- Department of Neurosurgery, Osaka Medical and Pharmaceutical University, Takatsuki City, Osaka, Japan,
- Department of Infection Control and Prevention, Osaka Medical and Pharmaceutical University, Takatsuki City, Osaka, Japan.
Correspondence Address:
Ryokichi Yagi, Department of Neurosurgery, Osaka Medical and Pharmaceutical University, Takatsuki City, Osaka, Japan.
DOI:10.25259/SNI_78_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ryokichi Yagi1, Yukimasa Ooi2, Naosuke Nonoguchi1, Masahiko Wanibuchi1. Brain abscess caused by Nocardia thailandica infection in systemic lupus erythematosus patient with steroid therapy. 08-Apr-2022;13:126
How to cite this URL: Ryokichi Yagi1, Yukimasa Ooi2, Naosuke Nonoguchi1, Masahiko Wanibuchi1. Brain abscess caused by Nocardia thailandica infection in systemic lupus erythematosus patient with steroid therapy. 08-Apr-2022;13:126. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11525
Abstract
Background: Nocardia is an aerobic Gram-negative bacillus belonging to actinomycetes and has been reported to be an infectious disease in healthy individuals. However, more than 60% have some underlying illnesses and are said to be opportunistic infections.
Case Description: The case was a 69-year-old man who had been on long-term steroids for systemic lupus erythematosus. He developed with nausea and gait disturbance and was suspected of having a brain abscess by imaging. Abscess drainage detects actinomycetes that appear to belong to the genus Nocardia and brain abscess by Nocardia thailandica using matrix-assisted laser desorption/ionization time-of-flight mass analysis (MALDITOFMS) I was diagnosed. He died during the course of the treatment, and his organs did not detect his N. thailandica at necropsy, so he concluded that bacterial death from long-term administration of antibiotics was the cause of death.
Conclusion: N. thailandica is a very rare bacterium belonging to Nocardia asteroides and is said to easily form brain lesions. In immunocompromised patients, prophylaxis with antibiotics and detailed examination of lung lesions for surveillance were considered necessary. This paper is the first report of a brain abscess caused by N. thailandica, including a review of the literature.
Keywords: Brain abscess, Long-term steroid therapy, Nocardia thailandica, Nocardia, Systemic lupus erythematosus
INTRODUCTION
Opportunistic infections occur more frequently, or are more severe, in people with weakened immune systems (such as people with HIV); these can be life-threatening and cause severe complications in patients receiving long-term treatment with immunosuppressants and steroids. Nocardia, a naturally occurring actinomycete, cannot be detected by culture tests and, thus, requires genetic analysis. In human, almost 90% of Nocardia infections are caused by the Nocardia asteroides group. Nocardia thailandica is an extremely rare bacterium related to Nocardia asteroids. Of note, N. thailandica drug sensitivity is relatively good, and its standard treatments are β-lactam antibiotics, sulfamethoxazole/trimethoprim (ST) combination, tetracycline, and macrolides. To the best of our knowledge, this is the first clinical report of an N. thailandica associated brain abscess. In our case, after the diagnosis of an N. thailandica-associated brain abscess, the original disease was ameliorated by the administration of antibiotics; however, the patient died from microbial substitution caused by prolonged administration of antibiotics.
CASE REPORT
A 69-year-old male with systemic lupus erythematosus (SLE) presented with a chief complaint of nausea and gait disorder. The patient’s eldest daughter also had SLE. He had no smoking history, no drinking, no travel history, no outdoor activities, and no sexual activity.
The patient’s present medical history was as follows. The patient became aware of dyspnea and edema of the lower extremities during exercise; thus, he consulted a family doctor. The examination revealed hypertension, proteinuria, and hematuria. A chest X-ray revealed bilateral pleural effusion. Because of an increase in antinuclear antibodies (Anti-ds-DNA antibody 218.5) and a decline in complement (C3 51 and C4 12.3), a connective tissue disease was suspected, and the patient was referred to our Department of Rheumatic Collagen Disease. After 2 months, he was hospitalized for examination and diagnosed with SLE because of (i) renal lesions, (ii) leukopenia, (iii) positive anti-ds-DNA antibody, and (iv) positive antinuclear antibody. Based on the renal biopsy results 2 months later, he was diagnosed with lupus nephritis Class IV. Accordingly, steroid pulse therapy (methylprednisolone, 1000 mg) and tacrolimus (2 mg/day) were initiated intravenously. As SLE and lupus nephritis improved, the patient received prednisolone (25 mg/day) + tacrolimus (2 mg/day) + azathioprine (100 mg/day) orally and was discharged after 6 months. He returned for follow-up as an outpatient.
After 1 year, the patient visited a family doctor for nausea and dizziness. A brain computed tomography (CT) revealed a multilocular cystic lesion with edema. Thus, he was referred to our department for neurosurgery.
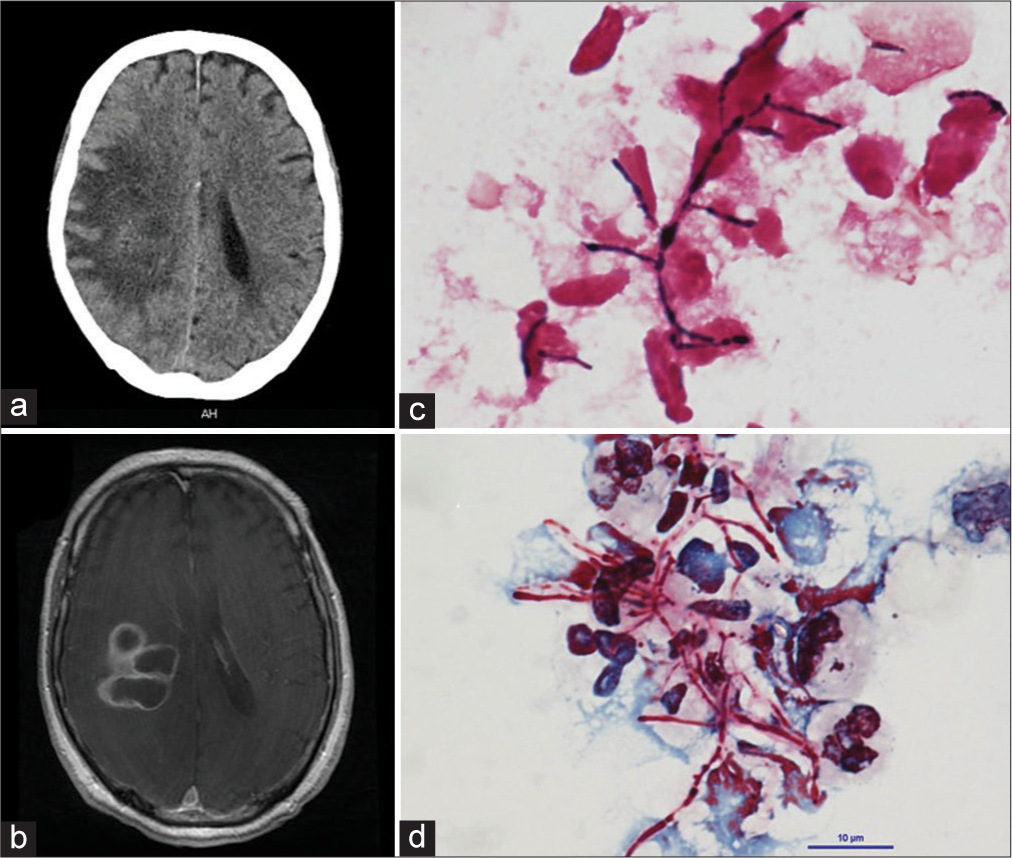
On day 1 of admission, the initial brain CT in the axial plane shows the low-density area in the right parietal lobe [
Figure 1:
(a) Preoperative the brain CT in the axial plane shows the low-density area in the right parietal lobe. (b) The enhanced T1-weighted MRI axial image revealed that polycystic ring-enhancing lesions with surrounding vasogenic edema in the right parietal lobe. (c) Gram stain for brain pus showing Gram-positive actinomyces with beaded staining pattern. (×1000). (d) Kinyoun smear stain showing a cluster of filamentous branching rods that are partially acid fast, which is characteristic for Nocardia (×1000).
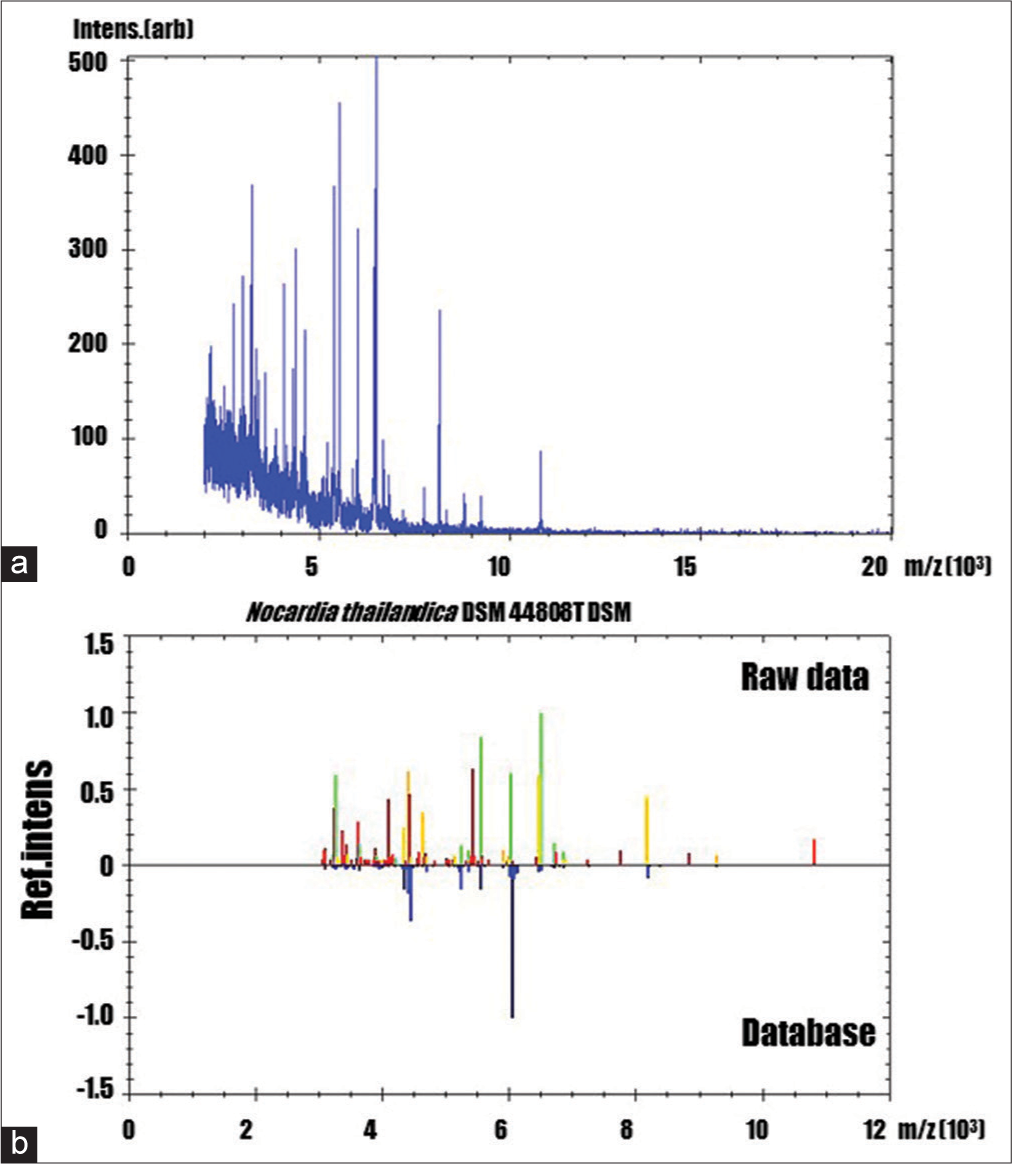
Figure 2:
(a) MALDI-TOF mass spectra of the extract from a patient’s abscess (b): Nocardia thailandica (identification score: 1.731) The comparison between the raw MALDI-TOF mass spectra and the most possibility reference mass spectra. Yellowish-green, yellowish, and reddish spectra indicated as correct, possible, and difference to the reference mass spectra of the database, respectively.
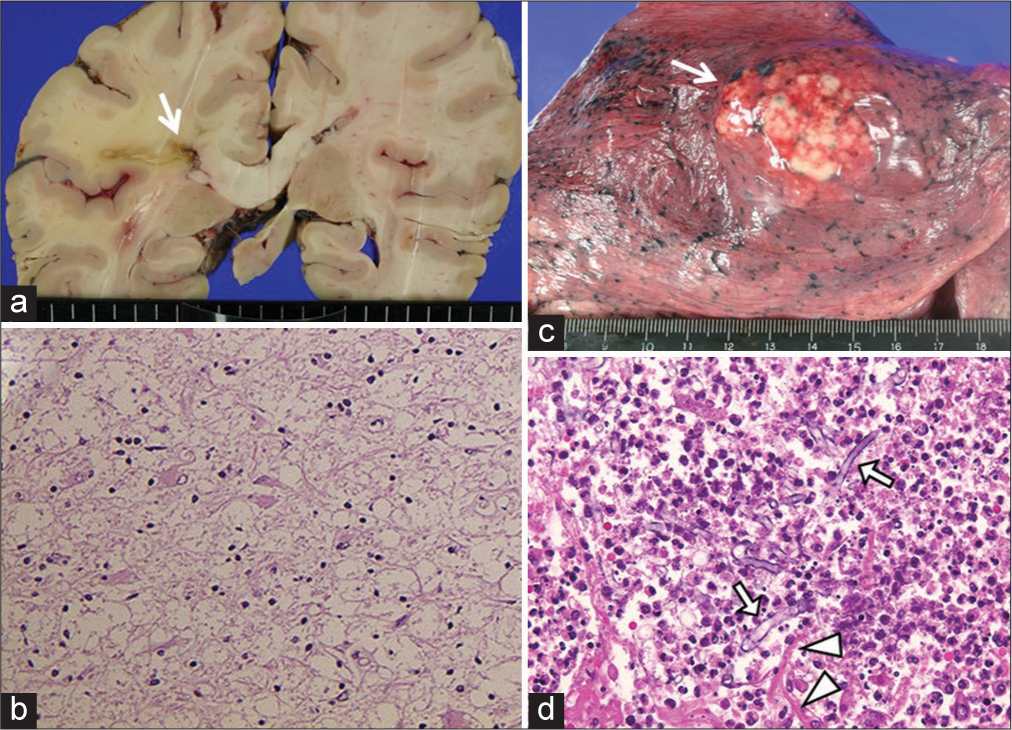
The patient remained hospitalized for 30 days without further deterioration of renal function; however, consciousness disorder appeared because of deteriorating central nervous system lupus. Consequently, oral administration became difficult; thus, we switched to ceftriaxone (CTRX; 2 g BID) intravenously. A brain CT revealed that the brain abscess had decreased gradually. The treatment of the abscess and SLE was progressing smoothly. However, on day 77 of admission, the patient developed a catheter-associated urinary tract infection and catheter-associated bloodstream infection due to Corynebacterium striatum and extended spectrum β-lactamase Escherichia coli. Gradually, the patient’s condition worsened and he died 86 days after admission. After death, a pathological autopsy was performed with the approval of the patient’s family. We established the formation of microabscesses in the lungs, mitral valve, liver, and kidneys; however, no Gram-positive bacilli suspected of being Nocardia were detected in the brain or other organs. Since filamentous fungi, suspected of Aspergillus, and yeast-like fungi, suspected of Candida, were detected in the abscesses and blood vessels, the cause of death was established as aspergillosis and disseminated candidiasis due to bacterial replacement caused by the prolonged administration of antibiotics [
Figure 3:
Postmortem specimens of the patient. (a) Gross brain section showing a collapsed abscess in the right parietal lobe (arrow). (b) H.E. stain revealed that no viable bacteria was in and around the cerebral abscess (×100). (c) Gross appearance of lung showing an abscess occupying the right inferior lobe (arrow). (d) Aspergillus (arrow) and Candida (arrow head) genus were seen in the lung abscess stained with H.E. (×100).
DISCUSSION
Nocardia is an aerobic Gram-negative bacillus belonging to the order actinomycetes and is found in soil, water, rotten plants, and animal excrement. Although infections in healthy individuals have been reported, more than 60% have some underlying diseases, and are said to be opportunistic infections that develop in an immunocompromised state with reduced cellular immunity.[
Since intestinal mucosal disorders occur in patients treated with steroids and antitumor drugs, it has been reported that Candida, which is a resident bacterium of the digestive tract and invades the body due to microbial substitution disease due to long-term antimicrobial administration.[
Opportunistic infections such as Nocardia are a serious complication for immunocompromised patients. In addition, long-term administration of antibacterial drugs in patients who need to continue administration of steroids and immunosuppressive drugs may cause other infections due to bacterial replacement phenomenon. Although it is of course possible to prevent opportunistic infections, early detection is important. We think that it is necessary to perform CT examinations regularly instead of simple X-ray examinations so as not to miss microscopic lesions 4. If any infection is suspected, antibiotics should be started early and, if possible, temporary discontinuation of steroids and immunosuppressants should be considered.
This case developed a brain abscess due to an opportunistic infection with N. thailandica during long-term steroid administration to SLE. This is the first case of a brain abscess of N. thailandica because we were able to identify N. thailandica using genetic analysis of the isolate. Although the patient was lost due to the bacterial replacement phenomenon associated with treatment, it is a valuable case to report.
CONCLUSION
To the best of our knowledge, this is the first case report of an N. thailandica-associated brain abscess. Although the patient died, the autopsy revealed that the cause of death was not the N. thailandica-associated brain abscess, but bacterial replacement due to the treatment of the brain abscess. Since opportunistic infections in immunocompromised patients with various underlying diseases are leading risk factors, prophylactic antimicrobial therapy before infection and regular chest CT screening could be beneficial for the patients’ prognosis.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
We are grateful to Dr. Shinji Kawabata and Motomasa Furuse for helpful discussions.
References
1. Beaman BL, Burnside J, Edwards B, Causey W. Nocardial infections in the United States, 1972-1974. J Infect Dis. 1976. 134: 286-9
2. Edwards U, Rogall T, Blocker H, Emde M, Bottger EC. Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res. 1989. 17: 7843-53
3. Jodlowski TZ, Melnychuk I, Conry J. Linezolid for the treatment of Nocardia spp. infections. Ann Pharmacother. 2007. 41: 1694-9
4. Kageyama A, Yazawa K, Ishikawa J, Hotta K, Nishimura K, Mikami Y. Nocardial infections in Japan from 1992 to 2001, including the first report of infection by Nocardia transvalensis. Eur J Epidemiol. 2004. 19: 383-9
5. Kageyama A, Yazawa K, Nishimura K, Mikami Y. Nocardia testaceus sp. Nov. and Nocardia senatus sp. Nov., isolated from patients in Japan. Microbiol Immunol. 2004. 48: 271-6
6. Kurahara Y, Tachibana K, Tsuyuguchi K, Akira M, Suzuki K, Hayashi S. Pulmonary nocardiosis: A clinical analysis of 59 cases. Respir Investig. 2014. 52: 160-6
7. Malincarne L, Marroni M, Farina C, Camanni G, Valente M, Belfiori B. Primary brain abscess with Nocardia farcinica in an immunocompetent patient. Clin Neurol Neurosurg. 2002. 104: 132-5
8. Mamelak AN, Obana WG, Flaherty JF, Rosenblum ML. Nocardial brain abscess: Treatment strategies and factors influencing outcome. Neurosurgery. 1994. 35: 622-31
9. McBride WJ.editors. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases 7th edition. Sex Health. 2010. 7: 218
10. Torack RM. Fungus infections associated with antibiotic and steroid therapy. Am J Med. 1957. 22: 872-82