- Departments of Neurological Surgery, University of Texas Southwestern, Dallas, Texas, United States.
Correspondence Address:
Salah G. Aoun, Department of Neurological Surgery, University of Texas Southwestern, Dallas, Texas, United States.
DOI:10.25259/SNI_759_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: James P. Caruso, Chen Shi, Benjamin Rail, Salah G. Aoun, Carlos A. Bagley. Aggressively recurring cervical intramedullary anaplastic astrocytoma in a pregnant patient. 13-Sep-2021;12:466
How to cite this URL: James P. Caruso, Chen Shi, Benjamin Rail, Salah G. Aoun, Carlos A. Bagley. Aggressively recurring cervical intramedullary anaplastic astrocytoma in a pregnant patient. 13-Sep-2021;12:466. Available from: https://surgicalneurologyint.com/surgicalint-articles/11104/
Abstract
Background: Many patients with spinal juvenile pilocytic astrocytoma can experience prolonged remission after resection. However, some reports suggest that pregnancy may be associated with progression.
Case Description: The authors provide an image report highlighting a case of rapid and aggressive transformation of an intramedullary astrocytoma of the cervical spine in a pregnant patient. Over the course of 1 year, the lesion progressed from a juvenile pilocytic astrocytoma to an anaplastic astrocytoma. Genetic testing revealed mutations associated with aggressive behavior.
Conclusion: The case and associated imaging demonstrate the importance of close neurologic monitoring and counseling regarding risk of progression in pregnant patients with spinal gliomas.
Keywords: Anaplastic astrocytoma, FGFR1, Intramedullary, Pilocytic astrocytoma, SETD2, TACC1
INTRODUCTION
While gliomas account for ~80% of primary malignant brain tumors, primary glioma of the spine remains a rare finding.[
CLINICAL IMAGE
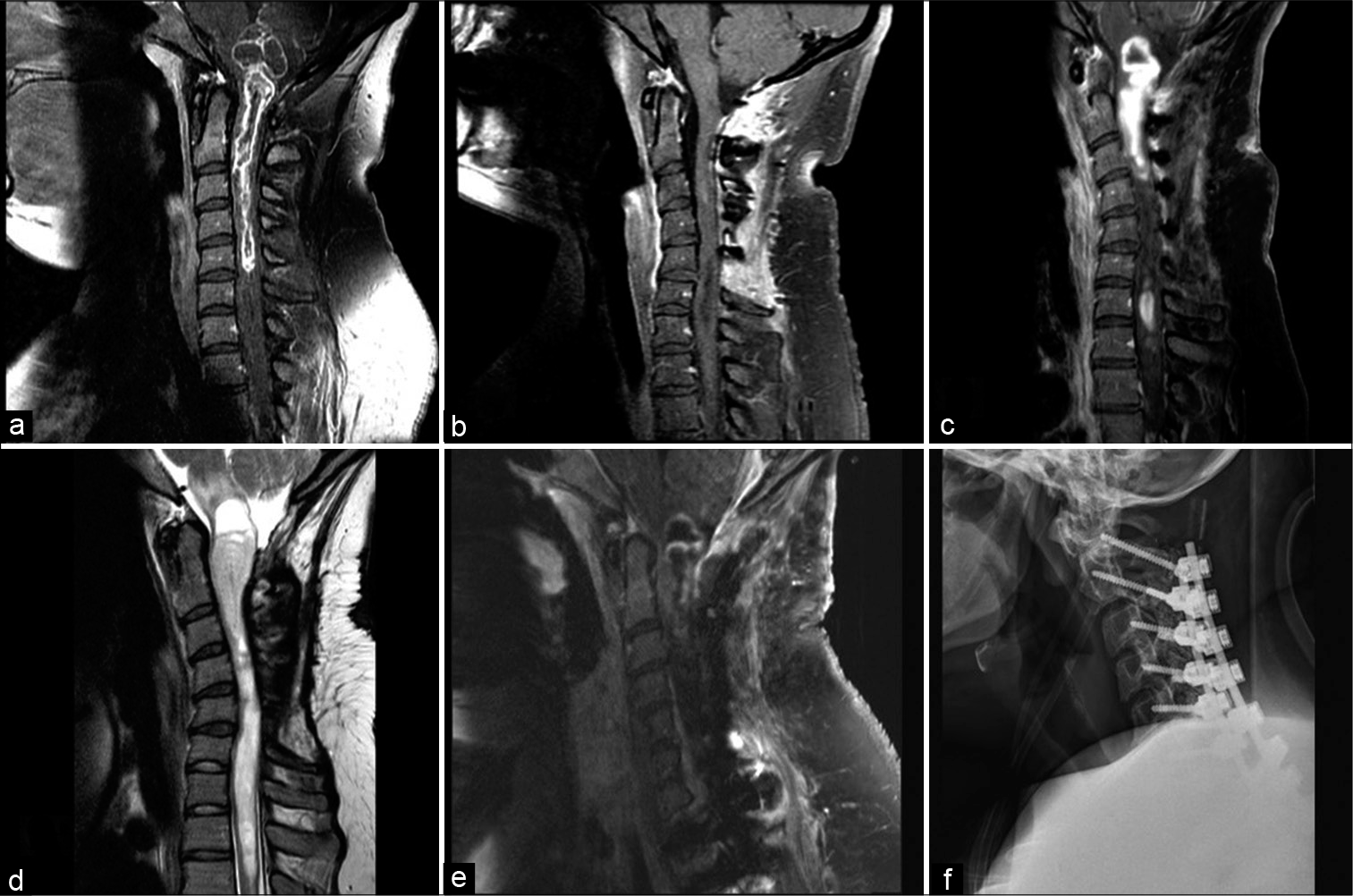
A woman with a progressive left hemiparesis and diffuse paresthesias presented for outpatient neurosurgical evaluation. Magnetic resonance imaging (MRI) of the cervical spine revealed a peripherally enhancing intramedullary lesion spanning from the medulla to C5 [
Figure 1:
(a) Sagittal MRI T1 sequence after gadolinium administration showing a peripherally enhancing intramedullary spinal mass spanning from the brainstem to C5. (b) Postoperative MRI after the initial resection showing a sagittal T1 sequence after gadolinium administration and minimal residual disease at the level of C3. (c) Sagittal MRI after gadolinium administration showing significant recurrence of the tumor with diffuse enhancement and cyst formation compressing the brainstem, within less than a year after her first surgery. (d) Sagittal MRI T2 sequence showing diffuse intramedullary edema spanning from the brainstem to the thoracic T2 segment, and worsening swan-neck deformity. (e) Sagittal MRI T1 sequence with gadolinium administration after the second tumor resection surgery showing decompression of the cyst with minimal peripheral residual enhancing mass. (f) Lateral postoperative X-ray of the cervical spine showing posterolateral instrumentation with restitution of lordosis.
After delivery, she noted progressively worsening weakness in her left hemibody. Of note, this weakness progressed more rapidly compared to her initial presentation. She underwent an MRI of the cervical spine, which revealed extensive disease recurrence and a new large cystic component of the tumor that compressed her medulla [
Her postoperative MRI revealed decreased tumor size, medullary decompression, and improved cervical alignment [
DISCUSSION
Case reports implicate pregnancy in the rapid transformation of gliomas.[
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Alifieris C, Trafalis DT. Glioblastoma multiforme: Pathogenesis and treatment. Pharmacol Ther. 2015. 152: 63-82
2. Fahey CC, Davis IJ. SETting the stage for cancer development: SETD2 and the consequences of lost methylation. Cold Spring Harb Perspect Med. 2017. 7: a026468
3. McDaniel SL, Strahl BD. Shaping the cellular landscape with Set2/SETD2 methylation. Cell Mol Life Sci. 2017. 74: 3317-34
4. Rosen A, Anderson V, Bercovici E, Laperriere N, D’Souza R. Brainstem gliomas in pregnancy: A systematic review†. J Matern Fetal Neonatal Med. 2020. 33: 2841-51
5. Schmidt BT, Hanna A. Deadly proliferation and transformation of pilocytic astrocytoma in pregnancy. World Neurosurg. 2020. 133: 99-103
6. Singh D, Chan JM, Zoppoli P, Niola F, Sullivan R, Castano A. Transforming fusions of FGFR and TACC genes in human glioblastoma. Science. 2012. 337: 1231-5