- Department of Neurosurgery, Sendai Medical Centre,
- Department of Neurosurgery, Graduate School of Medicine, Tohoku University, Sendai, Japan.
Correspondence Address:
Atsuhiko Ninomiya
Department of Neurosurgery, Graduate School of Medicine, Tohoku University, Sendai, Japan.
DOI:10.25259/SNI_318_2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Atsuhiko Ninomiya, Atsushi Saito, Tomohisa Ishida, Tomoo Inoue, Takashi Inoue, Masayuki Ezura, Shinsuke Suzuki, Hiroshi Uenohara, Teiji Tominaga. A surgical case of cerebellar tuberculoma caused by a paradoxical reaction while on therapy for tuberculosis spondylitis. 09-Aug-2019;10:162
How to cite this URL: Atsuhiko Ninomiya, Atsushi Saito, Tomohisa Ishida, Tomoo Inoue, Takashi Inoue, Masayuki Ezura, Shinsuke Suzuki, Hiroshi Uenohara, Teiji Tominaga. A surgical case of cerebellar tuberculoma caused by a paradoxical reaction while on therapy for tuberculosis spondylitis. 09-Aug-2019;10:162. Available from: http://surgicalneurologyint.com/surgicalint-articles/9568/
Abstract
Background: A paradoxical reaction (PR) is a phenomenon in which the primary tuberculous lesion worsens or another de novo tuberculous lesion appears while on anti-tuberculosis therapy. Here, we report a rare case of cerebellar tuberculoma caused by a PR during therapy for lumbar tuberculous spondylitis (Pott’s disease).
Case Description: A 47-year-old male with human immunodeficiency virus seronegative was diagnosed with lumber tuberculous spondylitis (Pott’s disease) and prescribed anti-tuberculous agents. His lower back pain and inflammatory condition recovered after initiation of anti-tuberculous therapy. Two months later, he complained of headache, nausea, and staggering. Magnetic resonance images revealed a ring-enhanced lesion located at the cerebellar hemisphere extending to the vermis, which caused perifocal edema and bilateral ventriculomegaly. These findings were consistent with his symptoms of hydrocephalus. He did not have preceding clinical findings of meningitis, and a PR was suggested to cause de novo aggregation of cerebellar tuberculoma. A lesionectomy was performed, and the surgical specimen was pathologically diagnosed as a tuberculoma. He recovered well from neurological disorders after the resection.
Conclusion: De novo formation of intracranial tuberculoma alone caused by a PR without preceding meningitis is very rare. Lesionectomy is needed for intracranial tuberculoma, which manifests as a mass effect, as well as antituberculous therapy.
Keywords: Anti-tuberculous therapy, Cerebellar tumor, Paradoxical reaction, Pott’s disease, Spinal tuberculosis, Tuberculoma
INTRODUCTION
Tuberculosis is still a high mortality disease and major public health issue even in developed countries. In Japan, 13.9 cases per 1 million persons were annually diagnosed with tuberculosis in 2016, and Japan is considered as a middle-burden country.[
A paradoxical reaction (PR) is a phenomenon in which the primary tuberculous lesion worsens or another de novo tuberculous lesion appears during anti-tuberculous therapy although clinical symptoms improve initially after antituberculous therapy.[
Hereby, we experienced a rare surgical case of cerebellar tuberculoma caused by a PR during antituberculous therapy for tuberculous spondylitis. We report clinical characteristics of this patient and describe this case compared to previous studies.
CASE REPORT
A 47-year-old male with seronegative human immunodeficiency virus (HIV) and diabetes mellitus suffered from lower back pain. He was diagnosed with bacterial spondylitis and iliopsoas abscess [
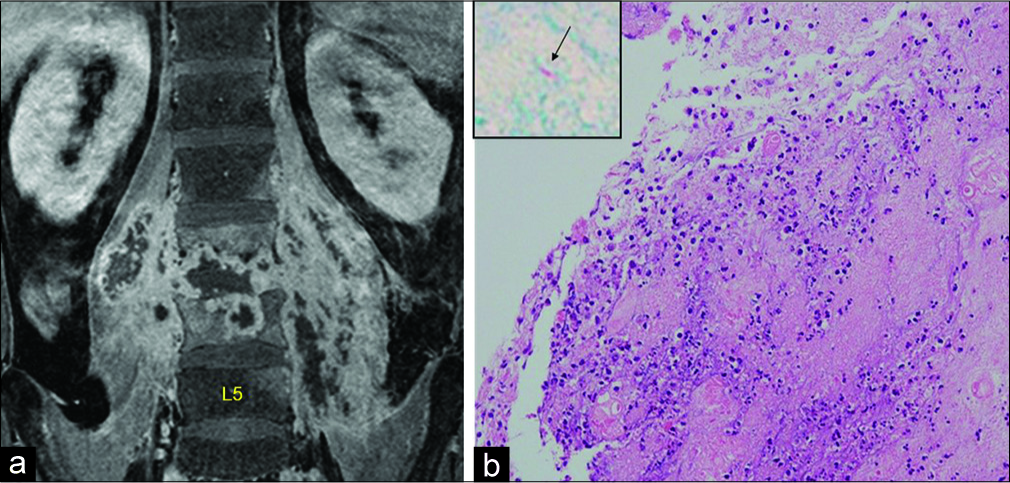
Figure 1:
(a) Gadolinium contrast-enhanced magnetic resonance imaging demonstrated inflammation of vertebral body at L3 and L4 with bilateral iliopsoas abscesses. (b) Microscopy of the surgical specimen of the vertebral interbody revealed granuloma aggregating of histiocytes by hematoxylin and eosin staining at low magnification. A punctured specimen from the abscess of iliopsoas muscle demonstrated Mycobacterium tuberculosis in the Ziehl–Neelsen stain at high magnification (left upper).
Two months after starting the antituberculous therapy, he complained of headache, nausea, and staggering. Physical findings on admission were that he was afebrile and did not have stiff neck. A right coordination deficit was identified, and other neurological deficits did not exist. Laboratory findings did not show inflammatory reaction. Chest computed tomography (CT) did not show any cavity lesions. Magnetic resonance (MR) images revealed a nodular enhanced lesion in the right cerebellar hemisphere extending to the vermis associated perifocal edema and bilateral ventriculomegaly [
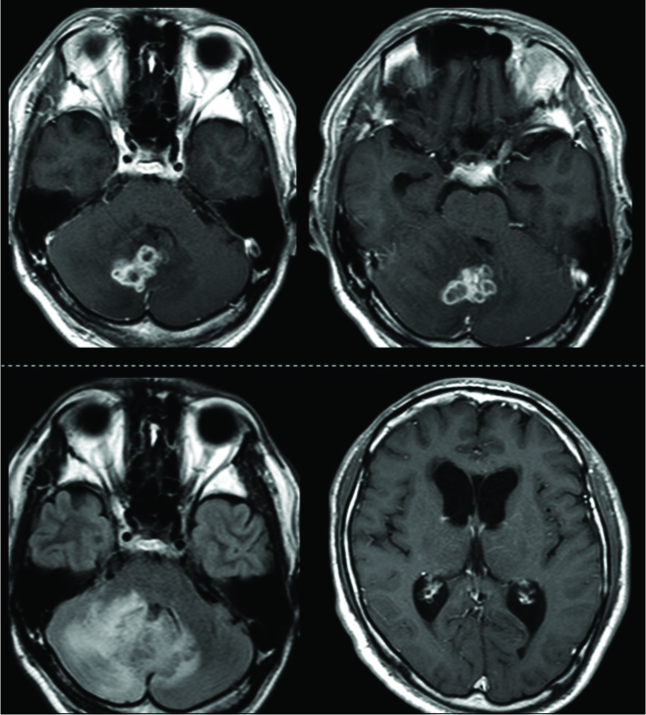
Figure 2:
Magnetic resonance images showed a nodular lesion with ring enhancement by gadolinium contrast medium in the cerebellar hemisphere extending to the vermis, compressing fourth ventricle (upper 2 slides). The lesion was associated with perifocal edema and bilateral ventriculomegaly (lower 2 slides).
Tuberculoma was suspected which appeared de novo or enlarged during the antituberculous therapy that was suggested to be a PR. The therapeutic strategy was a lesionectomy because he presented noncommunicating hydrocephalus, and malignant glioma could not be ruled out completely. Lesionectomy would be thought to relief noncommunicating hydrocephalus, and we did not plan to perform ventricular shunt.
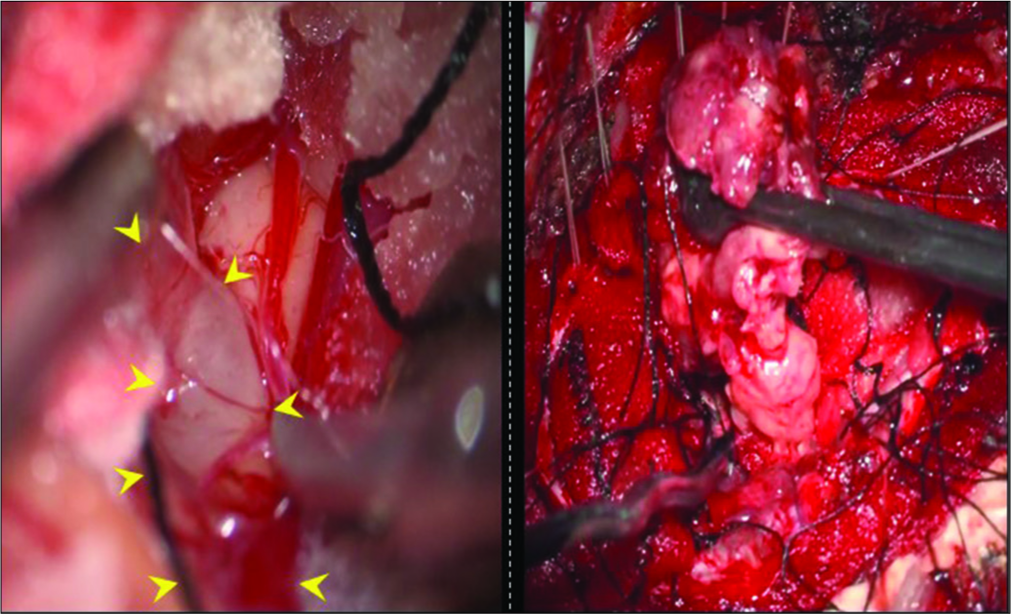
Surgery was performed by a midline occipital approach. A well-circumscribed mass with gray color was identified by opening the cerebellomedullary fissure [
Figure 4:
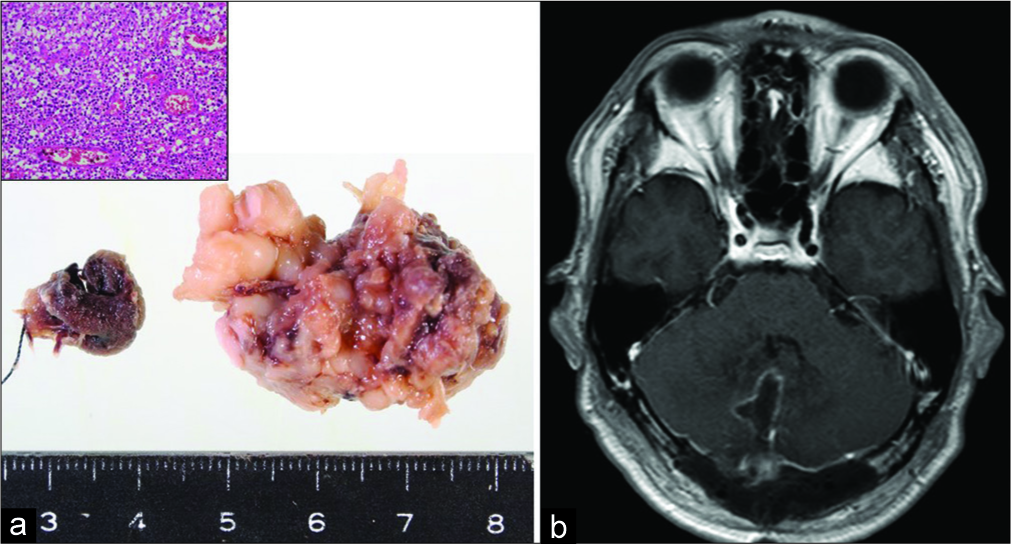
(a) Operative view: A lesionectomy was performed by opening the cerebellomedullary fissure, and a well-circumscribed mass with gray color encircled by arrowhead was identified (left). Total mass resection was performed (right). The size of the extracted mass was 3.5 cm. Pathology of the lesion revealed lymphocytes, macrophages, plasmacytes, and neutrophils aggregated around the tuberculoma by hematoxylin and eosin staining at 200-fold magnification (upper left). (b) Postoperative MRI showed a total mass resection.
DISCUSSION
The pathological mechanism of a PR is an exaggerated immunological reaction to viable microbes and debris of microbes destroyed by antituberculous therapy. This mechanism has been studied among patients who are coinfected with HIV and M. tuberculosis and received antiretroviral therapy (ART) and is relevant to the pathology of immune reconstitution inflammatory disease.[
Intracranial tuberculoma occurs in 1% of tuberculous patients.[
Here, we treated an immune-competent case with cerebellar tuberculoma caused by a PR during the therapy for tuberculous spondylitis. The cerebellar tuberculoma was suggested to be formed by PR against the preceding nonsymptomatic M. tuberculosis lesion in the cerebellum hematogenously infected from tuberculous spondylitis. He did not have a preceding clinical history of meningitis. Although the symptoms and clinical findings of tuberculous spondylitis markedly recovered after antituberculous therapy, annual neurological symptoms occurred after 2 months. The initiation of antituberculous therapy may induce the enlargement of the cerebellar lesion, which is considered a PR. Recently, it is reported that vertebral involvement of M. tuberculosis infection is a risk factor for PR of tuberculosis in central nervous system, which would support the clinical features of this case.[
Limitations of the present case were that the patient had not taken brain CT scans or MR images ahead of antituberculous therapy, and we could not identify when the lesion formed. However, he did not show any neurological symptoms before antituberculous therapy and enlargement of the lesion produced neurological disorders after the initiation of antituberculous therapy.
CONCLUSION
We treated a patient who had cerebellar tuberculoma caused by a PR and presented with neurological disorders during therapy for tuberculous spondylitis. Antituberculous therapy is the first choice for tuberculoma manifesting PR. Concurrent use of corticosteroids is recommended because of suppressing exaggerated immune reaction in tuberculoma causing PR and reducing perilesional brain edema. Surgery is considered for localized tuberculoma causing neurological symptoms.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Chambers ST, Hendrickse WA, Record C, Rudge P, Smith H. Paradoxical expansion of intracranial tuberculomas during chemotherapy. Lancet. 1984. 324: 181-4
2. Cheng VC, Ho PL, Lee RA, Chan KS, Chan KK, Woo PC. Clinical spectrum of paradoxical deterioration during antituberculosis therapy in non-HIV-infected patients. Eur J Clin Microbiol Infect Dis. 2002. 21: 803-9
3. Choudhury AR. Non-surgical treatment of tuberculomas of the brain. Br J Neurosurg. 1989. 3: 643-53
4. Farinha NJ, Razali KA, Holzel H, Morgan G, Novelli VM. Tuberculosis of the central nervous system in children: A 20-year survey. J Infect. 2000. 41: 61-8
5. Garg RK, Malhotra HS, Kumar N. Paradoxical reaction in HIV negative tuberculous meningitis. J Neurol Sci. 2014. 340: 26-36
6. Garg RK, Sharma R, Kar AM, Kushwaha RA, Singh MK, Shukla R. Neurological complications of miliary tuberculosis. Clin Neurol Neurosurg. 2010. 112: 188-92
7. Geri G, Passeron A, Heym B, Arlet JB, Pouchot J, Capron L. Paradoxical reactions during treatment of tuberculosis with extrapulmonary manifestations in HIV-negative patients. Infection. 2013. 41: 537-43
8. Gupta M, Bajaj BK, Khwaja G. Paradoxical response in patients with CNS tuberculosis. J Assoc Physicians India. 2003. 51: 257-60
9. Hagiya H, Koyama T, Zamami Y, Minato Y, Tatebe Y, Mikami N. Trends in incidence and mortality of tuberculosis in Japan: A population-based study, 1997-2016. Epidemiol Infect. 2018. 147: 1-10
10. Hejazi N, Hassler W. Multiple intracranial tuberculomas with atypical response to tuberculostatic chemotherapy: Literature review and a case report. Acta Neurochir (Wien). 1997. 139: 194-202
11. Hirasawa Y, Terada J, Kono C, Yamada Y, Sasaki Y, Tatsumi K. A case of repeated paradoxical reactions in tuberculous meningitis with miliary tuberculosis: Comparisons between the brain MRI and chest CT findings. Nihon Kokyuki Gakkai Zasshi. 2016. 5: 326-30
12. Kestens L, Seddiki N, Bohjanen PR. Immunopathogenesis of immune reconstitution disease in HIV patients responding to antiretroviral therapy. Curr Opin HIV AIDS. 2008. 3: 419-24
13. Liu Y, Wang Z, Yao G, Lu Y, Hu Z, Yao H. Paradoxical reaction in HIV-negative tuberculous meningitis patients with spinal involvement. Int J Infect Dis. 2019. 79: 104-8
14. Malone JL, Paparello S, Rickman LS, Wagner KF, Monahan B, Oldfield EC. Intracranial tuberculoma developing during therapy for tuberculous meningitis. West J Med. 1990. 152: 188-90
15. Marais S, Scholtz P, Pepper DJ, Meintjes G, Wilkinson RJ, Candy S. Neuroradiological features of the tuberculosis-associated immune reconstitution inflammatory syndrome. Int J Tuberc Lung Dis. 2010. 14: 188-96
16. Narita M, Ashkin D, Hollender ES, Pitchenik AE. Paradoxical worsening of tuberculosis following antiretroviral therapy in patients with AIDS. Am J Respir Crit Care Med. 1998. 158: 157-61
17. Nicolls DJ, King M, Holland D, Bala J, del Rio C. Intracranial tuberculomas developing while on therapy for pulmonary tuberculosis. Lancet Infect Dis. 2005. 5: 795-801
18. Okazaki A, Watanabe S, Yoneda T, Hara J, Nishitsuji M, Nishi K. Paradoxical reaction to antituberculosis therapy after 6 months of treatment for pulmonary tuberculosis: A case report. J Infect Chemother. 2016. 22: 748-51
19. Rajshekhar V. Surgery for brain tuberculosis: A review. Acta Neurochir (Wien). 2015. 157: 1665-78
20. Rock RB, Olin M, Baker CA, Molitor TW, Peterson PK. Central nervous system tuberculosis: Pathogenesis and clinical aspects. Clin Microbiol Rev. 2008. 21: 243-61
21. Sáenz B, Hernandez-Pando R, Fragoso G, Bottasso O, Cárdenas G. The dual face of central nervous system tuberculosis: A new janus bifrons. ? Tuberculosis (Edinb). 2013. 93: 130-5
22. Tai ML, Nor HM, Kadir KA, Viswanathan S, Rahmat K, Zain NR. Paradoxical manifestation is common in HIV-negative tuberculous meningitis. Medicine (Baltimore). 2016. 95: e1997-
23. Thwaites G, Fisher M, Hemingway C, Scott G, Solomon T, Innes J. British infection society guidelines for the diagnosis and treatment of tuberculosis of the central nervous system in adults and children. J Infect. 2009. 59: 167-87