- Department of Neurological Diseases and Neurosurgery, Peoples’ Friendship University of Russia, Moscow, Russia,
- Department of Neurosurgery, Morozov Children’s City Clinical Hospital, Center for the Treatment of Cerebrovascular Pathology in Children and Adolescents, Moscow, Russian Federation
- Department of Pediatrics and Child Health, Peoples Friendship University of Russia (RUDN University), Moscow, Russian Federation.
Correspondence Address:
Dimitri T. K. Ndandja, Department of Neurological Diseases and Neurosurgery, Peoples’ Friendship University of Russia, Moscow, Russia.
DOI:10.25259/SNI_308_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Dimitri T. K. Ndandja1, Gerald Musa1, Ilya Nosov2, Gennady E. Chmutin1, Matvey I. Livshitz2, Nazar S. Annanepesov1, Bupe Mumba Mwela3. A rare case of rebleeding brainstem cavernoma in a 5-month-old-girl. 30-Jun-2023;14:227
How to cite this URL: Dimitri T. K. Ndandja1, Gerald Musa1, Ilya Nosov2, Gennady E. Chmutin1, Matvey I. Livshitz2, Nazar S. Annanepesov1, Bupe Mumba Mwela3. A rare case of rebleeding brainstem cavernoma in a 5-month-old-girl. 30-Jun-2023;14:227. Available from: https://surgicalneurologyint.com/surgicalint-articles/12385/
Abstract
Background: Brainstem cavernomas (BSCs) are relatively rare intracranial vascular lesions that, if left untreated, can be devastating to the patient. The lesions are associated with a myriad of symptoms, depending on their size and location. However, medullary lesions present acutely with cardiorespiratory dysfunction. We present the case of a 5-month-old child with a BSC.
Case Description: A 5-month-old child presented for the 2nd time with sudden respiratory distress and excessive salivation. On the first presentation, brain magnetic resonance imaging (MRI) showed a 13 × 12 × 14 mm cavernoma at the pontomedullary junction. She was managed conservatively but presented 3 months later with tetraparesis, bulbar palsy, and severe respiratory distress. A repeat MRI showed enlargement of the cavernoma to 27 × 28 × 26 mm with hemorrhage in different stages. After hemodynamic stabilization, complete cavernoma resection was performed through the telovelar approach with neuromonitoring. Postoperatively, the child recovered motor function, but the bulbar syndrome persisted with hypersalivation. She was discharged on day 55 with a tracheostomy.
Conclusion: BSCs are rare lesions that are associated with severe neurological deficits due to the compactness of important cranial nerve nuclei and other tracts in the brainstem. Early surgical excision and hematoma evacuation for superficially presenting lesions can be lifesaving. However, the risk of postoperative neurological deficits is still a major concern in these patients.
Keywords: Brainstem hemorrhage, Brainstem, Bulbar palsy, Cavernoma, Telovelar approach
INTRODUCTION
Cavernous malformations, also known as cavernous angiomas, cavernous hemangiomas, or cavernomas, are well-circumscribed, low-flow, and angiographically occult lesions with no large arterial inflow or draining veins.[
CASE DESCRIPTION
A 5-month-old girl was admitted to the pediatric intensive care unit (ICU) with dyspnea, stridor, cyanosis, pronounced weakness, lethargy, and a progressively decreasing level of consciousness until she was comatose. This was the second admission to the ICU.
First admission
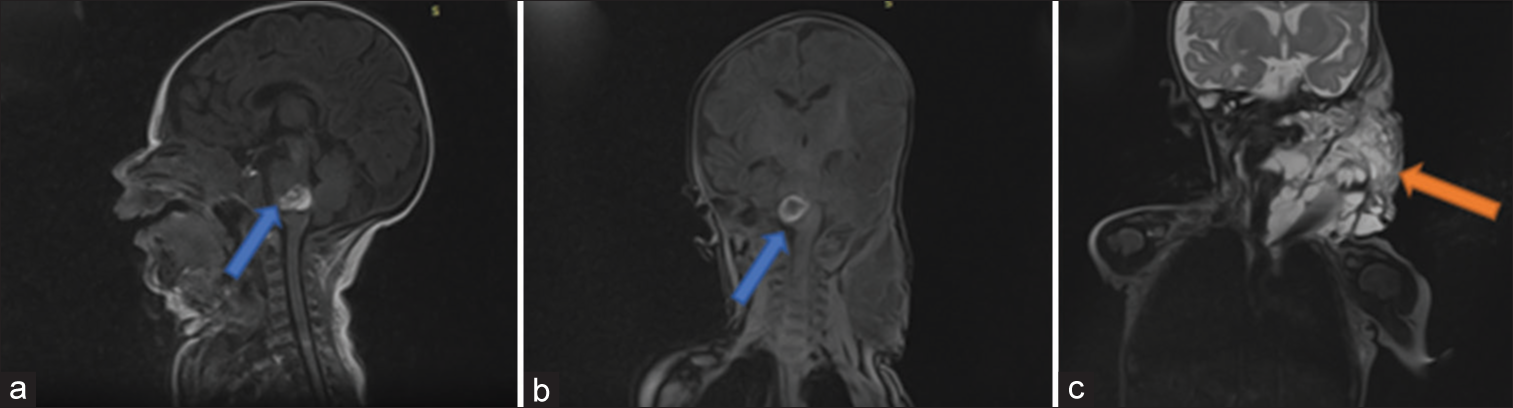
At 3 months old, the patient was admitted to the ICU on mechanical ventilation following sudden respiratory distress and excessive salivation. A head and neck computed tomography scan and magnetic resonance imaging (MRI) [
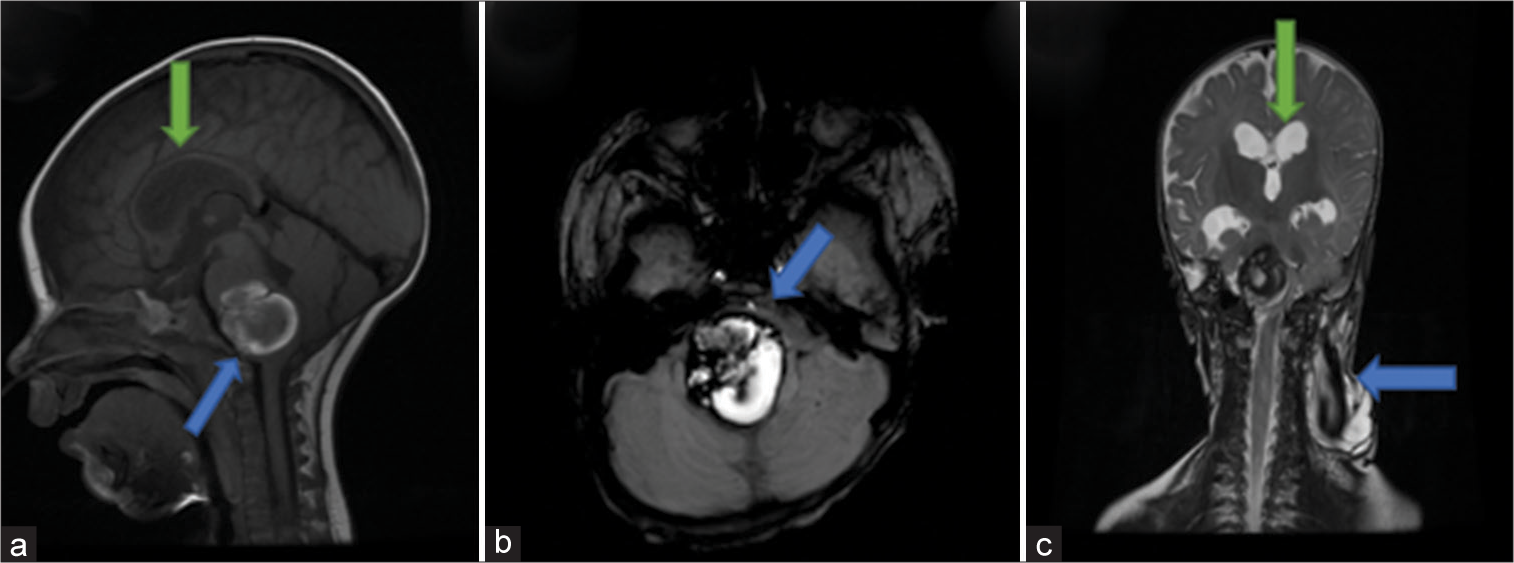
During the second admission, a brain MRI on admission revealed an increase in the size of the brainstem hematoma to 27 × 28 × 26 mm compared to the previous admission [
Figure 2:
Brain magnetic resonance imaging on the second admission. (a) Sagittal cut showing a 27 × 28 × 26 mm pontomedullary cavernoma (blue arrow) and hydrocephalus (green arrow). (b) Axial cut showing the heterogeneous lesion with a hemosiderin ring and severe compression of the brain stem (blue arrow). (c) Coronal cut showing hydrocephalus (green arrow) and significant regression of the lymphangioma (blue arrow) compared to magnetic resonance imaging on first admission.
On examination, the patient was in stupor with a four score of 10, with tetraparesis, bulbar palsy, and severe respiratory distress. In addition, she had metabolic and electrolyte disorders and anemia (Hb 80 g/L) with thrombocytosis (641 × 10*9/L).
Surgical technique
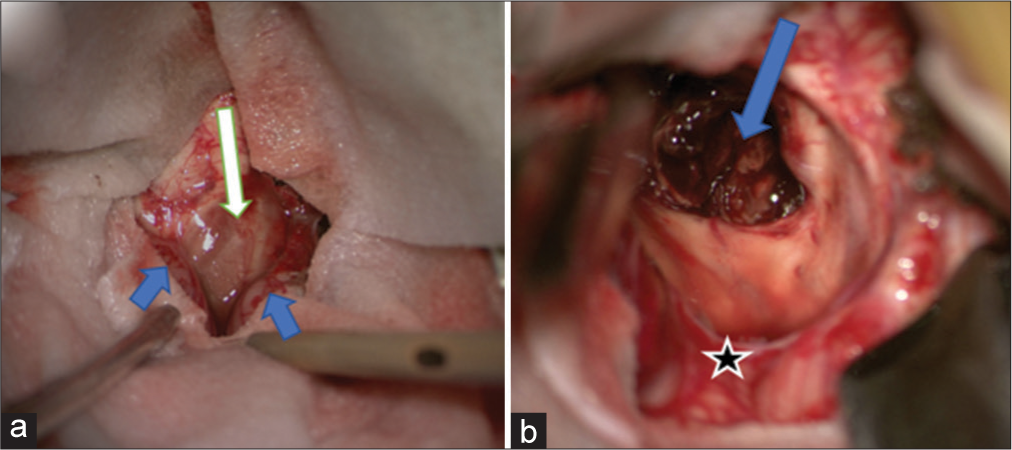
With the patient in the prone position, and the head fixed in a Mayfield 3 pin at maximum neck flexion, a standard telovelar approach to the brainstem through a midline suboccipital craniotomy was performed. The dura was opened in a curvilinear form, followed by dissection of the arachnoid between the tonsils to expose the telovelar membrane. With gentle tonsillar retraction, the telovelar membrane was opened, and the floor of the fourth ventricle was exposed. The dorsal surface of the upper medulla oblongata was seen bulging and hemosiderin-stained [
Figure 3:
Intraoperative images. (a) The exposure of the telovelor membrane and the bulging medulla oblongata with hemosiderin discoloration (white arrow). The cerebellar tonsils (blue arrows). (b) The opened cavernoma with the preserved developmental venous anomaly (DVA) (blue arrow). Cerebellar vermis (black star).
A small incision was made at the most presenting surface of the cavernoma, and the hematoma was evacuated and sent for histology [
There was no significant reduction in the brainstem-evoked potentials, and there was minimal iatrogenic injury to the neural tissue. After complete excision of the cavernoma, hemostasis was achieved using thin Surgicel slices, duroplasty was performed using NeoDura and Hemopatch grafts, and closure was performed as standard.
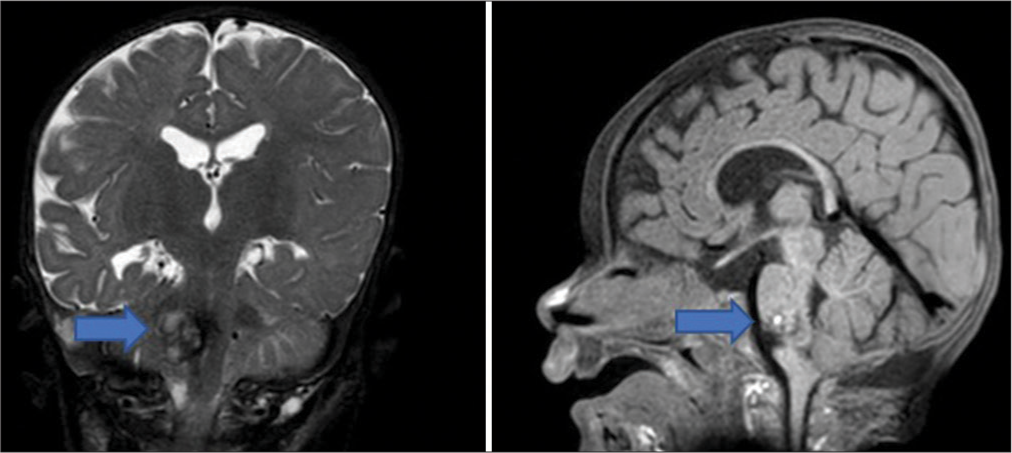
In the early postoperative period, the patient’s neurological functioning gradually improved. The patient was gradually weaned off of mechanical ventilation and transferred to auxiliary lung ventilation. By day 5, the patient had hemodynamically stabilized with a four score of 13. However, extubation was delayed considering the excessive salivation of approximately 500 mL/day, persistent bulbar palsy, and the patient’s age. A tracheostomy was performed on day 22 to manage the bulbar palsy. The child was discharged on hospital day 55 to continue outpatient treatment and rehabilitation. The 4 months postoperative MRI showed normal resolving postoperative changes [
Histopathology showed the typical hamartomatous cystic dilation of the single layered vascular spaces. Molecular and genetic studies showed no mutations associated with familial cavernomas like Krit 1.
DISCUSSION
Cavernous malformations are the most prevalent type of vascular malformation in the brain, with an incidence of 0.4–0.5% and a prevalence of 0.6/100,000 inhabitants. The condition is more prevalent in males between the ages of 20 and 40 and is found in approximately 25% of children. The dynamic nature of cavernous malformations, including enlargement, regression, and de novo formation, can be attributed to different stages of hemorrhage and hemorrhage resolution, which can be seen in characteristic MR images.[
The clinical course of brainstem cavernous malformations in children is highly variable, ranging from benign lesions to highly aggressive lesions with repetitive hemorrhages. Patients with brainstem cavernous malformations may present with cranial nerve deficits, sensory changes, headache, motor deficits of the extremities, diplopia, ataxia, and vertigo. Cranial nerve deficits are the most common early clinical presentation.[
Assessing symptoms in infants can be challenging, given the severity of the disease and the resulting neurological deficits.[
Rebleeding is more frequent in conservatively treated patients or those with incomplete cavernoma resection. The rebleeding rate is 10%/year, which is considered high in comparison with other cerebral vascular malformations.[
Given the longer life expectancy and greater functional recovery in children, surgical treatment should be considered early in young patients presenting with surgically accessible lesions and an aggressive clinical course.[
The telovelar approach is a less traumatic surgical approach that uses natural planes. Through a C1 laminectomy, the rostral extent of the intraventricular exposure can be expanded, giving more flexible inferior-to-superior working angles. Moreover, the telovelar approach offers a corridor through noneloquent arachnoid planes and a safe and adequate working environment.[
Using this approach with functional radiological studies, intraoperative navigation, and motor functional mapping allowed for precise resection of the lesion with minimal damage to the surrounding neural tissue. In addition to neuromonitoring, there are safe entry zones to the brainstem that has been shown to be safe and effective in avoiding injury to the vital nuclei and tracts.[
The patient regained significant motor function but had persistent bulbar syndrome on discharge, which was expected considering the size and location of the lesion. Close follow-up after surgery is essential for the early detection of potential complications, such as rebleeding, neurological deficits, or recurrence of the lesion. Imaging studies, such as MRI, can help monitor the progression of the lesion and detect any changes that may require further intervention.
CONCLUSION
BSCs are rare lesions that are associated with severe neurological deficits due to the compactness of important cranial nerve nuclei and other tracts in the brainstem. Early surgical excision and hematoma evacuation for superficially presenting lesions can be lifesaving. However, the risk of postoperative neurological deficits is still a major concern in these patients.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Abla AA, Turner JD, Mitha AP, Lekovic G, Spetzler RF. Surgical approaches to brainstem cavernous malformations. Neurosurg Focus. 2010. 29: E8
2. Arauz A, Patiño-Rodriguez HM, Chavarria-Medina M, Becerril M, Longo GM, Nathal E. Rebleeding and outcome in patients with symptomatic brain stem cavernomas. Cerebrovasc Dis. 2017. 43: 283-9
3. Catapano JS, Benner D, Rhodenhiser EG, Rumalla K, Graffeo CS, Srinivasan VM. Safety of brainstem safe entry zones: Comparison of microsurgical outcomes associated with superficial, exophytic, and deep brainstem cavernous malformations. J Neurosurg. 2022. p. 1-11
4. Deshmukh VR, Figueiredo EG, Deshmukh P, Crawford NR, Preul MC, Spetzler RF. Quantification and comparison of telovelar and transvermian approaches to the fourth ventricle. Neurosurgery. 2006. 58: ONS-202-6 discussion ONS-206-7
5. Ebrahim KS, Toubar AF. Telovelar approach versus transvermian approach in management of fourth ventricular tumors. Egypt J Neurosurg. 2019. 34: 1-10
6. Fotakopoulos G, Andrade-Barazarte H, Kivelev J, Tjahjadi M, Goehre F, Hernesniemi J. Brainstem cavernous malformations management: Microsurgery vs. Radiosurgery, a meta-analysis. Front Surg. 2022. 8: 630134
7. Gavrjushin A, Sufianov R. Resection of pontine cavernoma with hemorrhage via unilateral uvulotonsillar keyhole approach. World Neurosurg. 2022. 164: 256
8. Horne MA, Flemming KD, Su I, Stapf C, Jeon JP, Li D. Clinical course of untreated cerebral cavernous malformations: A meta-analysis of individual patient data. Lancet Neurol. 2016. 15: 166-73
9. Kupersmith MJ, Kalish H, Epstein F, Yu G, Berenstein A, Woo H. Natural history of brainstem cavernous malformations. Neurosurgery. 2001. 48: 47-54 discussion 53-4
10. Porter PJ, Willinsky RA, Harper W, Wallace MC. Cerebral cavernous malformations: Natural history and prognosis after clinical deterioration with or without hemorrhage. J Neurosurg. 1997. 87: 190-7
11. Salman RA, Hall JM, Horne MA, Moultrie F, Josephson CB, Bhattacharya JJ. Untreated clinical course of cerebral cavernous malformations: A prospective, population-based cohort study. Lancet Neurol. 2012. 11: 217-24
12. Santos AN, Rauschenbach L, Saban D, Chen B, Herten A, Dinger TF. Natural course of cerebral cavernous malformations in children: A five-year follow-up study. Stroke. 2022. 53: 817-24
13. Velz J, Özkaratufan S, Krayenbühl N, Beccaria K, Akeret K, Attieh C. Pediatric brainstem cavernous malformations: 2-center experience in 40 children. J Neurosurg Pediatr. 2022. 29: 1-12
14. Wang CC, Liu A, Zhang JT, Sun B, Zhao YL. Surgical management of brain-stem cavernous malformations: Report of 137 cases. Surg Neurol. 2003. 59: 444-54 discussion 454
15. Yasui T, Komiyama M, Iwai Y, Yamanaka K, Matsusaka Y, Morikawa T. A brainstem cavernoma demonstrating a dramatic, spontaneous decrease in size during follow-up: Case report and review of the literature. Surg Neurol. 2005. 63: 170-3 discussion 173
16. Yuen J, Whitfield P. Brainstem cavernous malformationsno longer a forbidden territory? A systemic review of recent literature. Neurochirurgie. 2020. 66: 116-26