- Department of Neurosurgery, Hospital Santa Maria, Lisboa Portugal.
- Department of Neurological Imaging, Hospital Santa Maria, Lisboa Portugal.
- Department of Neurosurgery, Hospital Garcia de Orta, Almada, Portugal.
# These are two co-first authors.
Correspondence Address:
Samuel Sequeira Lemos, Department of Neurosurgery, Hospital Santa Maria, Lisboa, Portugal.
DOI:10.25259/SNI_111_2024
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Samuel Sequeira Lemos1, Maria Inês Sá2, Carlos Daniel Casimiro3, Carla Guerreiro2, Claudia Faria1. A ventral brainstem neurenteric cyst – A case report and review of the pre-brainstem location. 07-Jun-2024;15:195
How to cite this URL: Samuel Sequeira Lemos1, Maria Inês Sá2, Carlos Daniel Casimiro3, Carla Guerreiro2, Claudia Faria1. A ventral brainstem neurenteric cyst – A case report and review of the pre-brainstem location. 07-Jun-2024;15:195. Available from: https://surgicalneurologyint.com/surgicalint-articles/12926/
Abstract
Background: Neurenteric cysts are uncommon, benign endoderm-derived lesions that result from aberrant embryologic development of the notochord. They are typically located in the intradural extramedullary spinal cord and rarely located intracranially. Contrary to spinal-located cysts, intracranial cysts are rarer in the pediatric population. Clinically, they may present with symptoms of mass effect, or they can be incidentally discovered.
Case Description: A 10-year-old healthy female child presented with recurrent headaches. The physical and neurological examination was unremarkable. Brain magnetic resonance imaging (MRI) showed a well-demarcated lesion anterior to the pontomedullary junction with striking T1 and T2/T2 fluid-attenuated inversion recovery high-signal intensity and a small rounded nodule within of low signal on T1, T2, and T2*. On initial conservative strategy with serial brain MRI, there was a progressive enlargement of the lesion with significant mass effect on the brainstem. The patient underwent a right retrosigmoid craniotomy, and the cyst wall was fenestrated and drained. Part of the cyst wall and the solid nodule were adherent to the brainstem and basilar artery and were not removed. The histologic findings were consistent with the diagnosis of a benign endodermal cyst. The postoperative period was uneventful.
Conclusion: We report a successful surgical treatment of this rare congenital cyst located in the ventral brainstem. We present pre-and post-operative imaging findings, intraoperative microscopic images of the procedure, and a brief review of relevant clinical literature on the topic.
Keywords: Brainstem, Endodermal cyst, Intracranial neurenteric cyst, Retrosigmoid craniotomy, Ventral
INTRODUCTION
Neurenteric cysts (NECs) are uncommon, benign lesions and are regarded as ectopic endodermal cysts lined by respiratory or gastrointestinal mucin-secreting epithelium. They are thought to arise during the third embryonic week after abnormal or late closure of the neurenteric canal which communicates endoderm and ectoderm through the notochord.[
They are typically located in the intradural extramedullary ventral spinal cord and rarely located intracranially. Intracranially, they are typically found in the pre-pontine cistern, cerebellopontine cistern, or in the 4th ventricle.[
The best surgical management remains controversial due to the rarity of the condition and the limited follow-up data. Complete resection of the NEC, including the cyst wall, is generally recommended.[
We present a case of a 10-year-old child with an NEC located ventrally to the BS, which showed progressive growth in follow-up exams. We present the pre-and post-operative imaging findings and a brief review of the relevant clinical literature on the topic.
CASE DESCRIPTION
History and imaging
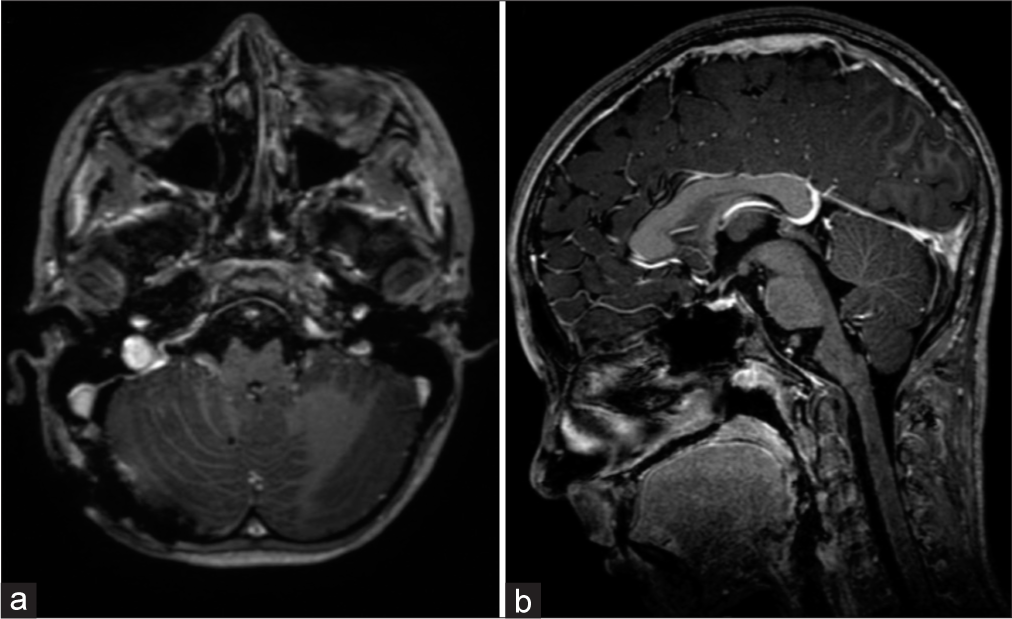
A 10-year-old healthy female child presented with recurrent occipital headaches over 1 year. The physical and neurological examinations were unremarkable. Head computed tomography imaging showed a small, rounded, hyperdense lesion located anterior to the pontomedullary junction. On MRI, the lesion seemed extra-axial and was mostly hyperintense on T1 and T2/T2 fluid-attenuated inversion recovery, with facilitated diffusion, no chemical-shift artifact (not a fatty lesion), and no gadolinium enhancement, with a small nodule within of low signal on T1, T2, and T2*, suggestive of calcification. The lesion measured about 10 × 9 × 8 mm in size (anterior-posterior × transversal × craniocaudal) and 280 mm3 of volume and had no surrounding edema, with a slight mass effect on the BS [
On an initial conservative strategy with serial follow-up brain MRIs, there was a progressive growth of the lesion, which became more lobulated and extended to the pre-bulbar and pre-pontine cisterns, measuring 19 × 28 × 23 mm and 5400 mm3 of volume, with significant mass effect. The prepontine vasculature was further anteriorly displaced but not encased. This led to the decision of neurosurgical treatment [
Figure 2:
Brain magnetic resonance imaging on a 2-year follow-up, showing an increase in lesion volume and mass effect on the brainstem. A small nodule within of low signal on T1, T2, and T2* is apparent. An anteriorly displaced but not encased basilar artery is also visible (a) sagittal T1, (b) sagittal T1 with contrast, (c) axial T2, and (d) axial T2.
Operative details
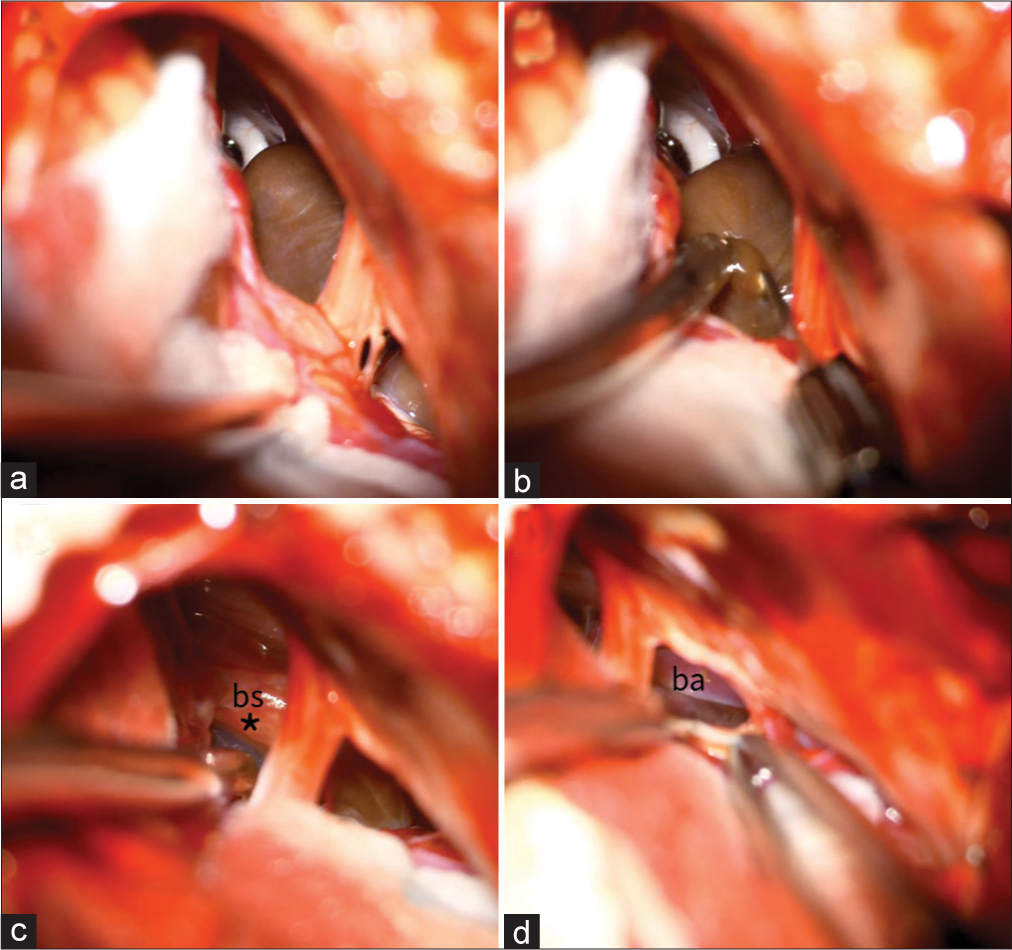
The patient was positioned in a lateral position, and we performed a right retro mastoid craniotomy with an extension to the foramen magnum. On an operating microscope and neuronavigation-assisted, the cyst wall was exposed, widely fenestrated, and its fluid was drained, revealing a thick yellow liquid content. After aspiration and profuse irrigation of the cyst, part of the cyst wall was removed. The part of the cyst wall adherent to the BS and basilar artery was not removed [
Figure 3:
(a) Exposure of the lesion’s capsule through the cerebellopontine angle between the 5th and the 7th nerve and its inferior part of the cyst viewed between the 7th and the 9th nerves, (b) the capsule was opened, and a dense yellow fluid was aspirated, and (c and d) final view after fluid drainage and partial removal of the capsule, part of the capsule (*) left adherent overlying the brainstem (bs). The basilar artery (ba) is also viewed.
Histopathology and postoperative details
The histologic findings were of a pseudo-stratified epithelium with no anaplasia, consistent with the diagnosis of a benign endodermal cyst. In the postoperative period, the patient experienced no neurological alterations, and postoperative MRI showed complete drainage of the cyst and the persistence of a small T2 hypointense nodule. At 1 year follow-up, the patient was well, active, and with no symptoms, and imaging showed no evidence of recurrence of the lesion [
DISCUSSION
Intracranial NEC accounts for 0.01% of central nervous system tumors.[
In the presented case, despite the significant growth of the lesion, the patient had no neurological signs, which we relate to the slow growth of the cyst to the prepontine cistern. This contrasts with some cases that reported quicker growth.[
Despite its rarity, the imaging features of an intracranial NEC can be suggestive of the diagnosis. NEC can present with a variety of different signal intensities on T1 and T2 related to differences in protein levels, rendering the diagnosis challenging.[
The existence of a nodule has been attributed to calcification or previous hemorrhage, but in a recent report of removal of such an associated nodule, the histologic analysis showed a xanthogranulomatous reaction with melanin. It has been proposed that this nodule/hemorrhage favors inflammation, which might explain the transformation from a silent cyst to an active cyst with headache and cyst expansion.[
Complete surgical resection is the gold standard for the treatment of NEC and is associated with the most favorable outcomes.[
Despite a 1-year follow-up MRI showing no signs of recurrence, a long follow-up is recommended since an age under 30 years old is a risk factor for recurrence.[
In recurrent cases, different approaches have been reported. Newly formed adhesions may complicate reoperation with new drainage and further attempt to remove the cyst wall. A case of an intra-axial BS recurrent NEC was treated with intra-cystic Interferon-α and another with intra-cystic H2O2, with good outcomes.[
CONCLUSION
We report a surgical resection of this rare congenital cyst located ventral to the BS. This case adds to previously reported cases on the management of prepontine NEC in which adherence to critical structures precludes total removal. It also evokes questions on the natural history of the lesion and the causes of growth. More data on this pathology and its natural history are needed to optimize management and recurrence avoidance.
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Agresta G, Sokol D, Kaliaperumal C, Kandasamy J, Gallo P. A novel management proposal for intrinsic brainstem neurenteric cysts: Case report. J Neurosurg Pediatr. 2020. 25: 83-7
2. Anderson T, Kaufman T, Murtagh R. Intracranial neurenteric cyst: A case report and differential diagnosis of intracranial cystic lesions. Radiol Case Rep. 2020. 15: 2649-54
3. Birinyi PV, Bieser S, Reis M, Guzman MA, Agarwal A, Abdel-Baki MS. Impact of DTI tractography on surgical planning for resection of a pediatric pre-pontine neurenteric cyst: A case discussion and literature review. Childs Nerv Syst. 2015. 31: 457-63
4. Chen CT, Lee CY, Lee ST, Chang CN, Wei KC, Wu CT. Neurenteric cysts: Risk factors and management of recurrence. Acta Neurochir. 2016. 158: 1325-31
5. Gauden AJ, Khurana VG, Tsui AE, Kaye AH. Intracranial neuroenteric cysts: A concise review including an illustrative patient. J Clin Neurosci. 2012. 19: 352-9
6. Harris CP, Dias MS, Brockmeyer DL, Townsend JJ, Willis BK, Apfelbaum RI. Neurenteric cysts of the posterior fossa: Recognition, management, and embryogenesis. Neurosurgery. 1991. 29: 893-7 discussion 897-8
7. Hayashi Y, Tachibana O, Muramatsu N, Tsuchiya H, Tada M, Arakawa Y. Rathke cleft cyst: MR and biomedical analysis of cyst content. J Comput Assist Tomogr. 1999. 23: 34-8
8. Ko BS, Jung S, Jung TY, Moon KS, Kim IY, Kang SS. Neurenteric cyst with xanthomatous changes in the prepontine area: Unusual radiological findings. J Neurosurg Pediatr. 2008. 2: 351-4
9. Oliveira RS, Cinalli G, Roujeau T, Sainte-Rose C, Pierre-Kahn A, Zerah M. Neurenteric cysts in children: 16 consecutive cases and review of the literature. J Neurosurg. 2005. 103: 512-23
10. Osborn AG, Preece MT. Intracranial cysts: Radiologicpathologic correlation and imaging approach. Radiology. 2006. 239: 650-64
11. Pang D, Dias MS, Ahab-Barmada M. Split cord malformation part I: A unified theory of embryogenesis for double spinal cord malformations. Neurosurgery. 1992. 31: 451-80
12. Shimizu Y, Fujita N, Akiyama O, Suzuki M, Kondo A. A rare presentation of a pediatric neurenteric cyst as an intra-axial pontine lesion: A case report with a 5-year follow-up. Surg Neurol Int. 2019. 10: 236
13. Wang L, Zhang J, Wu Z, Jia G, Zhang L, Hao S. Diagnosis and management of adult intracranial neurenteric cysts. Neurosurgery. 2011. 68: 44-52
14. Wong ST, Moes GS, Yam KY, Fong D, Pang D. Intrinsic brainstem neurenteric cyst with extensive squamous metaplasia in a child. J Neurosurg Imaging Tech. 2016. 1: 26-37
15. Yamamoto J, Shimajiri S, Akiba D, Nakano Y, Nishizawa S. Intracranial neurenteric cyst with an enhanced mural nodule and melanin pigmentation: Radiologic-pathologic correlation. World Neurosurg. 2017. 97: 758.e11-9