- Department of Neurological Surgery, Harborview Medical Center, UW Medicine, Seattle, Washington, USA
- Department of Neurosurgery, Dell Children's Medical Center, Austin, Texas, USA
- Department of Rehabilitation Medicine, Seattle Children's Hospital, Seattle, Washington, USA
- Department of Neurosurgery, Seattle Children's Hospital, Seattle, Washington, USA
Correspondence Address:
Samuel R. Browd
Department of Neurosurgery, Seattle Children's Hospital, Seattle, Washington, USA
DOI:10.4103/2152-7806.196236
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Brian W. Hanak, Luke Tomycz, Robert G. Oxford, Erin Hooper, Susan D. Apkon, Samuel R. Browd. An algorithmic approach to the management of unrecognized hydrocephalus in pediatric candidates for intrathecal baclofen pump implantation. 20-Dec-2016;7:105
How to cite this URL: Brian W. Hanak, Luke Tomycz, Robert G. Oxford, Erin Hooper, Susan D. Apkon, Samuel R. Browd. An algorithmic approach to the management of unrecognized hydrocephalus in pediatric candidates for intrathecal baclofen pump implantation. 20-Dec-2016;7:105. Available from: http://surgicalneurologyint.com/surgicalint_articles/an-algorithmic-approach-to-the-management-of-unrecognized-hydrocephalus-in-pediatric-candidates-for-intrathecal-baclofen-pump-implantation/
Abstract
Background:Complications of intrathecal baclofen (ITB) pump implantation for treatment of pediatric patients with spasticity and dystonia associated with cerebral palsy remain unacceptably high. To address the concern that some patients may have underlying arrested hydrocephalus, which is difficult to detect clinically because of a low baseline level of neurological function, and may contribute to the high rates of postoperative cerebrospinal fluid leak, wound breakdown, and infection associated with ITB pump implantation, the authors implemented a standardized protocol including mandatory cranial imaging and assessment of intracranial pressure (ICP) by lumbar puncture prior to ITB pump implantation.
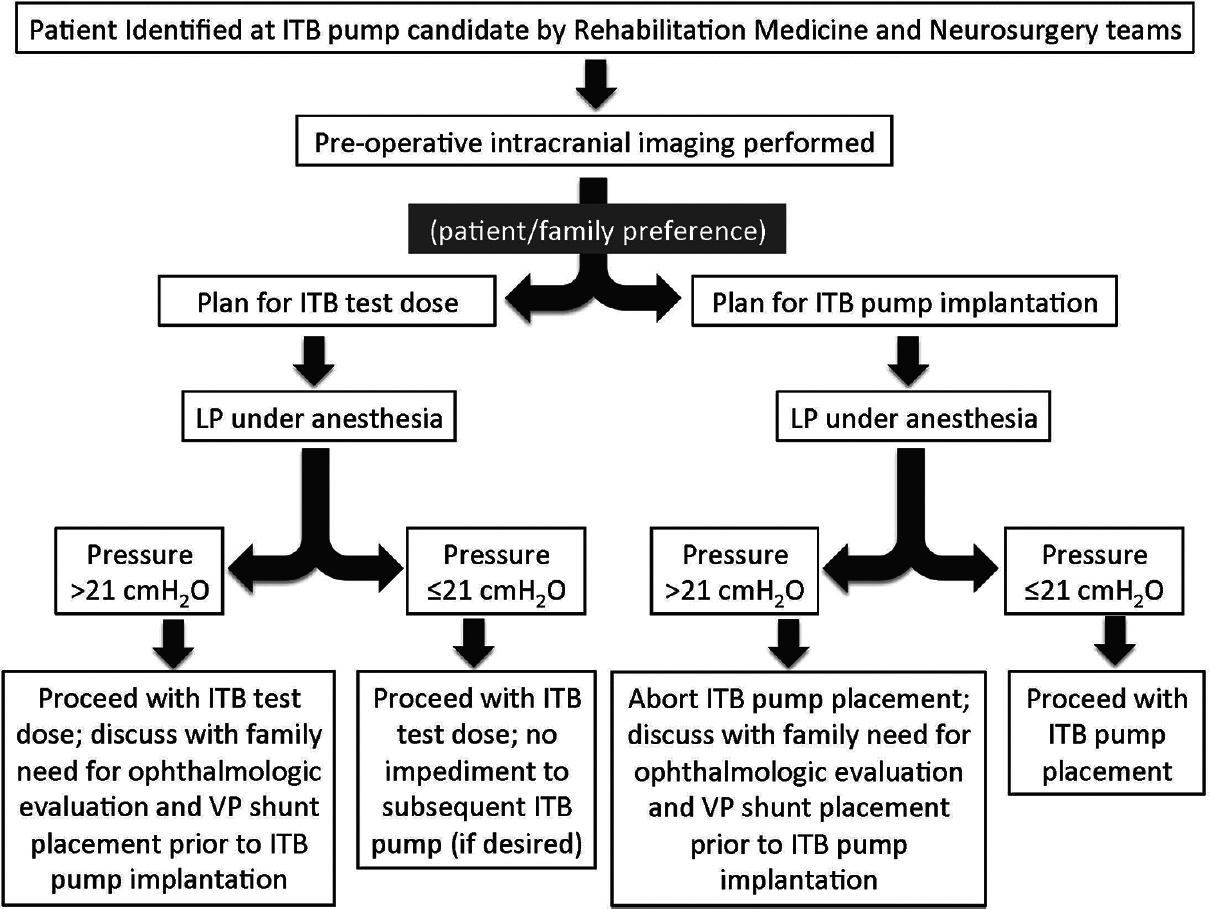
Methods:A retrospective case series of patients considered for ITB pump implantation between September 2012 and October 2014 at Seattle Children's Hospital is presented. All patients underwent lumbar puncture under general anesthesia prior to ITB pump implantation and, if the opening pressure was greater than 21 cmH2O, ITB pump implantation was aborted and alternative management options were presented to the patient's family.
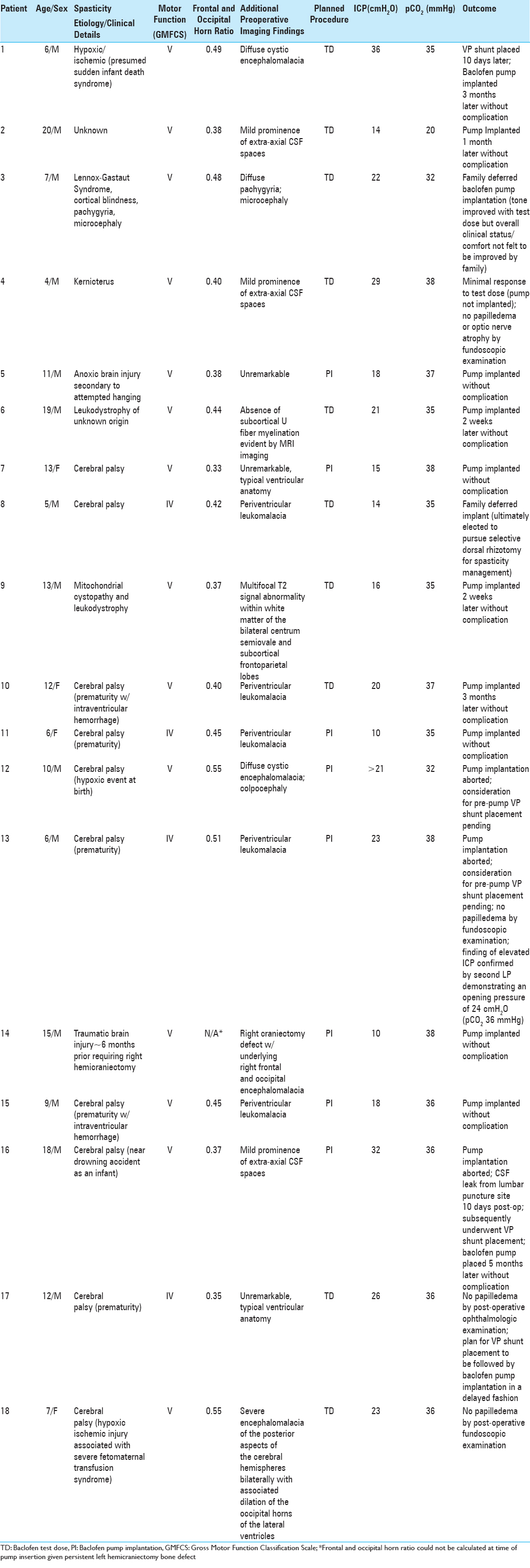
Results:Eighteen patients were treated during the study time period. Eight patients (44.4%) who had ICPs in excess of 21 cmH2O on initial LP were identified. Eleven patients (61.1%) ultimately underwent ITB pump implantation (9/10 in the “normal ICP” group and 2/8 in the “elevated ICP” group following ventriculoperitoneal shunt placement), without any postoperative complications.
Conclusions:Given the potentially high rate of elevated ICP and arrested hydrocephalus, the authors advocate pre-implantation assessment of ICP under controlled conditions and a thoughtful consideration of the neurosurgical management options for patients with elevated ICP.
Keywords: Arrested hydrocephalus, cerebral palsy, CSF leak, intrathecal baclofen pump, hydrocephalus, spasticity
INTRODUCTION
Intrathecal baclofen (ITB) delivery is one of the most common and effective treatments for spasticity and dystonia associated with cerebral palsy; however, complications of ITB pump implantation continue to be unacceptably high, with some reports citing a combined risk of infection and malfunction exceeding 40%.[
In many patients with cerebral palsy and spastic quadriparesis, arrested hydrocephalus may exist concurrently but may be difficult to clinically detect because of many factors including, in some, the limited ability to communicate symptoms associated with hydrocephalus. Albright et al. (2005) demonstrated elevated intracranial pressures (ICP; range: 22–41 cm H2O) by lumbar puncture (LP) in 23 of 24 (96%) children with cerebral palsy and ventriculomegaly.[
ILLUSTRATIVE CASE
Prior to establishing a formal protocol for screening of occult hydrocephalus, our center recorded rates of complications after pump implantations that were in line with published occurrence rates. One such case illustrates the need for ICP interrogation prior to intrathecal pump implantation. A 17-year-old male patient with a history of hypoxic ischemic injury with resultant spastic dystonic quadriplegia and severe developmental delay functioning at the Gross Motor Function Classification System (GMFCS) level V was recommended for baclofen pump implantation for ease-of-care considerations by the Rehabilitation Medicine team. No intraoperative complications were encountered at the time of baclofen pump implantation. The catheter was placed through a paraspinal transfascial approach. ICP was not measured at the time of pump implantation. In the postoperative period, the patient developed a tense pseudomeningocele at the operative site that ultimately resulted in wound breakdown and a frank CSF leak. Approximately 2 weeks after the initial surgery, a wound revision was performed. After exploring the patient's lumbar pseudomeningocele, we observed CSF egress around the intrathecal catheter. A purse-string suture was placed around the intrathecal catheter that effectively eliminated intraoperative evidence of the CSF leak. Following this surgery, there was temporary improvement in the appearance of the patient's lumbar wound, however, over the following 2 weeks, the patient's lumbar pseudomeningocele recurred and, again, a frank CSF leak was noted. A second, more extensive wound revision was performed at that time in an attempt to definitely address the CSF leak; this procedure consisted of an additional catheter site purse-string suture, a blood patch, and local paraspinal muscle advancement flaps. Unfortunately, the CSF leak persisted, and a decision was made to remove the patient's baclofen pump system in its entirety 5 days later. Upon removal of the lumbar catheter, ICP was measured via lumbar puncture and found to be 38 cmH2O (pCO2 Of 32 mmHg). No prior history of hydrocephalus was noted in review of the patient's history. In a delayed fashion, the patient underwent placement of a right frontal ventriculoperitoneal (VP) shunt. Three months after VP shunt placement, the patient again underwent baclofen pump insertion. Intraoperative measurement of ICP at the time of pump insertion was 8 cmH2O (pCO2 Of 38 mmHg) in the setting of a working VP shunt. After baclofen pump reinsertion, no postoperative pseudomeningocele and no wound healing concerns developed. The patient's family noted a dramatic improvement in the patient's tone, with appropriate titration of the patient's intrathecal baclofen. Of note, the patient's family did not note any improvement in functional status or cognition after VP shunt placement; the patient remained nonverbal and dependent for eating, dressing, and mobility, as was the case at his preoperative baseline.
MATERIALS AND METHODS
The case series presented is a retrospective review of patients considered for ITB pump implantation between September 2012 and October 2014 at the Seattle Children's Hospital. Patients were deemed candidates for ITB pump implantation after being seen by both the Rehabilitation Medicine and Neurosurgery teams. Head computed tomography (CT) or brain magnetic resonance imaging (MRI) was performed on all patients preoperatively to evaluate ventricular size and configuration. Frontal and occipital horn ratios were calculated as a means of estimating ventricular size.[
RESULTS
Twenty patients were considered for ITB pump implantation during the period of the study. Of these, 18 patients ranging in age from 4 to 20 years met the criteria for inclusion in the study [
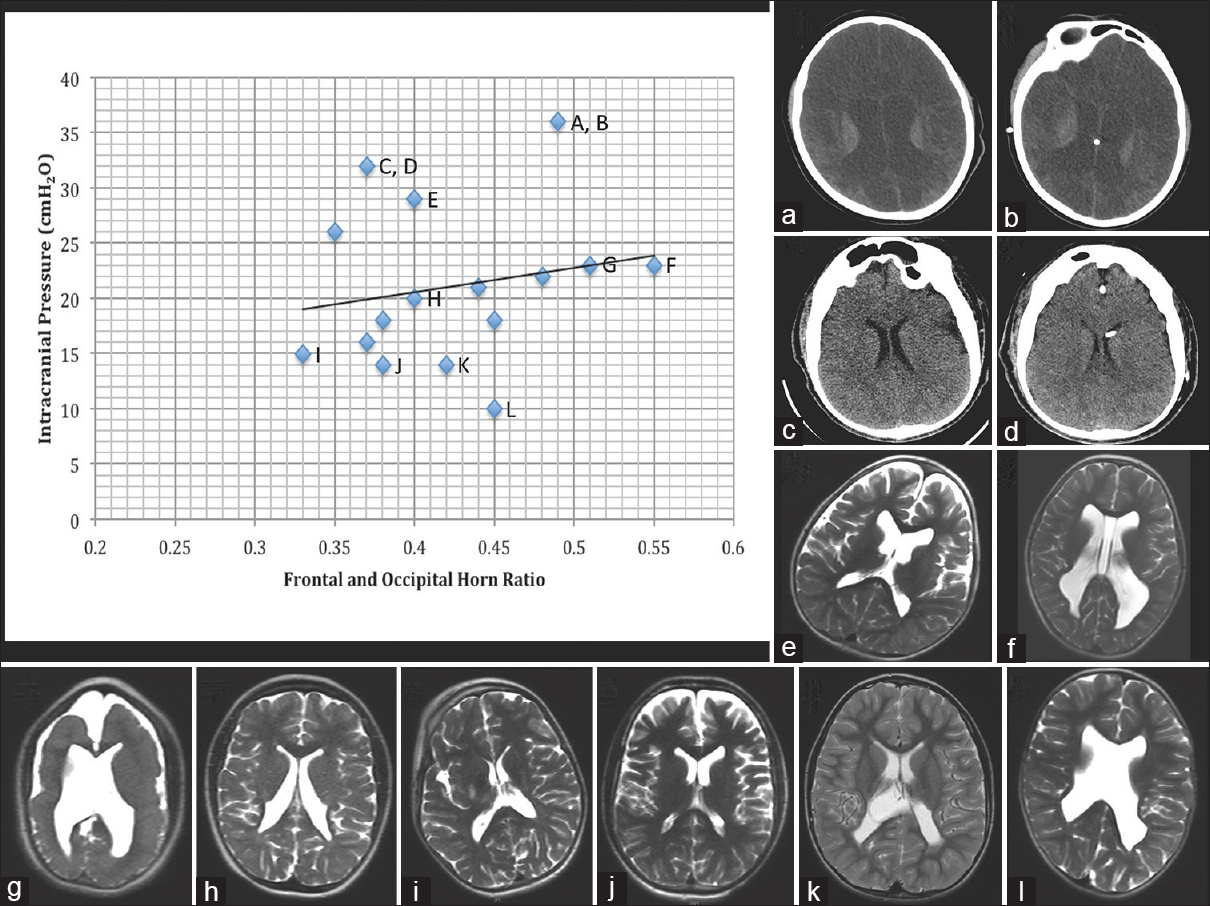
Among the 18 patients included in the study population, 8 (44.4%) had an ICP in excess of 21 cmH2O on initial LP. Nine of the 10 patients with ICP ≤21 cmH2O underwent ITB pump, implantation and there were no complications in this group. The family of the one patient (#8) in the “normal ICP” group who did not undergo ITB pump implantation ultimately decided to pursue an alternative procedure (selective dorsal rhizotomy) for spasticity management. One patient (#13) who was found to have an elevated ICP of 23 cmH2O by LP prior to a planned ITB pump insertion subsequently underwent a second LP to confirm the finding and, on repeat procedure, was found to have an ICP of 24 cmH2O. There was a mild positive correlation between preoperatively measured frontal and occipital horn ratios on cranial imaging and measured opening pressures on sedated LP (R2 =0.038) [
Figure 2
Scatter plot of preoperative frontal and occipital horn ratios and measured opening pressures on sedated lumbar puncture (LP) with selected imaging. Only absolute intracranial pressure (ICP) values were plotted; therefore, patient #12 (who had a recorded opening pressure of >21 cmH2O) was not included in the plot or linear regression analysis. In addition, patient #14 was not included in the plot or linear regression analysis because his hemicraniectomy bone defect precluded reliable measurement of a frontal and occipital horn ratio. The line of best fit is y = 22.19x + 11.674; R2= 0.038. Letters next to select scatter plot points correspond to the imaging studies presented in panels a-l. (a, b) Pre- and post-ventriculoperitoneal (VP) shunt placement noncontrast head computed tomographies (CTs), respectively, for patient #1. (c, d) Pre- and post-VP shunt placement noncontrast head CTs, respectively, for patient #16. In both patients who underwent VP shunt placement, a modest reduction in ventricular volume was noted after shunting. The remaining panels demonstrate preoperative T2-weighted magnetic resonance imagings (MRIs) for patients #4 (e), #18 (f), #13 (g), #10 (h), #7 (i), #2 (j), #8 (k), and #11 (l). Note the heterogeneity of ventricular anatomy. The presence of periventricular leukomalacia is common in the cerebral palsy patient population; it is particularly pronounced in patients #13 (g) and #11 (l). Significant variability is also noted with respect to the volume of extra-axial cerebrospinal fluid (CSF) spaces, with generous extra-axial CSF seen in cases with elevated (#4, e), borderline (#13, g), and normal (#2, j) CSF opening pressures
Two of the 8 patients in the “elevated ICP” group (#1, #16) ultimately underwent ITB pump implantation and, in both cases, VP shunt placement was performed prior to ITB pump implantation. Patient #16 required VP shunt placement to manage a CSF leak from the LP site created at the time of the LP. In a 3-week follow-up after shunt placement, there was no difference in the patient's cognition compared with his baseline. Five months after VP shunt placement, the patient underwent placement of an ITB pump without complication. In patient #1, a 6-year-old boy with a seizure disorder and diffuse cystic encephalomalacia on imaging, a test dose LP revealed an opening pressure of 36 cmH2O, and placement of a VP shunt was offered. After VP shunt placement, the patient demonstrated a significant reduction in seizure frequency, with a decrease from 10–15 seizures per day to 4–5 seizures per day. However, no cognitive or functional improvement was noted after shunt placement. The remaining 6 patients with elevated ICP either decided to forgo baclofen pump placement and proceed with ophthalmologic evaluation and possible repeat LP at a later date or opted for a referral to another provider for consideration of implantation using standard implantation guidelines.
DISCUSSION
Intrathecal baclofen delivery has become an effective treatment for spasticity, however, it continues to be associated with a high risk of complications and morbidity.[
In this series, 8/18 (44.4%) pediatric patients with spasticity and dystonia being considered for ITB pump implantation had ICPs in excess of 21 cmH2O as assessed by LP. While alarming in isolation, the observed rate of occult hydrocephalus in our series is actually considerably lower than the 96% rate of occult hydrocephalus observed by Albright et al. in their 2005 study of 24 pediatric patients with cerebral palsy.[
Patients with cerebral palsy may harbor arrested (i.e., nonprogressive) hydrocephalus; however, because of the frequent presence of developmental delay and the inability of the child to communicate symptoms, it is not a commonly pursued diagnosis in this patient population. Unfortunately, impairments in language cognition make it difficult to detect underlying symptoms from elevated ICP in many patients. The finding of occult hydrocephalus in patients who experienced wound breakdown with CSF leak after ITB pump implantation prompted a change in our institutional protocol such that each patient is now screened preoperatively with a cranial imaging study as well as a LP under general anesthesia for assessment of opening pressure.
In our series, we noted a trend toward increased ICP by LP evaluation and frontal and occipital horn ratios; however, this correlation was quite weak. The lack of a significant correlation between ventricular size and measured ICP is consistent with Albright et al., who found no correlation in this regard.[
ITB pumps confer significant benefits to patients suffering from spasticity, however, surgeons implanting these devices continue to search for ways to limit the historically high rates of associated complications. In this small series of patients, we have mitigated the potential for development of postoperative pseudomeningoceles and CSF leaks since implementing protocol presented. We believe that the assessment of ICP may help identify an especially high-risk cohort of patients for whom ITB pump implantation would likely lead to wound breakdown from CSF leak.[
We provide clarity to families that our protocol is not a standard practice currently in the neurosurgical management and assessment of baclofen pump patients, and we offer alternative options to families that wish to consider implantation regardless of ICP measurements. Given the potentially high rate of elevated ICP in the pediatric cerebral palsy population, we advocate for pre-implantation assessment of ICP under controlled conditions and a thoughtful consideration of the neurosurgical management options for patients with elevated ICP. Our protocol reflects our belief that elevated ICP is a risk factor for CSF leak following not just ITB pump implantation, but, any neurosurgical procedure performed that involves durotomy/dural puncture. Therefore, when LP reveals a marked elevation in intracranial pressure we feel an ethical obligation to convey this finding to the family before proceeding with the elective procedure of pump implantation. We fully expect that the institutional protocol presented in this manuscript will be viewed as somewhat controversial and we hope that it inspires debate. We certainly acknowledge that the threshold of ICP >21 cmH2O used in our current clinical algorithm is rather arbitrary and that ICP elevations above this threshold may be tolerated by many patients without wound breakdown or resulting complications. Future work is needed to confirm that patients with elevated ICP are at significantly increased risk of postoperative complications following ITB pump implantation and should seek to identify an ICP threshold above which there is a significantly higher procedural complication rate. Given that ITB pump implantation can result in dramatic improvements in the quality of life for cerebral palsy patients, continued efforts are needed to make this complication fraught procedure safer.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Albright AL, Ferson S, Carlos S. Occult hydrocephalus in children with cerebral palsy. Neurosurgery. 2005. 56: 93-6
2. Albright AL, Gilmartin R, Swift D, Krach LE, Ivanhoe CB, McLaughlin JF. Long-term intrathecal baclofen therapy for severe spasticity of cerebral origin. J Neurosurg. 2003. 98: 291-5
3. Anderson R, Connolly E, McKhann G, Huang J, Choudhri T, Komotar R, Mocco JIntrathecal Baclofen Therapy. Fundamentals of Operative Techniques in Neurosurgery. New York: Thieme; 2010. p.
4. Borowski A, Littleton AG, Borkhuu B, Presedo A, Shah S, Dabney KW. Complications of intrathecal baclofen pump therapy in pediatric patients. J Pediatr Orthop. 2010. 30: 76-81
5. Campbell WM, Ferrel A, McLaughlin JF, Grant GA, Loeser JD, Graubert C. Long-term safety and efficacy of continuous intrathecal baclofen. Dev Med Child Neurol. 2002. 44: 660-5
6. Ghosh D, Mainali G, Khera J, Luciano M. Complications of intrathecal baclofen pumps in children: Experience from a tertiary care center. Pediatr Neurosurg. 2013. 49: 138-44
7. Hoving MA, van Raak EP, Spincemaille GH, Palmans LJ, Becher JG, Vles JS. Efficacy of intrathecal baclofen therapy in children with intractable spastic cerebral palsy: A randomised controlled trial. Eur J Paediatr Neurol. 2009. 13: 240-6
8. Kulkarni AV, Drake JM, Armstrong DC, Dirks PB. Measurement of ventricular size: Reliability of the frontal and occipital horn ratio compared to subjective assessment. Pediatr Neurosurg. 1999. 31: 65-70
9. Sponheim S, Skraastad O, Helseth E, Due-Tonnesen B, Aamodt G, Breivik H. Effects of 0.5 and 1.0 MAC isoflurane, sevoflurane and desflurane on intracranial and cerebral perfusion pressures in children. Acta Anaesthesiol Scand. 2003. 47: 932-8