- Department of Neurosurgery, Desert Regional Medical Center, Palm Springs, California, United States,
- Department of Neurology, University of Washington, Seattle, Washington, United States,
- Department of Neurosurgery, Redlands Community Hospital, Redlands, California, United States
Correspondence Address:
Brian Fiani
Department of Neurosurgery, Redlands Community Hospital, Redlands, California, United States
DOI:10.25259/SNI_946_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: James B. Fowler1, Brian Fiani1, Kasra Sarhadi2, Vladimir Cortez3. Cerebral fat embolism in the absence of a long bone fracture: A rare case report. 02-Mar-2021;12:78
How to cite this URL: James B. Fowler1, Brian Fiani1, Kasra Sarhadi2, Vladimir Cortez3. Cerebral fat embolism in the absence of a long bone fracture: A rare case report. 02-Mar-2021;12:78. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10621
Abstract
Background: The classic triad of fat embolism syndrome consists of pulmonary distress, mental status change, and petechial rash. Typically, symptoms manifest 24–48 hours after a long bone fracture, but case reports have demonstrated fat embolism syndrome without long bone fracture. These cases are initiated by a stress response, mobilizing free fatty acids into the circulation.
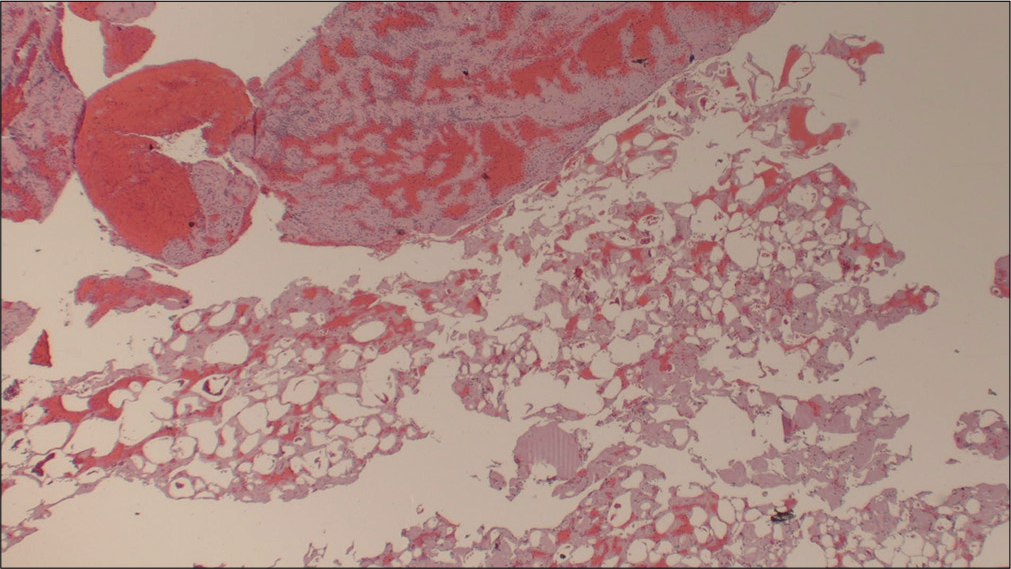
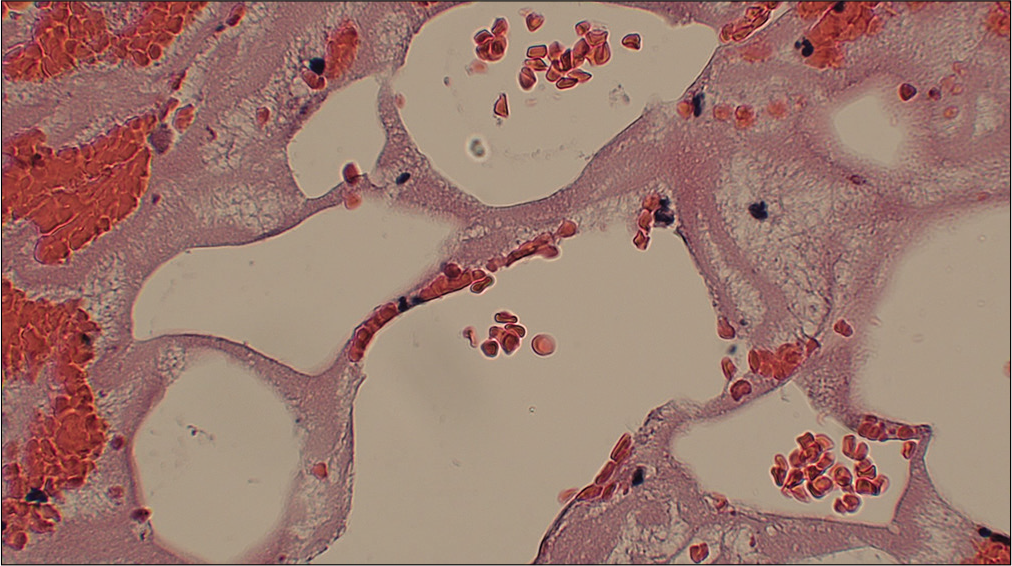
Case Description: Herein, we present the case of a 70-year-old male who presented with the left-sided hemiparesis and was subsequently found to have tandem lesions of the right internal carotid artery (ICA) and right middle cerebral artery (MCA) warranting emergent mechanical thrombectomy (MT). The ensuing pathology report determined the source of ischemic stroke to be caused by fat embolism, a rare and intriguing case of cryptogenic large vessel occlusion (LVO) with unique features distinguishing it from other reports in the literature.
Conclusion: According to the biochemical theory, a catecholamine surge can precipitate fat globules forming in the circulatory system, leading to tissue hypoxia, injury, and ischemia. While the majority of cerebral fat emboli cause reversible ischemia of small diameter vessels, our case presents with LVO and tandem lesions in both the ICA and MCA resulting in infarct and residual hemiparesis.
Keywords: Endovascular, Fat emboli, Interventional neurosurgery, Neurointervention, Thrombectomy
INTRODUCTION
Fat embolism, described in the literature as early as 1862, is characterized by the release of fat droplets into the systemic circulation, commonly as a complication of long bone fracture or following an orthopedic procedure. Less commonly, severe burns, infection, kidney transplant, liposuction, cardiopulmonary bypass, gastrectomy, and transfusions may result in fat emboli syndrome (FES).[
CLINICAL PRESENTATION
A 70-year-old male with hypertension presented to the emergency department after being involved in a motor vehicle collision in the early afternoon. He awoke that morning with the left-sided weakness and impaired coordination. He initially proceeded to carry out his daily routine, until the time of the accident. Emergency medical services at the scene noted left facial droop and left upper extremity weakness, without other apparent injury, and initiated a stroke code. His time last known well (LNW) was 14 h before presentation. Baseline modified Rankin scale (MRS) was 0.
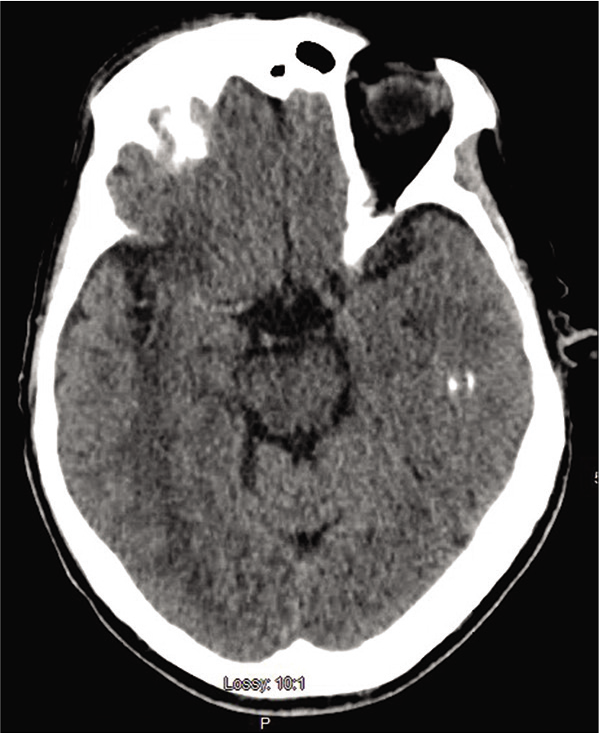
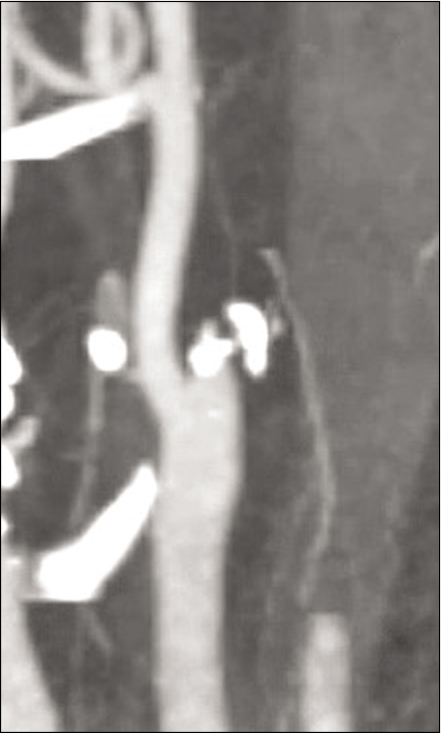
On arrival, blood pressure, heart rate, and oxygen saturation were within normal limits and National Institute of Health Stroke Scale (NIHSS) was calculated to be 4 for partial left facial droop and left upper extremity drift. No other injuries were identified. Computed tomography (CT) head without contrast demonstrated a hyperdense MCA [
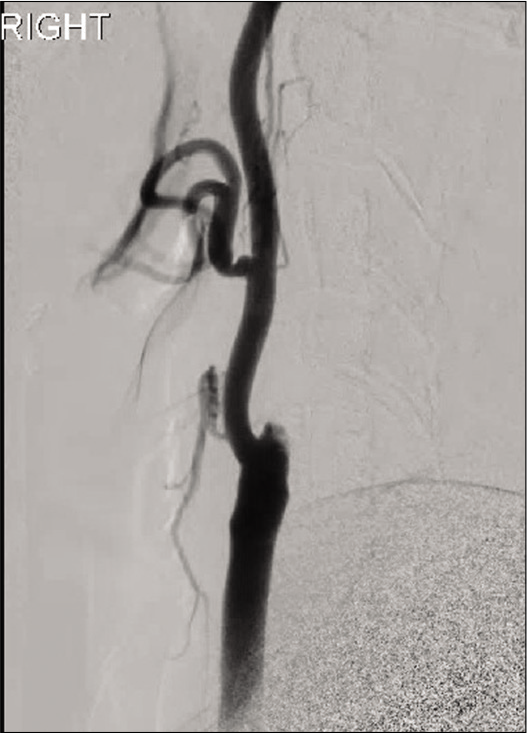
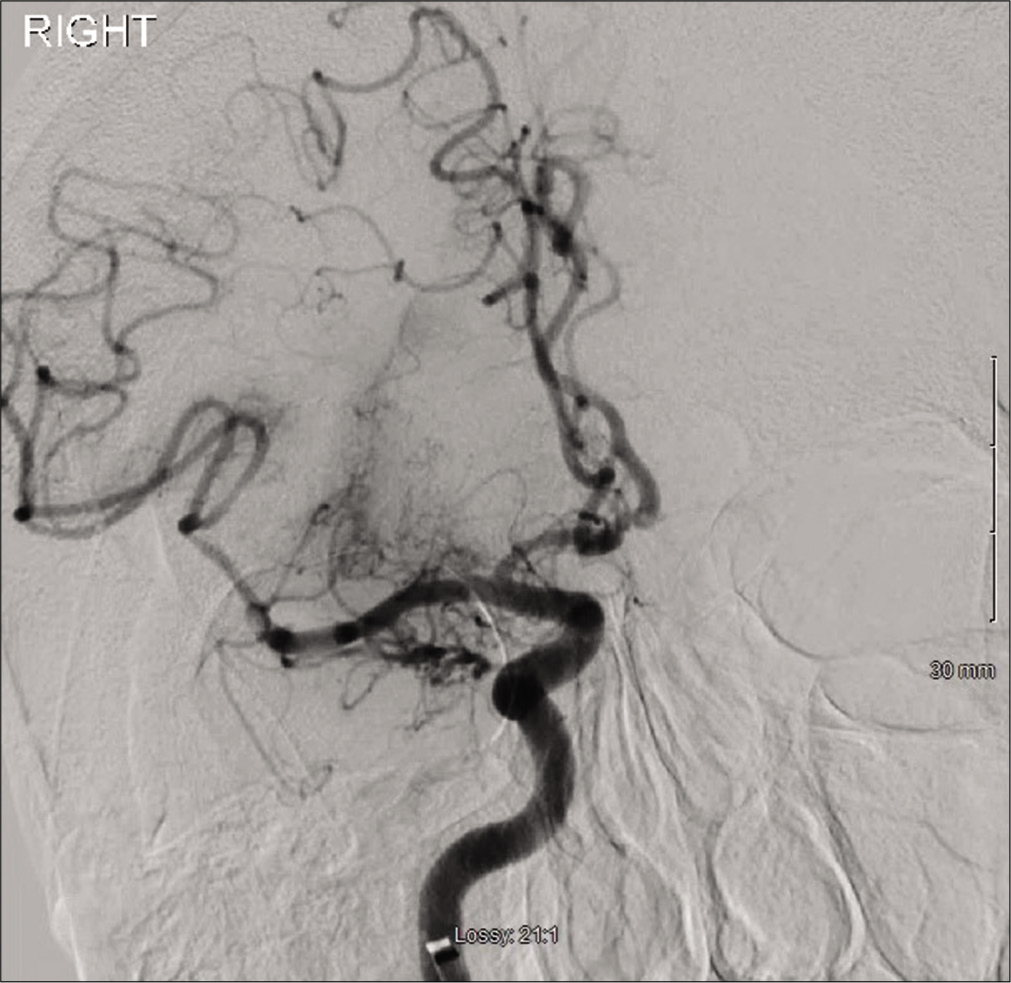
Intraoperatively, there was complete occlusion at the proximal right ICA [
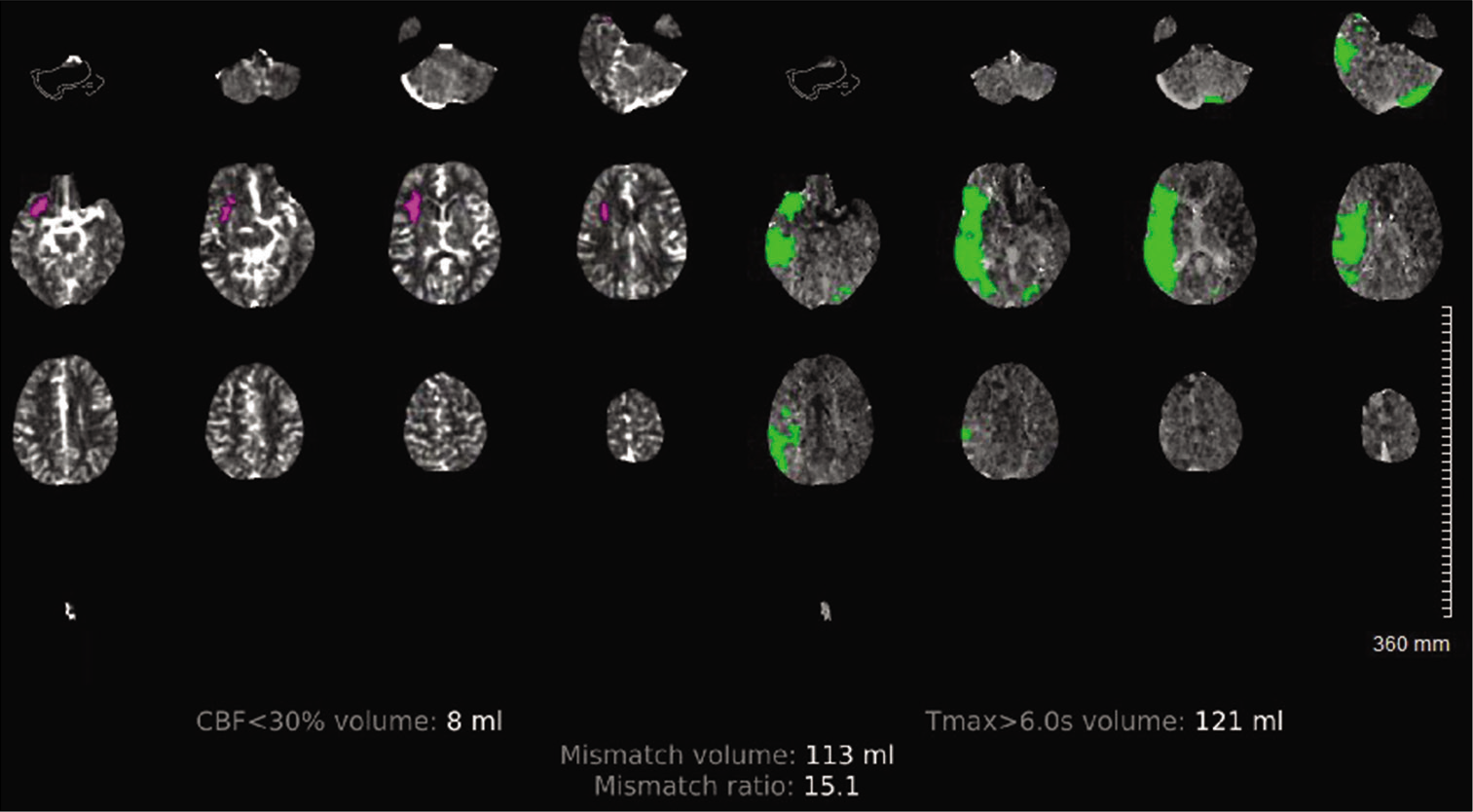
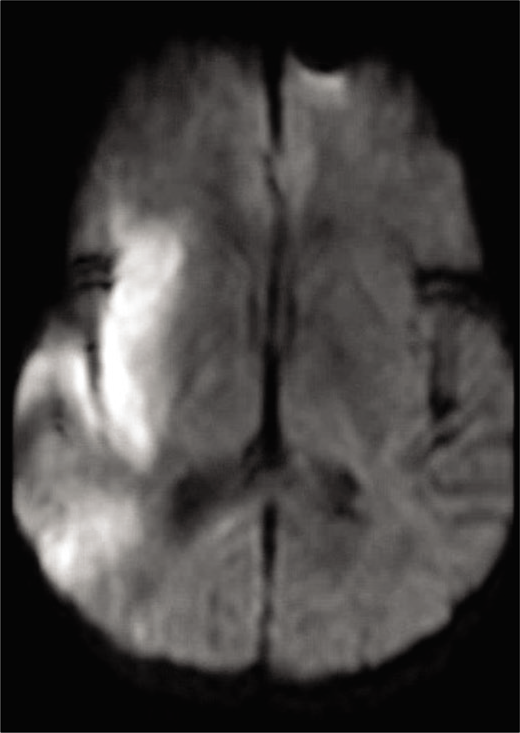
The patient was monitored in the intensive care unit after the procedure. Magnetic resonance imaging (MRI) of the brain on postprocedure day 1 showed right MCA territory infarct [
DISCUSSION
The case presented has several unique features that distinguish it from prior reports of FES described in the literature. While the classic triad of FES consists of pulmonary distress, mental status change, and petechial rash, a variety of symptoms (or lack thereof) may be present. Gurd and
Wilson described at least two major criteria or one major and four minor criteria to be presented to diagnose FES.[
Second, fat emboli typically affects small caliber vessels, while FES presenting as a LVO is exceedingly rare.[
In the case presented, the patient had no long bone fracture. While the majority FES reported in the literature is in association with long bone fracture, less common etiologies include severe burns, infection, kidney transplant, liposuction, cardiopulmonary bypass, gastrectomy, and transfusions.[
Figure 8:
Axial magnetic resonance imaging brain, DWI sequence postprocedure demonstrating hyperintensity in the right middle cerebral artery territory, primarily in the insular region, consistent with restricted diffusion. There is some scattered hyperintensity posteriorly consistent with “starfield” pattern.
Despite an overall mortality rate of 7.7–36%,[
CONCLUSION
Fat embolism syndrome typically presents with respiratory distress, mental status change, and petechial rash. While cerebral fat emboli in itself are not a clinical rarity, our case presents numerous unique aspects. Namely, our patient demonstrated isolated neurological manifestations in the absence of long bone fracture. According to the biochemical theory, a catecholamine surge can precipitate fat globules forming in the circulatory system, leading to tissue hypoxia, injury, and ischemia. While the majority of cerebral fat emboli cause reversible ischemia of small diameter vessels, our case presents with LVO and tandem lesions in both the ICA and MCA resulting in infarct and residual hemiparesis.
Declaration of patient consent
Patient’s consent was not required as the identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Allardyce DB, Meek RN, Woodruff B, Cassim MM, Ellis D. Increasing our knowledge of the pathogenesis of fat embolism: A prospective study of 43 patients with fractured femoral shafts. J Trauma. 1974. 14: 955-62
2. Baker PL, Pazell JA, Peltier LF. Free fatty acids, catecholamines, and arterial hypoxia in patients with fat embolism. J Trauma. 1971. 11: 1026-30
3. Bardana D, Rudan J, Cervenko F, Smith R. Fat embolism syndrome in a patient demonstrating only neurologic symptoms. Can J Surg. 1998. 41: 398-402
4. Chang RN, Kim JH, Lee H, Baik HJ, Chung RK, Kim CH. Cerebral fat embolism after bilateral total knee replacement arthroplasty-a case report. Korean J Anesthesiol. 2010. 59: S207-10
5. DeFroda SF, Klinge SA. Fat embolism syndrome with cerebral fat embolism associated with long-bone fracture. Am J Orthop (Belle Mead NJ). 2016. 45: E515-21
6. Erdem E, Namer IJ, Saribas O, Aras T, Tan E, Bekdik C. Cerebral fat embolism studied with MRI and SPECT. Neuroradiology. 1993. 35: 199-201
7. Gossling HR, Pellegrini VD. Fat embolism syndrome: A review of the pathophysiology and physiological basis of treatment. Clin Orthop Relat Res. 1982. 165: 68-82
8. Gregorakos L, Sakayianni K, Hroni D, Harizopoulou V, Markou N, Georgiadou F. Prolonged coma due to cerebral fat embolism: Report of two cases. J Accid Emerg Med. 2000. 17: 144-6
9. Gurd AR, Wilson RI. The fat embolism syndrome. J Bone Joint Surg Br. 1974. 56B: 408-16
10. Ibañez NC, Collado RN, Gaviña IS, de Medrano MG, Chicharro FM. Fat embolism syndrome without respiratory failure. Clin Intensive Care. 2011. 10: 101-3
11. Ihn YK, Baik JH. Cerebral fat embolism as a rare complication of postgastrectomy: Case report. J Korean Radiol Soc. 2004. 51: 591-4
12. Liu L, Yin M, Liu S, Hu M, Zhang B. Facial filler causes stroke after development of cerebral fat embolism. Lancet. 2020. 395: 449
13. Muller C, Rahn BA, Pfister U, Meinig RP. The incidence, pathogenesis, diagnosis, and treatment of fat embolism. Orthop Rev. 1994. 23: 107-17
14. Parchani A, Shaikh N, Bhat V, Kattren MA. Fat embolism syndrome: Clinical and imaging considerations: Case report and review of literature. Indian J Crit Care Med. 2008. 12: 32-6
15. Parizel PM, Demey HE, Veeckmans G, Verstreken F, Cras P, Jorens PG. Early diagnosis of cerebral fat embolism syndrome by diffusion-weighted MRI (starfield pattern). Stroke. 2001. 32: 2942-4
16. Peltier LF. Fat embolism. Clin Orthop Relat Res. 2004. 422: 148-53
17. Pinney SJ, Keating JF, Meek RN. Fat embolism syndrome in isolated femoral fractures: Does timing of nailing influence incidence?. Injury. 1998. 29: 131-3
18. Replogle RL. The nature of blood sludging, and its relationship to the pathophysiological mechanisms of trauma and shock. J Trauma. 1969. 9: 675-83
19. Robb HJ. Microembolism in the pathophysiology of shock. Angiology. 2016. 16: 405-11
20. Ryu CW, Lee DH, Kim TK, Kim SJ, Kim HS, Lee JH. Cerebral fat embolism: Diffusion-weighted magnetic resonance imaging findings. Acta Radiol. 2005. 46: 528-33
21. Satoh H, Kurisu K, Ohtani M, Arita K, Okabayashi S, Nakahara T. Cerebral fat embolism studied by magnetic resonance imaging, transcranial doppler sonography, and single photon emission computed tomography. J Trauma. 1997. 43: 345-8
22. Takahashi M, Suzuki R, Osakabe Y, Asai JI, Miyo T, Nagashima G. Magnetic resonance imaging findings in cerebral fat embolism. J Trauma. 1999. 46: 324-7
23. Tsai IT, Hsu CJ, Chen YH, Fong YC, Hsu HC, Tsai CH. Fat embolism syndrome in long bone fracture-clinical experience in a tertiary referral center in Taiwan. J Chin Med Assoc. 2010. 73: 407-10