- Department of Neurosurgery, Memfys Hospital, University of Nigeria Teaching Hospital, Enugu, Nigeria.
- Department Neurology, Memfys Hospital, University of Nigeria Teaching Hospital, Enugu, Nigeria.
- Department of Medicine, University of Nigeria Teaching Hospital, Enugu, Nigeria.
Correspondence Address:
Francis Chukwuebuka Campbell, Department of Neurosurgery, Memfys Hospital, Enugu, Nigeria.
DOI:10.25259/SNI_636_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Francis Chukwuebuka Campbell1, Bibiana Oti2, Ned Michael Ndafia1, Idawarifagha Hart1, Okwuoma Okwunodulu1, Chika Anele Ndubuisi1, Birinus Adikaibe Ezeala-Adikaibe2,3, Samuel Chukwunonyerem Ohaegbulam1. Balo’s concentric sclerosis a rare variant of multiple sclerosis in a Nigerian adult male: A case report. 28-Oct-2022;13:486
How to cite this URL: Francis Chukwuebuka Campbell1, Bibiana Oti2, Ned Michael Ndafia1, Idawarifagha Hart1, Okwuoma Okwunodulu1, Chika Anele Ndubuisi1, Birinus Adikaibe Ezeala-Adikaibe2,3, Samuel Chukwunonyerem Ohaegbulam1. Balo’s concentric sclerosis a rare variant of multiple sclerosis in a Nigerian adult male: A case report. 28-Oct-2022;13:486. Available from: https://surgicalneurologyint.com/surgicalint-articles/11968/
Abstract
Background: Balo’s concentric sclerosis (BCS) is a rare variant of multiple sclerosis (MS) that usually runs a benign course. Typically, brain magnetic resonance imaging (MRI) shows concentric lamella of areas of demyelination alternating with normal zones of myelin preservation. Clinically, it may be mistaken for other intracranial space-occupying lesions, especially in our subregion, where neuroimaging facilities are scarce or not affordable. The authors report a rare case of BCS in a young Nigerian male diagnosed and managed in Enugu, Nigeria. This is possibly the first reported case in sub-Saharan Africa.
Case Description: The patient presented with behavioral changes, focal seizures, cranial neuropathy, long tract signs, and cerebellar dysfunction causing severe disability and functional impairment with a Modified Rankin Score (mRS) of 4. Brain MRI showed multiple heterogeneous lesions in the right cerebral hemisphere, brainstem, and cerebellum with features in keeping with BCS. Cerebrospinal fluid oligoclonal bands were negative and C-reactive protein was normal. He had high-dose steroids and immunosuppressive therapy. He made a gradual neurological improvement and significant symptom resolution and was able to return to work after a year (mRS-1).
Conclusion: BCS is a rare variant of MS in sub-Saharan Africa. The mainstay of diagnosis is MRI which shows the typical concentric demyelinating pattern. Steroids and immunosuppressive therapy are effective in the treatment of BCS.
Keywords: Balo’s concentric sclerosis, Demyelination, Intracranial space-occupying lesion, Multiple sclerosis
INTRODUCTION
Balo’s concentric sclerosis (BCS) is a rare neuroinflammatory disease characterized by concentric rings of white matter demyelination. It is a variant of multiple sclerosis (MS) initially described by Joseph Balo, in 1928, which runs a benign course with a fairly good outcome when compared to other demyelinating lesions.[
Brain magnetic resonance imaging (MRI) is the diagnostic investigation of choice. A collaborative report in the literature confirmed the characteristic concentric lamella of demyelination alternating with normal zones of myelin preservation seen in both FLAIR, T1- and T2-weighted images which give the lesion an onion peel appearance.[
The authors report a rare case of BCS in a young Nigerian male diagnosed and managed in Enugu Nigeria. This is probably the first reported case in sub-Saharan Africa.
CASE DESCRIPTION
The patient is a 31-year-old man who presented with a history of recurrent throbbing headaches of 10 weeks duration (Visual analog score 7-8/10). There were associated focal seizures involving the left upper limb which resolved with the administration of antiepileptic medications, personality changes characterized predominately by aggressive and irrational behavior as well as gradual onset but progressive weakness of the left side of the body.
Examination revealed that he was fully conscious with dysarthria. He had left facial nerve palsy upper motor neuron type, but vision, hearing, and other lower cranial nerves were normal. He also had left-sided spastic hemiparesis with a power of 3 to 4-5 (medical research council scale) and a significant left hemisensory deficit. The gait was grossly ataxic with associated recurrent falls predominately to the left. His Modified Rankin Score (mRS) was 4 at presentation. He had a course of low-dose oral steroids in the referral hospital before presentation to our center without any improvement.
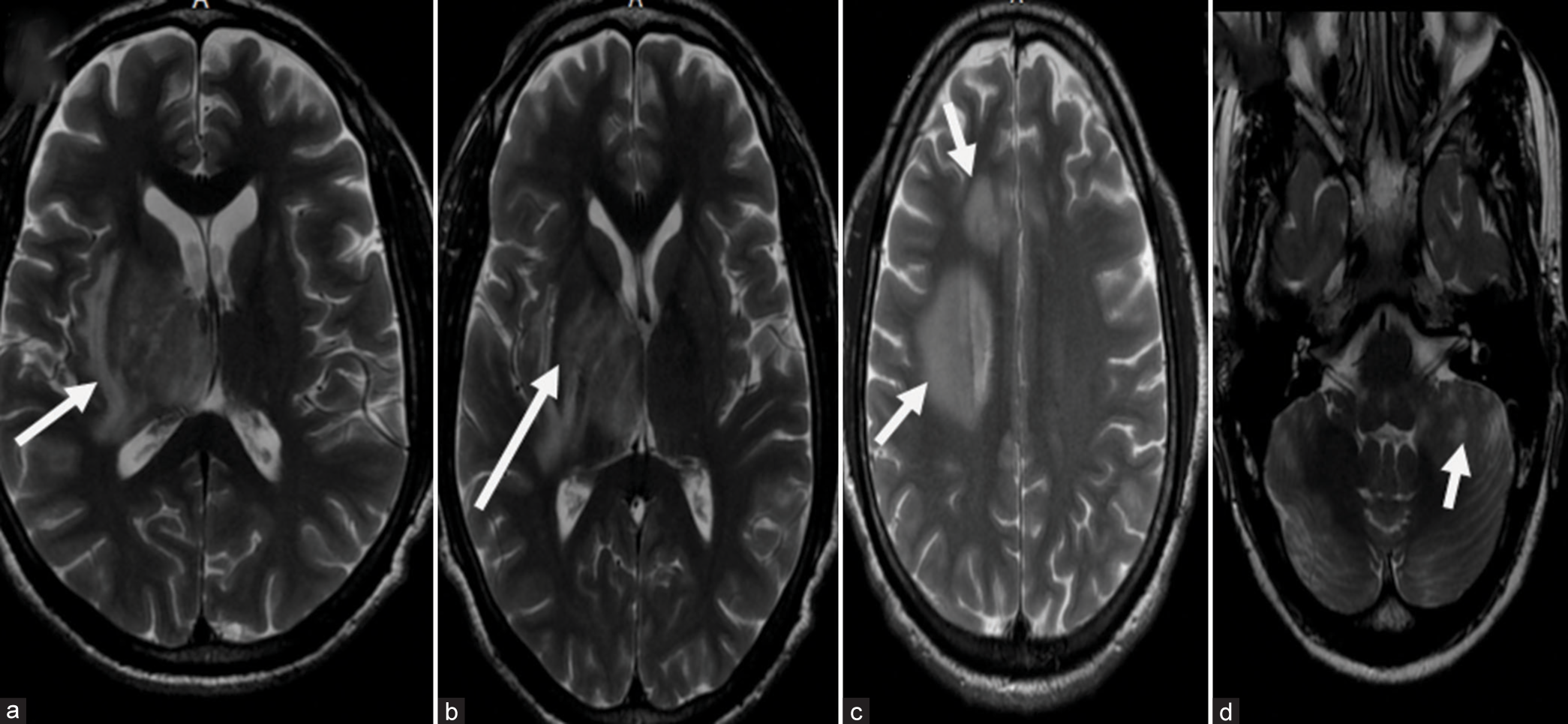
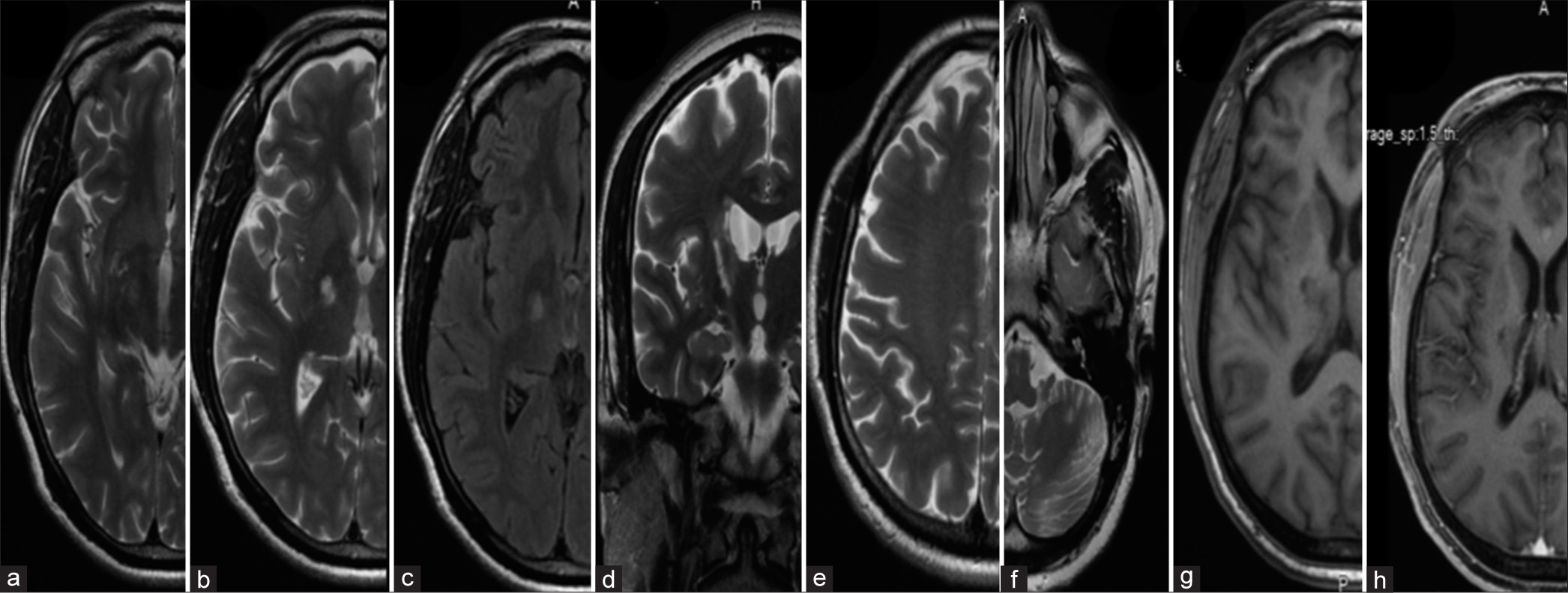
Brain MRI showed multiple heterogeneous lesions in the right cerebral hemisphere, brainstem, and cerebellum. The largest lesion was in the right capsuloganglionic area extending into the deep white matter and displaying alternating isointense and hypointense concentric rings/ bars on T1WI [
Figure 1:
Brain magnetic resonance imaging (MRI) of the patient at presentation (a-d) axial cuts of T2-weighted image of brain MRI of index patient showing multiple hyperintense lesions largest in the right capsuloganglionic area and adjacent white matter displaying concentric rings/bars (a-c). (d) Shows a similar hyperintense lesion in the left cerebellar hemisphere (white arrow depicts the lesion).
Cerebrospinal fluid (CSF) oligoclonal bands were negative, and CSF chemistry and cell count were normal. CSF culture was negative; however, the cytology showed fluffy cotton wool exudates in keeping with the presence of a possible degenerative or neuroinflammatory lesion. CRP was normal (<2.50 mg/dl.)
He was treated with a combination of steroids and immunosuppressive agents. The first course was high-dose steroids (intravenous methylprednisolone 1 g daily for 5 days followed by a maintenance dose of oral prednisolone). He made remarkable improvement after treatment with resolution of the headaches, speech deficits, and hemibody weakness. He had significant improvement in his personality; however, the gait and coordination problems persisted causing some limitations in his ability to carry out activities of daily living (mRS of 2).
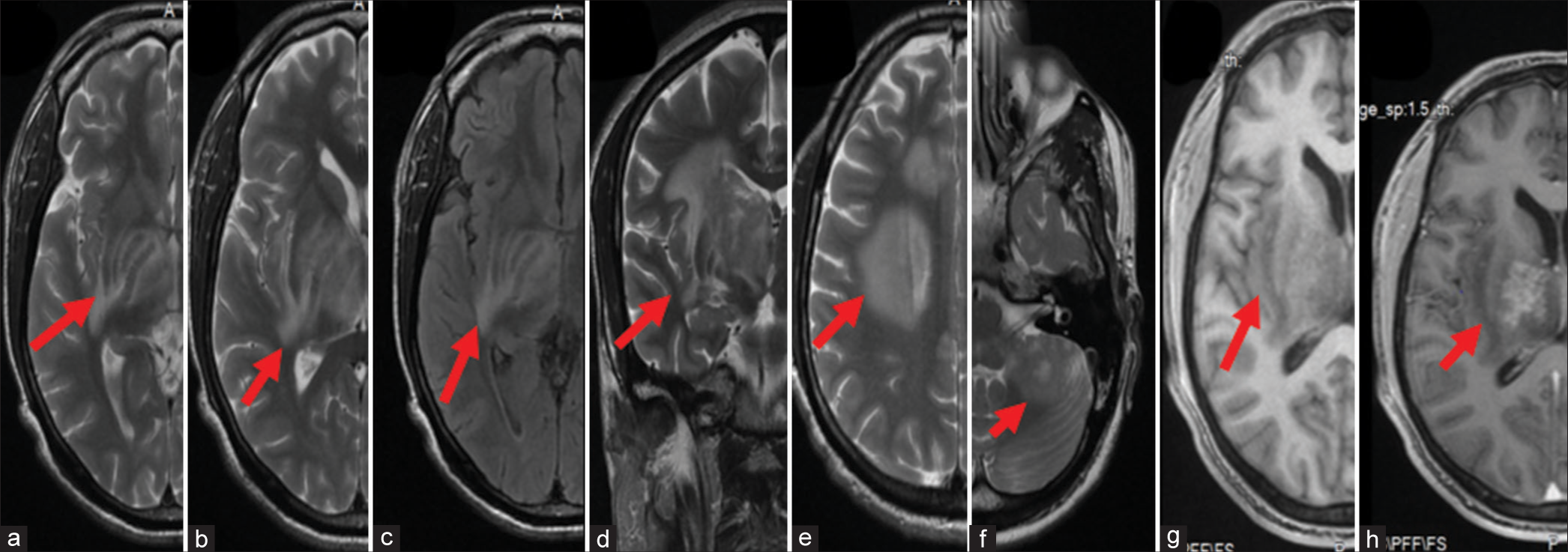
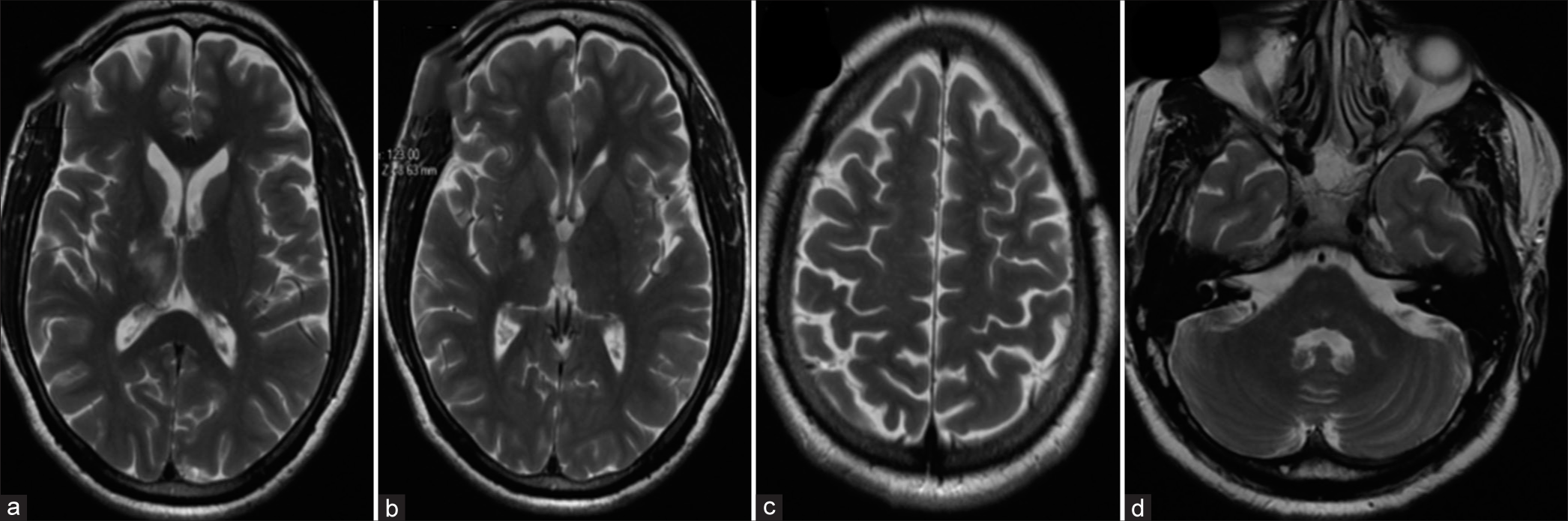
Five months after the first course of steroid therapy, he had progressive worsening of speech, gait, and limb weakness that necessitated a repeat course of treatment and maintenance with oral azathioprine. He made a gradual but progressive improvement and almost complete resolution of the speech deficits, hemibody weakness, gait, and coordination problems for 2–3 months. Brain MRI repeated 2 months after the second course showed almost complete resolution of the lesions with no associated edema or contrast enhancement [
He had regular physical therapy and rehabilitation and was able to return to work with mRS of 1 at 18 months follow-up.
CASE DISCUSSION
BCS is described as a variant of MS and usually presents with single or multiple concentric tumefactive-like lesions which causes widespread neurologic dysfunction.[
The MRI striking features of BCS are reminiscent of histopathology comparison of this rare demyelinating disease. These characteristic MRI imaging findings from the high tesla images allow antemortem diagnosis of BCS when performed at the onset of the disease. This helps to solve the diagnostic dilemma associated with this disease which before now was made postmortem or following biopsy of a suspected ICSOL.
Our index patient had behavioral changes, cranial neuropathy, long tract signs, and cerebellar dysfunction that significantly affected his quality of life and MRI features showing multiple demyelinating plaques in the subcortical, deep white matter, and the cerebellum with features in keeping with BCS. On treatment with high-dose steroids, he showed remarkable clinical response with an improvement of mRS from 4 to 1 and almost complete disappearance of the lesions on repeat brain MRI [
The cause of the neuroinflammation in BCS is not clear, but it is believed that unknown toxins or substances are released from the perivenular zone at the geometric center of the lesion and spread outward.[
Although the usual concentric lamella of demyelinating plaques alternating with areas of myelin preservation is the predominant findings of BCS, other studies have noted varied presentation patterns on MRI including distorted rings, rosettes, carnation pattern, and even parallel and rectangular bars.[
About half of BCS coexists with MS and this may explain the varying expression of oligoclonal bands in CSF of patients with BCS.[
Although BCS runs a benign course, it is essential to counsel patients about the risk of recurrence which may occur anytime during care and follow-up. This may arise from a preexisting lesion(s) or denovo from previously uninvolved sites typically disseminated in space and time as observed in MS.[
BCS, although rare, requires a high index of suspicion during clinical and neuroimaging evaluation of patients suspected to have multiple intracranial lesions. Resource-constrained regions with limited MRI facilities may miss this potential variant of MS that runs a rather benign course resulting in a diagnostic dilemma, unnecessary biopsies, and delay to institute appropriate management.
CONCLUSION
BCS, a rare variant of MS, exists in sub-Saharan Africa. The mainstay of diagnosis is MRI which shows the typical concentric demyelinating pattern in FLAIR, T1-, and T2-sequences. Its course is often benign but can present with multiple recurrences and relapses. Steroids and immunosuppressive agents are often effective in the management.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Amini Harandi AA, Esfandani A, Pakdaman H, Abbasi M, Sahraian MA. Balo’s concentric sclerosis: An update and comprehensive literature review. Rev Neurosci. 2018. 29: 873-82
2. Balo J. Encephalitis periaxialis concentrica. Arch Neur Psych. 1928. 19: 242-64
3. Caracciolo JT, Murtagh RD, Rojiani AM, Murtagh FR. Pathognomonic MR imaging findings in Balo concentric sclerosis. AJNR Am J Neuroradiol. 2001. 22: 292-3
4. Dobson R, Giovannoni G. Multiple sclerosis a review. Eur J Neurol. 2019. 26: 27-40
5. Hardy TA, Miller DH. Baló’s concentric sclerosis. Lancet Neurol. 2014. 13: 740-6
6. Hardy TA, Tobin WO, Lucchinetti CF. Exploring the overlap between multiple sclerosis, tumefactive demyelination and Baló’s concentric sclerosis. Mult Scler. 2016. 22: 986-92
7. Karaarslan E, Altintas A, Senol U, Yeni N, Dincer A, Bayindir C. Balo’s concentric sclerosis: Clinical and radiologic features of five cases. AJNR Am J Neuroradiol. 2001. 22: 1362-7
8. Kastrup O, Stude P, Limmroth V. Balo’s concentric sclerosis. Evolution of active demyelination demonstrated by serial contrast-enhanced MRI. J Neurol. 2002. 249: 811-4
9. Kira J. Astrocytopathy in Balo’s disease. Mult Scler. 2011. 17: 771-9
10. Ndubuisi CA, Ndukuba KO, Okwuoma O, Iroegbu LU, Ohaegbulam SC. Dominant hemisphere and upper cervical cord tumefactive multiple sclerosis in a Nigerian teenager initially misdiagnosed and managed. Orient J Med. 2019. 31: 61-6
11. Poser CM, Vernant JC. La sclérose en plaques dans les populations noires Multiple sclerosis in the black population. Bull Soc Pathol Exot. 1993. 86 5 Pt 2: 428-32
12. Wang C, Zhang KN, Wu XM, Huang G, Xie XF, Qu XH. Balo’s disease showing benign clinical course and co-existence with multiple sclerosis-like lesions in Chinese. Mult Scler. 2008. 14: 418-24