing-rupture-risk-in-patients-with-intracranial-arteriovenous-malformations/
Abstract
Background:Whether cerebral arteriovenous malformations (AVMs) should be treated remains an ongoing debate. Nevertheless, there is a need for predictive factors that assist in labelling lesions as low or high risk for future rupture. Our aim was to design a new classification that would consider hemodynamic and anatomic factors in the rapid assessment of rupture risk in patients with AVMs.
Methods:This was a retrospective study that included 639 patients with ruptured and unruptured AVMs. We proposed a new classification score (1–4 points) for AVM rupture risk using three factors: feeding artery mean velocity (Vm), nidus size, and type of venous drainage. We employed descriptive statistics and logistic regression analysis.
Results:A total of 639 patients with cerebral AVMs, 388 (60%) had unruptured AVMs and 251 (40%) had ruptured AVMs. Logistic regression analysis revealed a significant effect of Vm, nidus size, and venous drainage type in accounting for the variability of rupture odds (P = 0.0001, R2 = 0.437) for patients with AVMs. Based in the odds ratios, grades 1 and 2 of the proposed classification were corresponded to low risk of hemorrhage, while grades 3 and 4 were associated with hemorrhage: 1 point OR = (0.107 95% CI; 0.061–0.188), 2 point OR = (0.227 95% CI; 0.153–0.338), 3 point OR = (3.292 95% CI; 2.325–4.661), and 4 point OR = (23.304 95% CI; 11.077–49.027).
Conclusion:This classification is useful and easy to use, and it may allow for the individualisation of each cerebral AVM and the assessment of rupture risk based on a model of categorisation.
Keywords: Cerebral arteriovenous malformations, cerebral hemodynamics, Doppler ultrasound, cerebral venous drainage
INTRODUCTION
The Spetzler and Martin (SM) classification of arteriovenous malformations (AVMs), published in 1986,[
The aim of this study is to propose a classification system that allows for an estimation of bleeding risk in patients with cerebral AVMs. This proposed classification may help to differentiate between patients at a high risk of bleeding in need of aggressive management and patients with a low risk of bleeding who would benefit more from conservative treatment.
MATERIALS AND METHODS
This was a retrospective study that analysed 639 patients diagnosed with cerebral AVMs, from 1 March 2006 to 31 October 2015 at the Department of Neuroendovascular Therapy at the Instituto Nacional de Neurologia y Neurocirugía Manuel Velasco Suárez in Mexico City. The scientific and ethics committees of our institution approved the conduct of this research. We adhered to the Declaration of Helsinki.
The inclusion criteria were as follows: any patient with a diagnosis of cerebral AVM of any grade according to the Spetzler Martin classification, of any sex or age, a complete medical record and an imaging workup including at least a diagnostic cerebral angiogram and a transcranial Doppler ultrasound (TCD). All patients included underwent a head computed tomography scan, a brain magnetic resonance imaging or both. However, these imaging modalities were not used in the classification.
We included patients with ruptured and unruptured AVMs. The diagnosis of ruptured AVMs was confirmed by a head computed tomography scan performed 1 to 24 h after the onset of clinical symptoms in the patient. In these cases, the cerebral angiograms used for this classification were chosen 2 months after bleeding.
Transcranial Doppler ultrasound
TCD was performed by a single operator (F.P.V.) to measure the state of cerebral circulation in real time, using a Siemens Acuson Antares, premium edition, B©. All patients were measured in both anterior cerebral arteries (A1), the middle cerebral artery (M1), and the posterior cerebral artery (P1). Regardless of their afferences, the major arterial trunks (bilateral A1, M1 and P1) were measured with TCD.
All TCD parameters were evaluated in a previous study, where we measured the Vm, resistance index, pulsatility index, and Reynold's number to determine the degree of hemodynamic characteristics of the AVMs in normotensive patients. In this study, we observed that the hemodynamic parameters can be summarized in the value of the Vm. In this classification, we included all the main feeding artery Vm. Vm was calculated from the systolic velocity plus two diastolic velocities divided by 3 [
Digital subtraction angiography
This procedure was performed on an Artis zee/zeego robotic arm angiography system and VB21C Syngo Workplace postprocessing stations (Siemens Erlangen, Germany©). The size of the malformation nidus was determined by employing DSA in the measurement of the largest diameter of the AVM nidus and the patterns of venous drainage from the AVM (described below).
Description of the proposed AVM classification system
Three independent variables of all AVMs were analysed: afferent pedicles (measured by mean velocities), nidus size, and efferent vessels (venous drainage). Initially we analyzed the odds ratios of AVM bleeding from all the range of Vm (each analysis was stratified in 10cm/s, e.g. 10–19 cm/s) afferent pedicles and nidus size (each cm) to obtain the highest odds ratios related to ruptured AVMs. Then we chose the dichotomous (Vm [> or <90 cm/s] and nidus size [> or <3 cm]) or categorical variables (venous drainage type) to be included in the regression model. Venous drainage was categorised according to the venous patterns found in the cerebral angiograms.
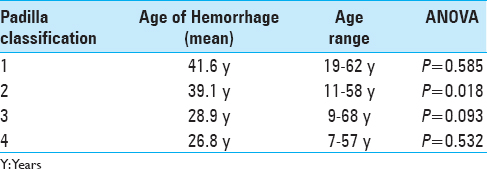
The cut-off mean velocities of the main feeding artery were obtained from a previous analysis conducted by our group in which we estimated the odds ratio of bleeding in patients with different categorical Vm values. According to these results, a Vm <90 cm/s is a risk factor for hemorrhage (OR = 6.5, 95% CI = 1.6, 26.6) compared to a Vm ≥90 cm/s (OR = 0.51, 95% CI = 0.340, 0.792) in the principal feeder of the AVM.
The second variable was the malformation nidus size; a nidus <3 cm (OR = 4.15, 95% CI = 1.29–13.3) had a risk 4-fold higher than a nidus ≥3 cm (OR = 0.55, 95% CI = 0.34, 0.89).
The third variable was venous drainage, which behaves differently in each AVM. Based on visual angiographic cerebral AVMs in the present study (cf. infra), we classified the AVMs according to venous drainage behaviour, dividing them into three types with several subtypes.
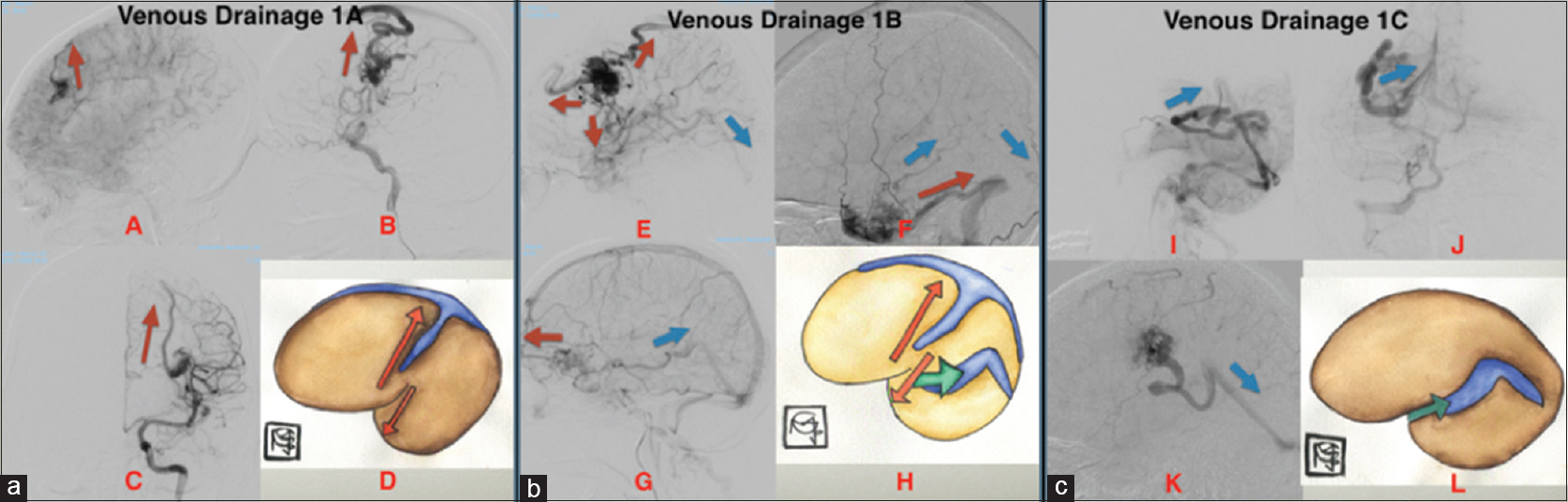
*Type 1 venous drainage: Anterograde (downstream or normal flow)
Anterograde venous drainage was considered normal venous drainage, where the region above the superficial sylvian vein drains (either by cortical veins or the vein of Trolard) to the superior sagittal sinus; below the superficial sylvian vein, the flow is directed toward the vein of Labbé, draining into the transverse sinus.
1A) Superficial venous drainage
1B) Superficial and deep venous drainage
1C) Deep venous drainage
[
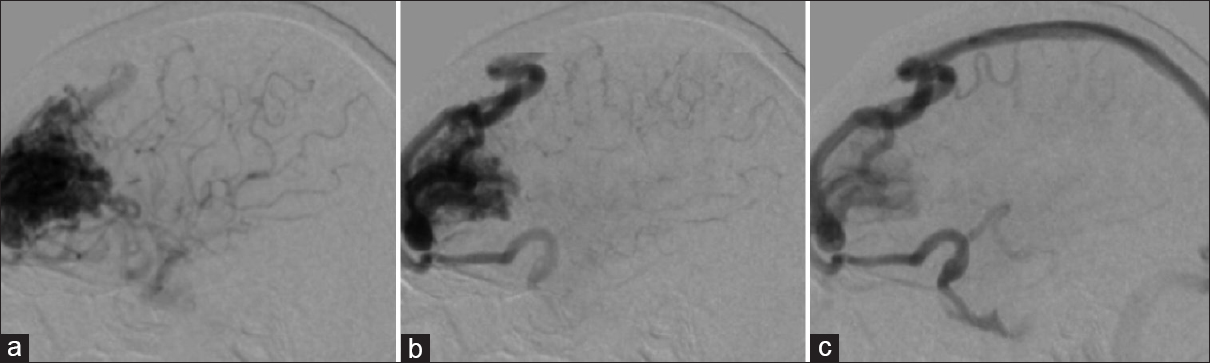
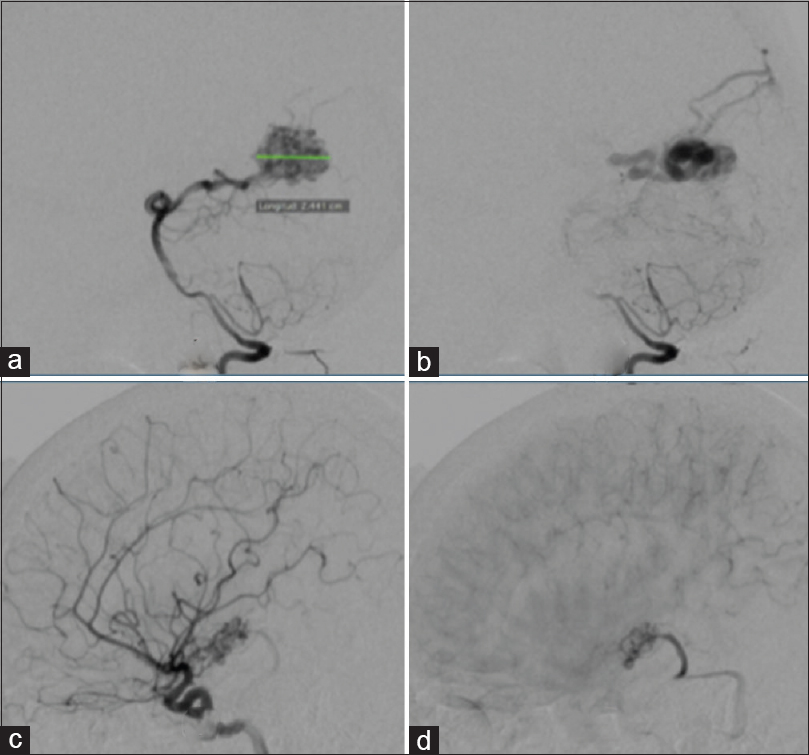
Figure 1
(a) Lateral (A, B) and anteroposterior (AP) (C) cerebral angiogram shows anterograde superficial venous drainage, (downstream): represented in the illustrative image (D). (b)Lateral cerebral angiogram (E, F, G) show mixed venous Drainage (superficial and deep), anterograde in which the superficial drainage is represent with red arrows and deep venous drainage with blue arrows in the illustrative image (H). (c) AP (J) and lateral (I, K) cerebral angiogram shows deep venous drainage anterograde (Type 1C), which is shown with blue arrow in the illustrative image (L)
In this type, we consider the Sylvian fissure to be a natural barrier. Lesions tend to drain away from the superficial sylvian vein, either from the superficial sylvian vein to the superior sagittal sinus or from the superficial sylvian vein to Labbé and the transverse sinus.
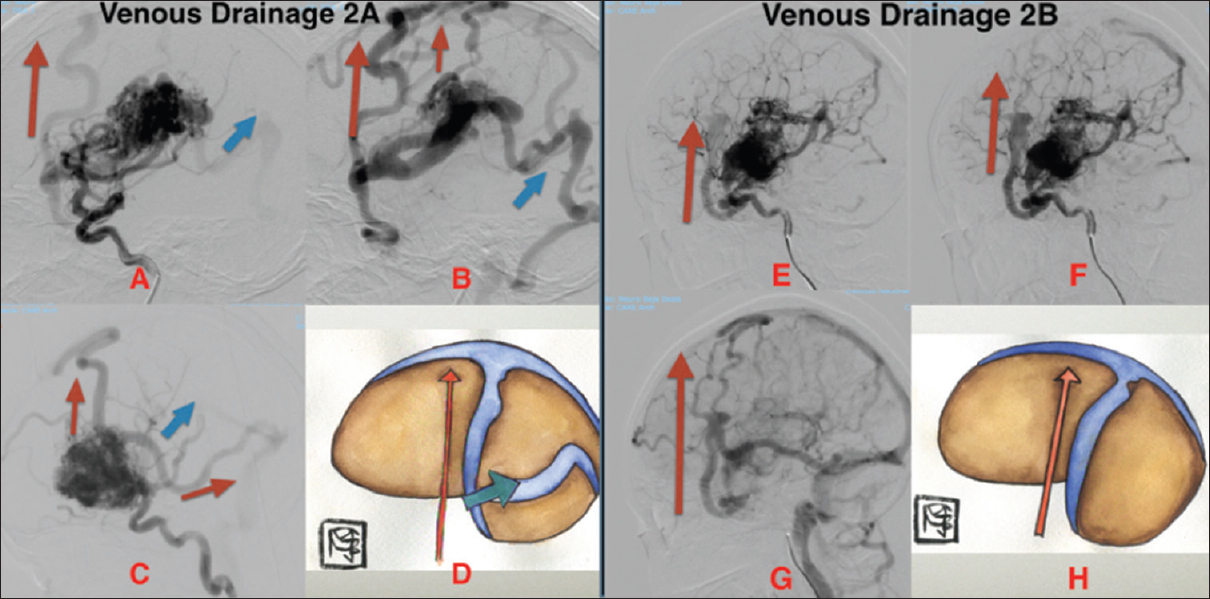
*Type 2 venous drainage: Retrograde (upstream or reverse flow)
Upstream drainage from the skull base to the superior sagittal sinus and/or downstream venous drainage to the contralateral hemisphere from the superior sagittal sinus toward the basal portion.
2A) Superficial and deep venous drainage
2B) Superficial venous drainage
[
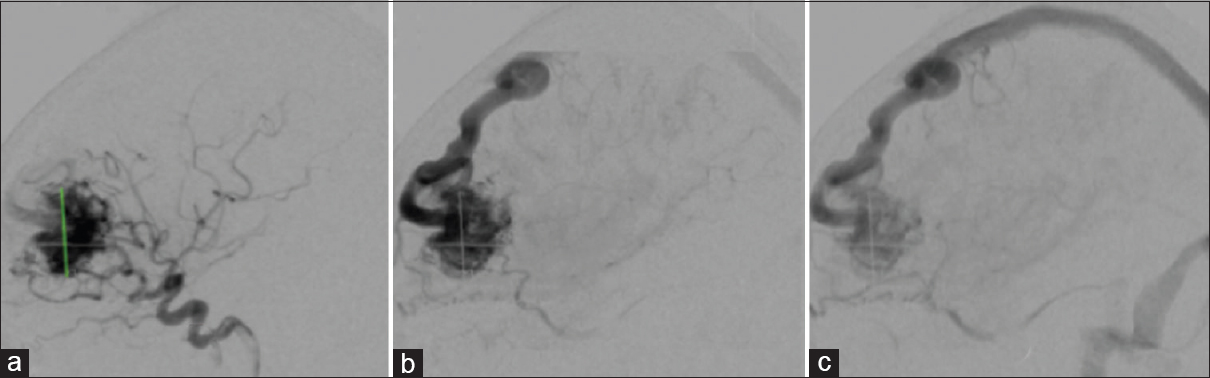
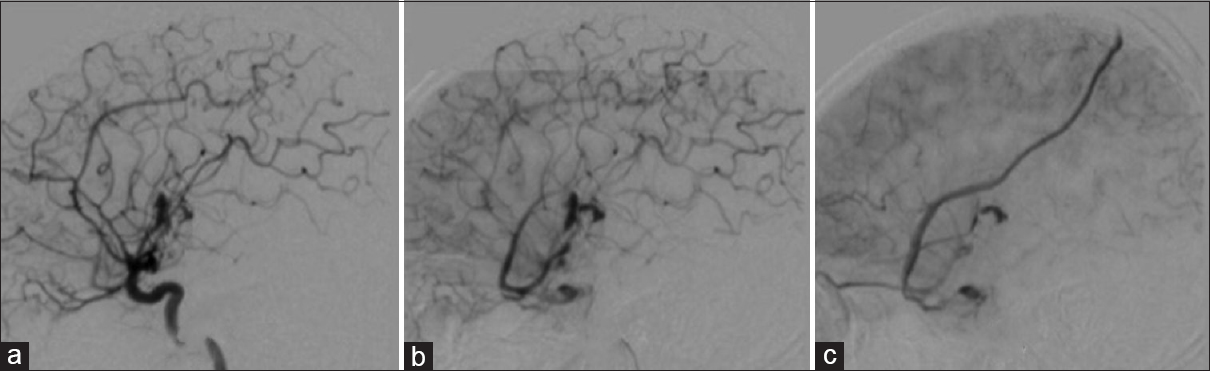
Figure 2
Type 2A: Lateral cerebral angiogram (A, B, C) shows mixed venous drainage (superficial and deep), retrograde (upstream). Superficial venous drainage (red arrow) and deep venous drainage (blue arrow) in (D). Type 2B: Lateral cerebral angiogram (E, F, G) shows superficial retrograde venous drainage (upstream), illustrated in (H)
In this type, the natural barriers are crossed anti-physiologically; basal lesions drain dorsally, crossing the Sylvian fissure, or hemispherical lesions cross the midline to be drained contralaterally, or convexity lesions drain ventrally, crossing the Sylvian fissure to the skull base.
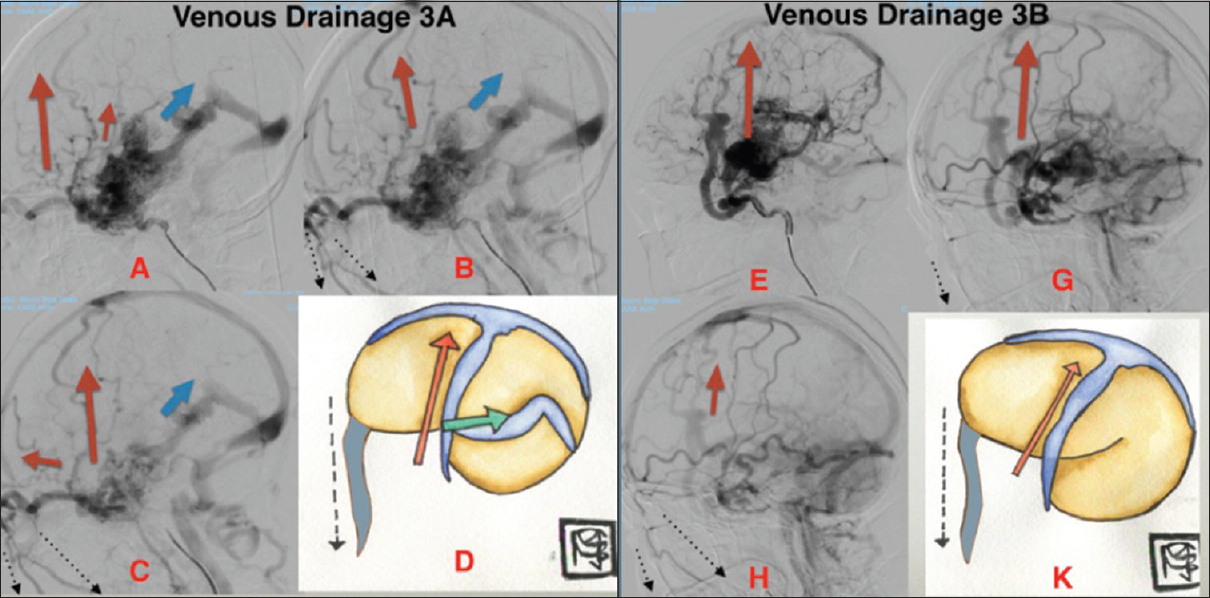
*Type 3 venous drainage: Retrograde (upstream or reverse flow) + facial venous drainage
Upstream drainage from the skull base to the Superior sagittal sinus and/or drainage in the downstream contralateral hemisphere from the superior sagittal sinus toward the basal portion associated with facial vein drainage.
3A) Superficial and deep venous drainage
3B) Deep venous drainage
[
Figure 3
3A: Lateral cerebral angiogram (LCA) (A, B, C) shows mixed venous drainage (superficial and deep), retrograde, (upstream), associated with drain through the facial vein. In (D) superficial venous drainage (red arrow), deep venous drainage (blue arrow) and drainage through the facial vein (dotted line) (D). 3B: LCA (E, G, H) shows superficial retrograde venous drainage (upstream), associated with drainage through the facial vein. Drawing shown in (K, red arrow) represents the retrograde superficial venous drainage and (dotted line) represents the drainage through the facial vein (K)
Determination of the grade of cerebral AVM
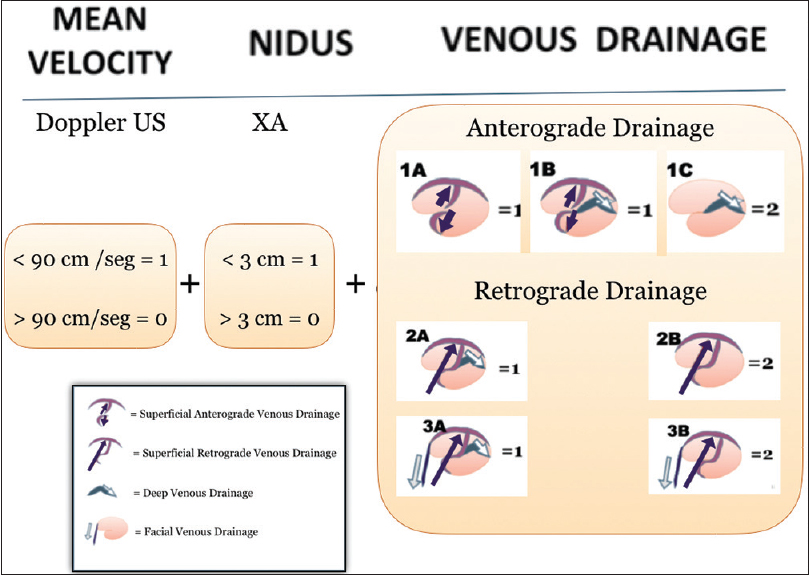
The proposed classification is calculated from the sum of the points from the three variables; Vm, nidus size, and venous drainage type. Thus, it gives a categorical scale [
Figure 4
Diagram showing the Padilla classification, which takes in account the three main components of an AVM (afference, is scored 0 or 1, according to mean velocity as measured by TCD; Nidus is given 0 or 1, according to size in cm; Efference is graded 1 or 2, following the topography and type of circulation as seen in DSA)
Statistical analysis
Categorical variables are presented as frequencies and percentages, and continuous variables are presented as means and ranges. Initially, we performed the Kolmogorov–Smirnov test for each variable analysed in order to evaluate normality. To define the categories of the afferent pedicles and the nidus size, we calculated the odds ratios (Pearson test) of AVM bleeding in 55 patients and then we established the dichotomous variables of these two parameters.
The significance of baseline differences was determined by the Chi-square test, Fisher's exact test or the Mann–Whitney U test, as appropriate. A two-sided P value of less than 0.05 was considered to indicate statistical significance.
We used a logistic regression model to estimate the extent and significance to which risk of bleeding of cerebral AVMs is explained by the proposed independent variables (afferent pedicles [measured by mean velocities], nidus size, and efferent vessels [venous drainage]). It is important to clarify that the proposed regression model is phenomenological in the sense that selection of some variables is not directly derived from theory but rather experimental and empirical observations. The regression model considered as dependent variable presence or absence of ruptured AVM and as independent variables afferent pedicles (measured by mean velocities), nidus size and efferent vessels (venous drainage) reviewed by TCD and digital subtraction angiograms. All the variables were categorical [
RESULTS
A total of 639 patients with cerebral AVMs were included and assessed; 321 (50%) males and 318 (50%) females with total mean age of 32.9 (5–82) years.
Sociodemographic findings
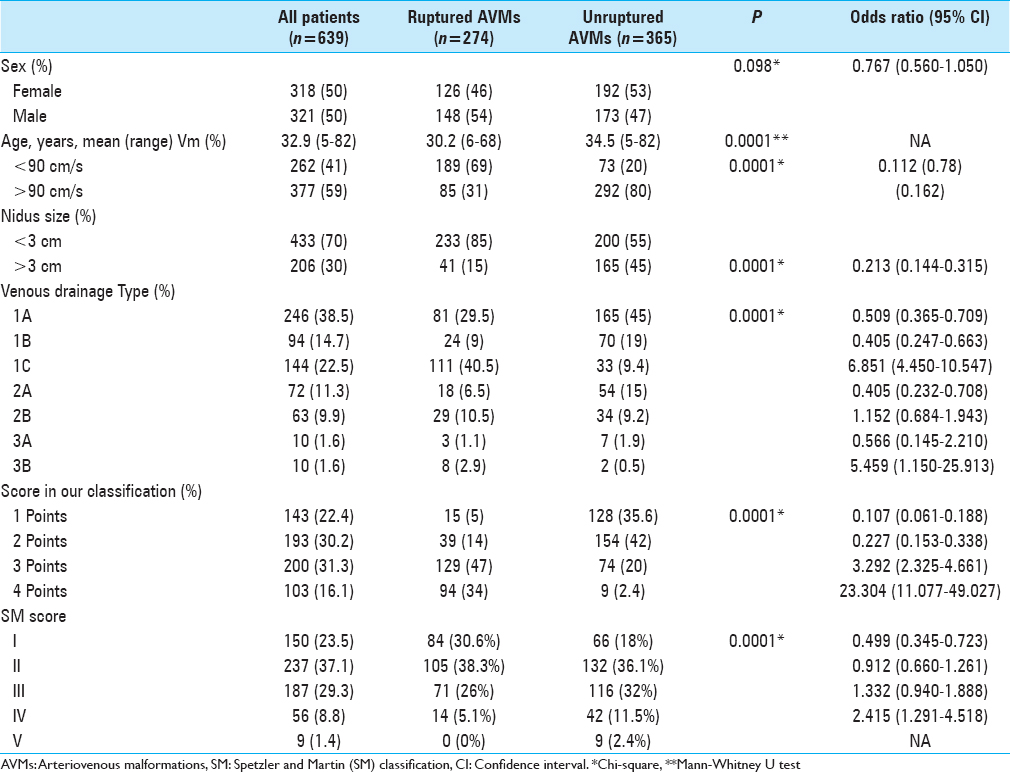
The sociodemographic characteristics, comparisons, and odd ratios of our patients according to the presence or absence of brain hemorrhage due to AVMs are summarised in
Clinical manifestations
In the group of patients with unruptured AVMs (n = 365), the clinical manifestations at the time of diagnosis were as follows: 202/365 (55.3%) seizures, 164/365 (44.9%) headache, 32/365 (8.7%) seizures, and headache, 30/365 (8.2%) steal phenomena (paresis, paraesthesia, transient amaurosis) and 10/365 (2.7%) were asymptomatic.
The group of patients with ruptured AVMs (n = 274) showed the following symptoms before bleeding: 22/274 (8%) headaches, 9/274 (3.2%) seizures, 2/274 (0.7%) steal phenomena, and 244/274 (89%) were asymptomatic.
Figures
Figure 7
Grade 3. Lateral cerebral angiogram (A, B) shows Spetzler Martin grade-II AVM, with deep venous drainage to the vein of Galen and straight sinus and finally the surface drainage anterograde through occipital cortical veins. TCD Mv <90 cm/sec. Grade 3. Lateral cerebral angiogram (C, D) shows temporal arteriovenous malformation Spetzler-Martin grade I, with anterograde superficial drainage through superficial temporal vein to the sigmoid sinus. TCD Mv <90 cm/sec
Logistic regression analysis of bleeding odds in AVMs
On the basis of Chi-square analyses and logistic regression, the influence of sex on bleeding odds was discarded. Nevertheless, Vm, nidus size, and venous drainage type were found to be significant (P = 0.0001). The obtained prediction model of bleeding odds in the context of AVMs is given as: ln (odds) = (-2.248) ·Vm + (-0.878) nidus size + (-0.452) venous drainage. This model allowed for the correct classification of 80.3% of patients without and 77.3% of patients with brain hemorrhage. The logistic regression indicated that anatomic and hemodynamic variables and so the proposed classification explain better the variability of bleeding odds in AVM patients (NR = 621.53, R2 = 0.437) as compared to the SM classification (NR = 847.048, R2 = 0.053).
The distribution of our patients’ AVMs according to the SM classification was: SM-I: 150 (23.4%); SM-II: 237 (37%); SM-III: 187 (29.2%); SM-IV: 56 (9%) and SM-V: 9 (1.4%).
DISCUSSION
The aim in the treatment of an AVM is to prevent future occurrence of intracranial hemorrhage.[
In the last decade, multiple scales for the classification of different aspects of an AVM have appeared. The SM, Spetzler-Ponce and Lawton's supplementary classifications[
Our classification based on scores of 1–4 shows an association between risk of rupture in patients with cerebral AVMs and anatomic and hemodynamic variables, thus it is useful in assessing the odds of rupture in AVM; it is easy to use and for the classification we employed very common diagnostic tests that are available in multiple medical centres around the world. In our opinion, all AVMs should be considered different and unique entities, since each of them may have characteristics that confer an individualised rupture risk thereupon. Our scale may help to establish a hierarchy for every AVM based on angiographic and hemodynamic variables, thus assessing a specific and exclusive bleeding risk and allowing for a more accurate determination of the optimal treatment.
The patients at low risk for rupture in our classification arrived at hospital with headache, seizures, or steal phenomenon symptoms. If these subsets of patients are partially embolised, a low-risk lesion can transform into a high-risk lesion and the patients who arrived by headache end up with cerebral hemorrhage. This phenomenon may occur even in near-totally embolised AVMs.[
With regard to venous drainage, it is well-known that single deep venous drainage is strongly correlated with brain hemorrhage; however, deep venous drainage associated with superficial venous drainage reduces rupture risk.[
Some multi-centre studies claim that patients with cerebral AVMs have a better prognosis when treated with pharmacological therapy, since endovascular and surgical procedures increase morbidity; nevertheless these are based on the SM classification, which we found is not predictive of rupture risk,[
In our study, we observed that a low percentage of patients (19% with a history of hemorrhage and 28% without a history of hemorrhage) exhibit upstream or reverse flow venous drainage. This type of venous drainage is almost always associated with fistulous shunts (low resistance and elevated mean velocity lesions with high turbulence and venous hypertension) and it has a high risk of rupture when it is purely superficial, since the flow and pressure in superficial and deep venous drainage have a better means of escape and dispersion.[
The limitations of our study are those that are common to retrospective studies, the use of selected anatomic and hemodynamic variables of the AVMs, the reproducibility, and consistency of TCD measurements. Notwithstanding the fact that our phenomenological regression modeling approach provided insight into the significance of the proposed variables in explaining rupture risk in patients with AVM, the use of predictive modeling will be necessary in a future study to quantify predictability of the chosen phenomenological variables.
CONCLUSIONS
The present classification is useful and easy to use, and it may allow us to individualise each AVM and assess the risk of rupture of a cerebral AVM based on a model of categorisation.
Conflicts of Interest/Disclosures
The authors declare that they have no financial or other conflicts of interest in relation to this research and its publication.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
The authors wish to thank María Guadalupe Padilla-Vázquez for her outstanding work with the venous drainage images. We also wish to recognise the revision done by our English language editor, Mr Patrick Bennett Weill.
References
1. Bradac O, Charvat F, Benes V. Treatment for brain arteriovenous malformation in the 1998-2011 period and review of the literature. Acta Neurochir. 2013. 155: 199-209
2. Cognard C, Gobin YP, Pierot L, Bailly AL, Houdart E, Casasco A. Cerebral dural arteriovenous fistulas: Clinical and angiographic correlation with a revised classification of venous drainage. Radiology. 1995. 194: 671-80
3. Conger A, Kulwin C, Lawton MT, Cohen-Gadol AA. Diagnosis and evaluation of intracranial arteriovenous malformations. Surg Neurol Int. 2015. 6: 76-
4. Ding D, Starke RM, Kano H, Mathieu D, Huang PP, Feliciano C. International multicentre cohort study of pediatric brain arteriovenous malformations. Part 1: Predictors of hemorrhagic presentation. J Neurosurg Pediatr. 2017. 19: 127-35
5. Dumont TM, Kan P, Snyder KV, Hopkins LN, Siddiqui AH, Levy EI. A proposed grading system for endovascular treatment of cerebral arteriovenous malformations: Buffalo score. Surg Neurol Int. 2015. 6: 3-
6. Fullerton HJ, Achrol AS, Johnston SC, McCulloch CE, Higashida RT, Lawton MT. Long-Term Hemorrhage risk in children versus adults with brain arteriovenous malformations. Stroke. 2005. 36: 2099-104
7. Gross BA, Du R. Natural history of cerebral arteriovenous malformations: A meta-analysis. J Neurosurg. 2013. 118: 437-43
8. Halim AX, Johnston SC, Singh V, McCulloch CE, Bennett JP, Achrol AS. Longitudinal risk of intracranial hemorrhage in patients with arteriovenous malformation of the brain within a defined population. Stroke. 2004. 35: 1697-702
9. Hofmeister C, Stapf C, Hartmann A, Sciacca RR, Mansmann U, terBrugge K. Demographic, Morphological and clinical characteristics of 1289 patients with brain arteriovenous malformation. Stroke. 2000. 31: 1307-10
10. Iwama T, Hayashida K, Takahashi JC, Nagata I, MHashimoto N. Cerebral hemodynamics and metabolism in patients with cerebral arteriovenous malformations: An evaluation using positron emission tomography scanning. J Neurosurg. 2002. 97: 1314-21
11. Kader A, Young WL, Pile-Spellman J, Mast H, Sciacca RR, Mohr JP. The influence of hemodynamic and anatomic factors on hemorrhage from cerebral arteriovenous malformations. Neurosurgery. 1994. 34: 801-7
12. Kim H, Su H, Weinsheimer S, Pawlikowska L, Young WL. Brain Arteriovenous malformation pathogenesis: A Response-to-injury Paradigm. Acta Neurochir Suppl. 2011. 111: 83-92
13. Kim H, Pourmohamad T, Westbroek EM, McCulloch CE, Lawton MT, Young WL. Evaluating performance of the spetzler-martin supplemented model in selecting patients with brain arteriovenous malformation for surgery. Stroke. 2012. 43: 2497-9
14. Lawton MT. Spetzler-Martin Grade III arteriovenous malformations: Surgical results and a modification of the grading scale. Neurosurgery. 2003. 52: 740-8
15. Lawton MT, Kim H, McCulloch CE, Mikhak B, Young WL. A supplementary grading scale for selecting patients with brain arteriovenous malformations for surgery. Neurosurgery. 2010. 66: 702-13
16. Lindegaard KF, Grolimund P, Aaslid R, Nornes H. Evaluation of cerebral AVM×s using transcranial Doppler ultrasound. J Neurosurg. 1986. 65: 335-44
17. Magro E, Gentric JC, Darsaut TE, Ziegler D, Msi , Bojanowski MW. Responses to ARUBA: A systematic review and critical analysis for the design of future arteriovenous malformation trials. J Neurosurg. 2017. 126: 486-94
18. Melo GG, Padilla VF, Escobar DVH, Mendizabal GR. Experiencia en el manejo de malformaciones arteriovenosas cerebrales en el Hospital Juarez de México. Rev Mex Neuroci. 2017. 18: 65-75
19. Melo-Guzman G, Padilla-Vazquez F, Escobar-de la Garma VH, Mendizabal GR. Experiencia en el manejo de malformaciones arteriovenosas cerebrales en el Hospital Juarez de México. Rev Mex Neuroci. 2017. 18: 65-75
20. Mohr JP, Parides MK, Stapf C, Moquete E, Moy CS, Overbey JR. Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): A multicentre, non-blinded, randomised trial. Lancet. 2014. 383: 614-21
21. Mohr JP, Moskowitz AF, Stapf C, Hartmann A, Lord K, Marshall SM. The ARUBA trial: Current Status, Future Hopes. Stroke. 2010. 41: e537-e540
22. Muñoz F, Clavel P, Molet J, Castaño C, de Teresa S, Solivera J. Current management of arteriovenous malformations, Retrospective study of 31 cases and literature review. Neurocirugía (Astur). 2007. 18: 394-405
23. Nornes H, Grip A. Hemodynamic aspects of cerebral arteriovenous malformations. J Neurosurg. 1980. 53: 456-64
24. Ondra SL, Troupp H, George ED, Schwab K. The natural history of symptomatic arteriovenous malformations of the brain: A 24-year follow-up assessment. J Neurosurg. 1990. 73: 387-91
25. Pandey P, Marks MP, Harraher CD, Westbroek EM, Chang SD, Do HM. Multimodality management of Spetzler-Martin Grade III arteriovenous malformations. J Neurosurg. 2012. 116: 1279-88
26. Peng T, Liu A, Jia J, Jiang C, Li Y, Wu Z. Risk factors for dural arteriovenous fistula intracranial haemorrhage. J Clin Neurosci. 2014. 21: 769-72
27. Reinard KA, Pabaney AH, Basheer A, Phillips SB, Kole MK, Malik GM. Surgical Management of giant intracranial arteriovenous malformations: A single center experience over 32 years. World Neurosurg. 2015. 84: 1765-78
28. Rutledge WC, Abla AA, Nelson J, Halbach VV, Kim H, Lawton MT. Treatment and outcomes of ARUBA-eligible patients with unruptured brain arteriovenous malformations at a single institution. Neurosurg Focus. 2014. 37: e8-
29. Shakur SF, Amin-Hanjani S, Mostafa H, Aletich VA, Charbel FT, Alaraj A. Relationship of pulsatility and resistance indices to cerebral arteriovenous malformation angioarchitectural features and haemorrhage. J Clin Neurosci. 2016. 33: 119-23
30. Spears J, Terbrugge KG, Moosavian M, Montanera W, Willinsky RA, Wallace MC. A discriminative prediction model of neurological outcome for patients undergoing surgery of brain arteriovenous malformations. Stroke. 2006. 37: 1457-64
31. Spetzler RF, Ponce FA. A 3-tier classification of cerebral arteriovenous malformations. Clinical article. J Neurosurg. 2011. 114: 842-9
32. Spetzler RF, Martin NA. A proposed grading system for arteriovenous malformations. J Neurosurg. 1986. 65: 76-83
33. Stapf C, Mast H, Sciacca RR, Choi JH, Khaw AV, Connolly ES. Predictors of hemorrhage in patients with untreated brain arteriovenous malformation. Neurology. 2006. p. 661350-5
34. Stefani MA, Porter PJ, terBrugge KG, Montanera W, Willinsky RA, Wallace MC. Large and Deep brain arteriovenous malformations are associated with risk of future hemorrhage. Stroke. 2002. 33: 1220-4
35. Valle RD, Zenteno M, Jaramillo J, Lee A, De Anda S. Definition of the key target volume in radiosurgical management of arteriovenous malformations: A new dynamic concept based on angiographic circulation time. J Neurosurg. 2008. 109: 41-50
36. Yamada S, Takagi Y, Nozaki K, Kikuta K, Hashimoto N. Risk factors for subsequent hemorrhage in patients with cerebral arteriovenous malformations. J Neurosurg. 2007. 107: 965-72
37. Zhao LB, Suh DC, Lee DG, Kim SJ, Kim JK, Han S. Association of pial venous reflux with hemorrhage or edema in dural arteriovenous fistula. Neurology. 2014. 82: 1897-904