- CNS Neurosurgery, Woolloomooloo, New South Wales, Australia.
DOI:10.25259/SNI_6_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Vini G. Khurana. Adverse impact of smoking on the spine and spinal surgery. 24-Mar-2021;12:118
How to cite this URL: Vini G. Khurana. Adverse impact of smoking on the spine and spinal surgery. 24-Mar-2021;12:118. Available from: https://surgicalneurologyint.com/surgicalint-articles/10662/
Abstract
Background: Tobacco smokers and companies are well aware that smoking increases the risks for cancers, vascular morbidity, and early mortality. This is a review of the plethora of adverse effects chronic smoking has on spinal tissues and spinal surgery.
Methods: Medline (PubMed) and Google Scholar databases were searched for pertinent literature through keywords related to smoking, spondylosis, and spinal surgery.
Results: Smoking accelerates spondylosis by impairing spinal tissue vascular supply through atherosclerosis and thrombosis, while inducing local hypoxia, inflammation, proteolysis, and cell loss. It, thus, compromises disc, cartilage, synovium, bone, and blood vessels. It can lead to early surgery, delayed wound healing, increased surgical site infection, failed fusion, more re-operations, and chronic spinal pain.
Conclusion: There is ample evidence to support surgeons’ declining to operate on chronic smokers. The need for immediate and permanent smoking cessation and its potential benefits should be emphasized for the patient considering or who has undergone spinal surgery.
Keywords: Pseudarthrosis, Smoking, Spinal fusion, Spondylosis, Surgery, Tobacco
INTRODUCTION
Mainstream cigarette smoke drawn into a smoker’s mouth consists of 8% tar and 92% gaseous components and contains thousands of toxic chemical compounds, about 1015 free radicals per puff, and the addictive substance, nicotine.[
METHODS
The literature was reviewed using keyword searches on Medline (PubMed) and Google Scholar search engines. Keywords included smoking, complications of spinal surgery, disc arthroplasty, disc degeneration, facet joint arthropathy, infection, pseudarthrosis, spinal fusion, spondylosis, vertebral bone, and wound healing.
RESULTS
Disc degeneration
The nutritional supply of intervertebral discs, which are nonvascular tissues, depends on diffusion from adjacent vertebral bodies.[
Cartilage degradation and facet arthropathy
Smoking alters joint tissue gene expression toward promotion of autoimmunity and chronic inflammation.[
Bone compromise
Osteoblast function, cellularity, bone mineral content, tensile strength, and vertebral body blood supply are all compromised by smoking/tobacco use.[
Vascular compromise
The gas and tar components of cigarette smoke contribute to oxidative stress attributed to reactive oxygen species that, in turn, contribute to atherosclerosis, increased inflammation, platelet aggregation, and thrombosis.[
Infection and impaired wound healing
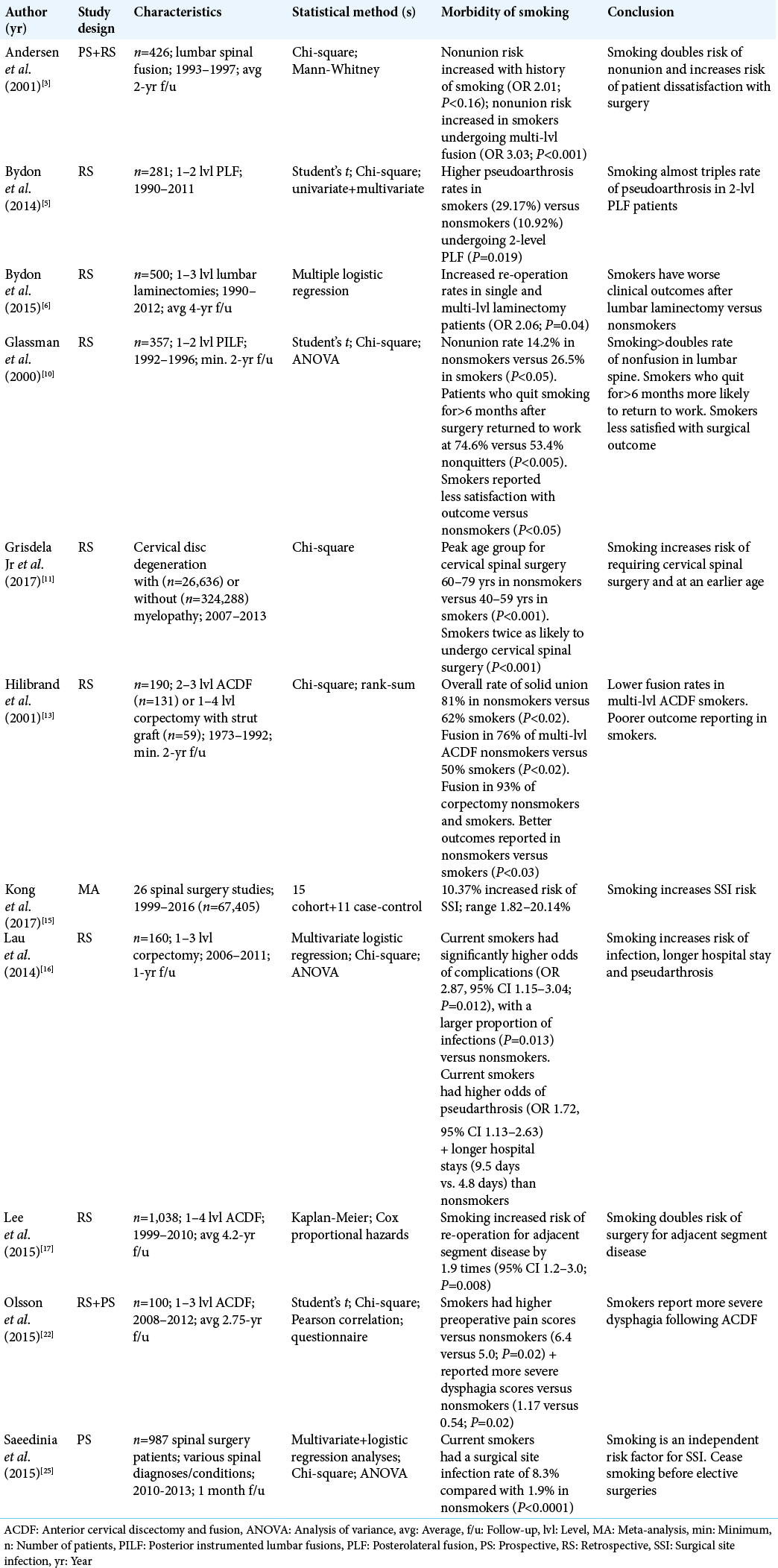
Meta-analyses have shown that smokers have significantly increased rates of spinal surgical site complications including more skin incision necrosis, delayed wound healing, more wound dehiscence, and increased susceptibility to infection [Table 1].[
Earlier operation and increased reoperation rate
Tobacco smokers are more likely to develop symptomatic cervical spondylotic myelopathy, typically warranting spinal surgery approximately two decades earlier than their nonsmoking counterparts.[
Smoking increases rates of fusion failure
Smoking is known to impede all stages of bone healing and fusion.[
CONCLUSION
Through a multitude of pathophysiological mechanisms, smoking negatively impacts the structure of the spine and the results of spinal surgery.[
Declaration of patient consent
Patient consent not required as patient identity not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ambrose JA, Barua RS. The pathophysiology of cigarette smoking and cardiovascular disease: An update. J Am Coll Cardiol. 2004. 43: 1731-7
2. Amin S, Niu J, Guermazi A, Grigoryan M, Hunter DJ, Clancy M. Cigarette smoking and the risk for cartilage loss and knee pain in men with knee osteoarthritis. Ann Rheum Dis. 2007. 66: 18-22
3. Andersen T, Christensen FB, Laursen M, Høy K, Hansen ES, Bünger C. Smoking as a predictor of negative outcome in lumbar spinal fusion. Spine (Phila Pa 1976). 2001. 26: 2623-8
4. Berman D, Oren JH, Bendo J, Spivak J. The effect of smoking on spinal fusion. Int J Spine Surg. 2017. 11: 229-38
5. Bydon M, de la Garza-Ramos R, Abt NB, Gokaslan ZL, Wolinsky JP, Sciubba DM. Impact of smoking on complication and pseudarthrosis rates after single-and 2-level posterolateral fusion of the lumbar spine. Spine (Phila Pa 1976). 2014. 39: 1765-70
6. Bydon M, Macki M, de la Garza-Ramos R, Sciubba DM, Wolinsky JP, Gokaslan ZL. Smoking as an independent predictor of reoperation after lumbar laminectomy: A study of 500 cases. J Neurosurg Spine. 2015. 22: 288-93
7. Chen T, Ehnert S, Tendulkar G, Zhu S, Arnscheidt C, AsperaWerz RH. Primary human chondrocytes affected by cigarette smoke-therapeutic challenges. Int J Mol Sci. 2020. 21: 1901
8. Chen Z, Li X, Pan F, Wu D, Li H. A retrospective study: Does cigarette smoking induce cervical disc degeneration?. Int J Surg. 2018. 53: 269-73
9. Fogelholm RR, Alho AV. Smoking and intervertebral disc degeneration. Med Hypotheses. 2001. 56: 537-9
10. Glassman SD, Anagnost SC, Parker A, Burke D, Johnson JR, Dimar JR. The effect of cigarette smoking and smoking cessation on spinal fusion. Spine (Phila Pa 1976). 2000. 25: 2608-15
11. Grisdela P, Buser Z, D’Oro A, Paholpak P, Liu JC, Wang JC. Trends analysis of surgical procedures for cervical degenerative disc disease and myelopathy in patients with tobacco use disorder. Eur Spine J. 2017. 26: 2386-92
12. Hadley MN, Reddy SV. Smoking and the human vertebral column: A review of the impact of cigarette use on vertebral bone metabolism and spinal fusion. Neurosurgery. 1997. 41: 116-24
13. Hilibrand A, Fye MA, Emery SE, Palumbo MA, Bohlman HH. Impact of smoking on the outcome of anterior cervical arthrodesis with interbody or strut-grafting. J Bone Joint Surg Am. 2001. 83: 668-73
14. Jackson KL 2nd, Devine JG. The effects of smoking and smoking cessation on spine surgery: A systematic review of the literature. Global Spine J. 2016. 6: 695-701
15. Kong L, Liu Z, Meng F, Shen Y. Smoking and risk of surgical site infection after spinal surgery: A systematic review and meta-analysis. Surg Infect (Larchmt). 2017. 18: 206-14
16. Lau D, Chou D, Ziewacz JE, Mummaneni PV. The effects of smoking on perioperative outcomes and pseudarthrosis following anterior cervical corpectomy: Clinical article. J Neurosurg Spine. 2014. 21: 547-58
17. Lee JC, Lee SH, Peters C, Riew KD. Adjacent segment pathology requiring reoperation after anterior cervical arthrodesis: The influence of smoking, sex, and number of operated levels. Spine (Phila Pa 1976). 2015. 40: 571-7
18. Lehr HA. Microcirculatory dysfunction induced by cigarette smoking. Microcirculation. 2000. 7: 367-84
19. Miķelsone I, Bormane I, Simsone Z, Jurka A, Tretjakovs P. The effect of chronic cigarette smoking on microvascular function, insulin resistance and inflammatory state. Environ Exp Biol. 2011. 9: 23-8
20. Mills E, Eyawo O, Lockhart I, Kelly S, Wu P, Ebbert JO. Smoking cessation reduces postoperative complications: A systematic review and meta-analysis. Am J Med. 2011. 124: 144-54
21. National Centre for Chronic Disease Prevention and Health Promotion. Centers for Disease Control and Prevention. Tips from Former Smokers. Available from: https://www.cdc.gov/tobacco/campaign/tips [Last accessed on 2021 Jan 02].
22. Olsson EC, Jobson M, Lim MR. Risk factors for persistent dysphagia after anterior cervical spine surgery. Orthopedics. 2015. 384: 319-23
23. Ospelt C, Camici GG, Engler A, Kolling C, Vogetseder A, Gay RE. Smoking induces transcription of the heat shock protein system in the joints. Ann Rheum Dis. 2014. 73: 1423-6
24. Patel RA, Wilson RF, Patel PA, Palmer RM. The effect of smoking on bone healing: A systematic review. Bone Joint Res. 2013. 2: 102-11
25. Saeedinia S, Nouri M, Azarhomayoun A, Hanif H, Mortazavi A, Bahramian P. The incidence and risk factors for surgical site infection after clean spinal operations: A prospective cohort study and review of the literature. Surg Neurol Int. 2015. 6: 154
26. Sørensen LT. Wound healing and infection in surgery: The pathophysiological impact of smoking, smoking cessation, and nicotine replacement therapy: A systematic review. Ann Surg. 2012. 255: 1069-79
27. Wang ZC, Shi JG, Chen XS, Xu GH, Li LJ, Jia LS. The role of smoking status and collagen IX polymorphisms in the susceptibility to cervical spondylotic myelopathy. Genet Mol Res. 2012. 11: 1238-44
28. Wong PK, Christie JJ, Wark JD. The effects of smoking on bone health. Clin Sci (Lond). 2007. 113: 233-41