- Tecnologico de Monterrey, Escuela de Medicina y Ciencias de la Salud, Monterrey, Mexico
- Department of Neurosurgery, National Institute of Neurology and Neurosurgery, Mexico City, Mexico
- Department of Neurosurgery, Hospital Ángeles Metropolitano, Mexico City, Mexico.
Correspondence Address:
Juan Luis Gomez Amador, Department of Neurosurgery, National Institute of Neurology and Neurosurgery, Mexico City, Mexico.
DOI:10.25259/SNI_915_2023
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Omar R. Ortega-Ruiz1, Jorge Armando Lara Olivas2, Marcos V. Sangrador-Deitos2, Ricardo Marian Magaña2, Jose Augusto Ruiz Gurria3, Juan Luis Gomez Amador2. Combined endoscopic transorbital and transnasal approach for the management of a solitary plasmacytoma of the sphenoid bone: A case report and literature review. 16-Feb-2024;15:45
How to cite this URL: Omar R. Ortega-Ruiz1, Jorge Armando Lara Olivas2, Marcos V. Sangrador-Deitos2, Ricardo Marian Magaña2, Jose Augusto Ruiz Gurria3, Juan Luis Gomez Amador2. Combined endoscopic transorbital and transnasal approach for the management of a solitary plasmacytoma of the sphenoid bone: A case report and literature review. 16-Feb-2024;15:45. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=12758
Abstract
Background: Parasellar plasmacytomas are rare neurosurgical entities. Intrinsic characteristics of these tumors, such as adjacent bone erosion and symptoms resulting from invasion and mass effect, may lead to the possibility of a solitary extramedullary plasmacytoma (SEP) as a differential diagnosis.
Case Description: We present the case of a 39-year-old male with a 1-month history of bilateral decreased visual acuity, retroocular pulsating pain, and chromatic vision loss. A computed tomography scan of the head revealed a parasellar lesion causing chiasmatic compression, as well as clival, orbital, sphenoidal, and ethmoidal invasion. A combined transorbital and endonasal endoscopic approach was found suitable, and gross total resection was achieved. Histological analysis of the lesion established the diagnosis of a SEP. After radiotherapy, a new magnetic resonance imaging was performed, revealing a recurrence of the lesion with a high grade of invasion. The patient was treated with palliative radiotherapy, as surgical resection did not seem feasible.
Conclusion: Surgical resection and radiotherapy may achieve remission of these lesions; however, recurrence rates remain high despite any treatment modality. Patients with this condition must be followed up with a multidisciplinary team due to the high risk of multiple myeloma progression.
Keywords: Endoscopic endonasal approach, Parasellar plasmacytoma, Solitary plasmacytoma, Transorbital endoscopic approach
INTRODUCTION
According to the International Myeloma Working Group, a solitary plasmacytoma (SP) can be classified as either a solitary extramedullary plasmacytoma (SEP) or an SP of the bone (SPB), in which a single mass of plasma cells with no evidence of bone marrow malignant transformation, is located in a single area of bone with no soft-tissue infiltration.[
CASE PRESENTATION
A 39-year-old male was admitted to the emergency department with a 1-month history of bilateral decreased visual acuity, intraocular pulsating pain, and loss of chromatic vision. Antibiotics and steroids were prescribed before his admission, causing symptom relief. However, recurrence after medication withdrawal prompted him to seek medical attention at our center.
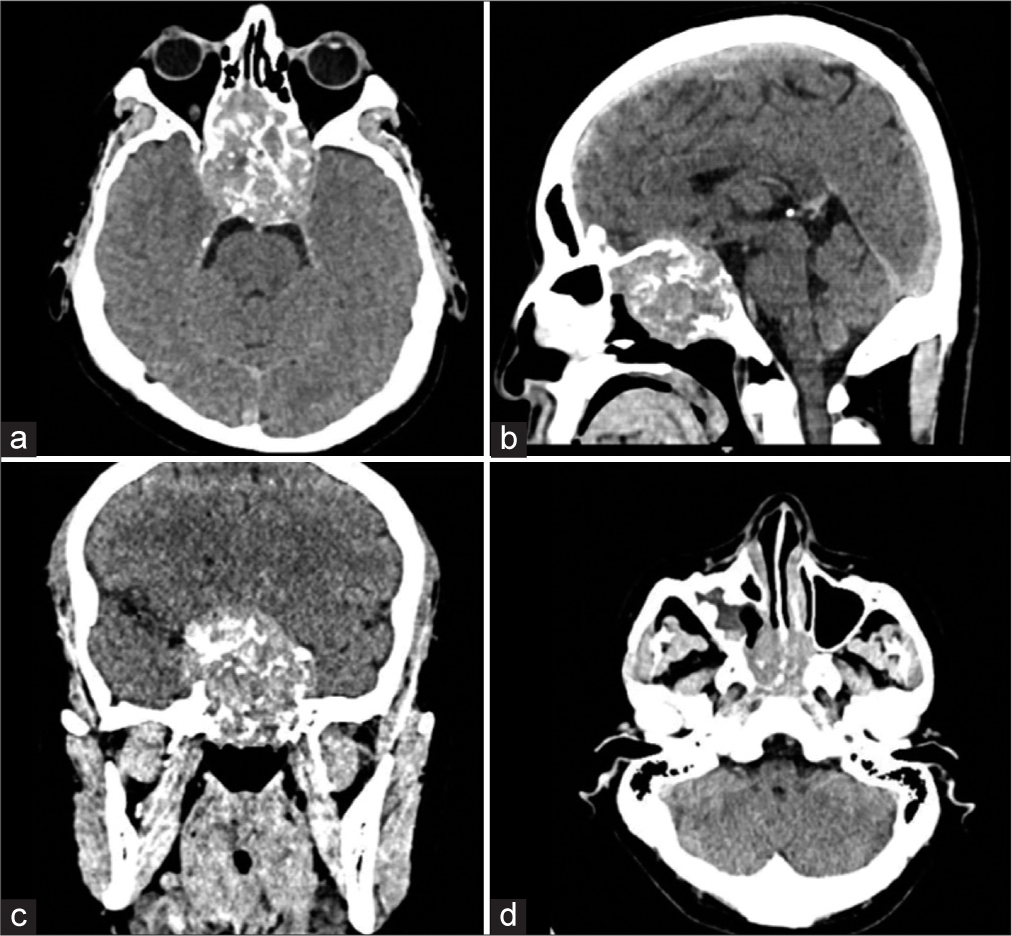
A mass in the nasal region with extension toward the right eye was found. On neurological examination, total blindness with loss of photomotor responses was found in the right eye, while vision in the left eye was preserved (20/40). An initial diagnosis of optic neuritis was made, and computed tomography (CT) scan revealed a 5.6 × 4.6 × 5.1 cm heterogeneous solid sellar lesion with chiasmatic compression, as well as clival, orbital, sphenoid, and ethmoid invasion. Protrusion into the nasal cavities and right maxillary sinus was also outlined [
TOA
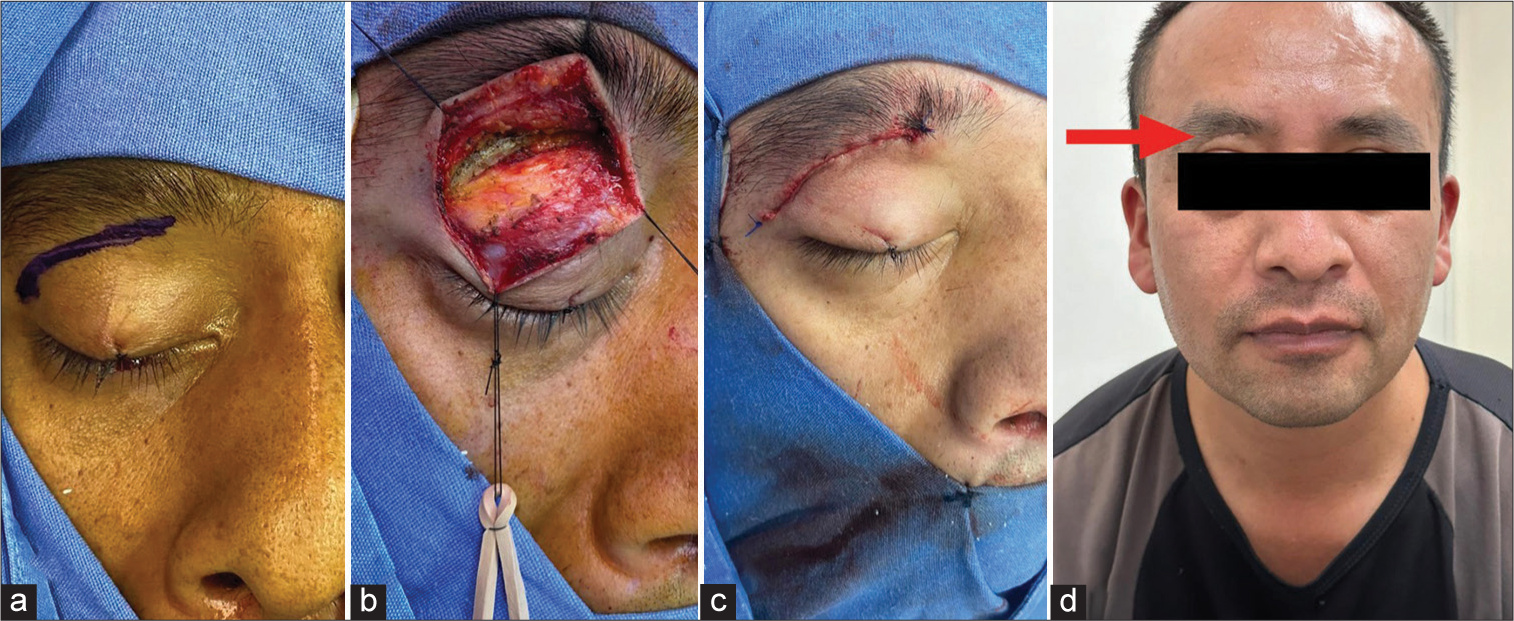
After conventional neuroanesthetic procedures, the patient was placed supine with the head in a neutral position. A superior eyelid crease incision was made, exposing the superolateral border of the orbital rim after the orbicularis muscle was incised. A subperiosteal dissection from the lateral margin of the orbit until identifying the superior orbital fissure (SOF) on the posterior wall of the orbit was performed. The recurrent meningeal artery was cauterized before reaching the SOF. High-speed drilling using a 3 mm diamond burr was performed from lateral to medial, protecting the periorbita with a malleable retractor. The frontal and temporal dura were exposed, and the lesser sphenoid wing was removed using the diamond burr and Kerrison rongeurs. Partial extradural clinoidectomy was achieved, and the supraorbital and lateral component of the tumor was removed. A dural opening was not necessary due to the extradural location of the tumor.
EEA
As for the TOA, a 0° angled rigid endoscope was used. The right nostril was approached first. The lesion was immediately reached as the sphenoidal sinus and the nasal fossa were occupied by the tumor. A soft and grayish lesion was resected using biopsy rongeurs and ringed curettes. Resection of all the tumor portions located inside the sphenoid sinus was achieved. A full decompression of the right optic nerve was completed after removing the infiltrated bone from the roof and floor of the optic canal. Once the GTR of the lesion was achieved, the dura was exposed with no evidence of cerebrospinal fluid leak. Neuronavigation was used to confirm the limits of the tumor, and hemostasis was performed using gelfoam on the surgical site.
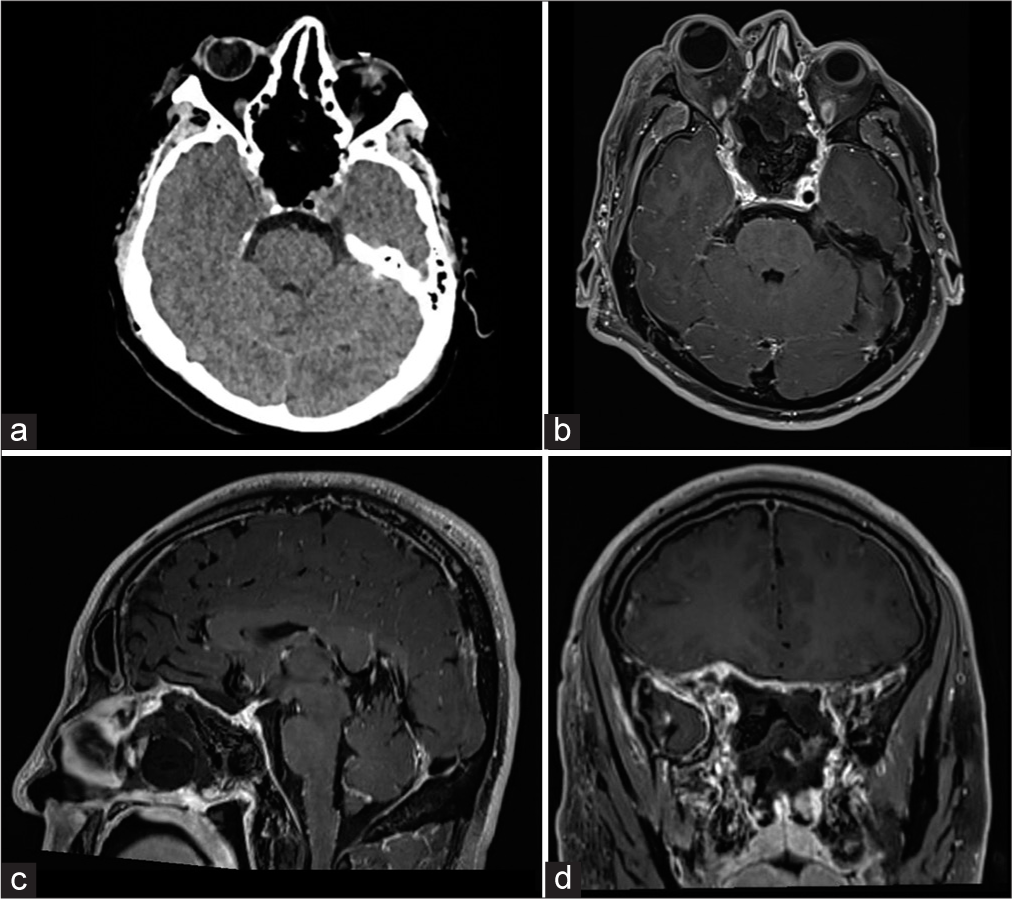
Morphological and immunohistochemical techniques revealed plasma cell dyscrasia and positive staining for lambda light chains consistent with extramedullary plasmacytoma. A PET scan with fluoro-D-glucose ruled out multiple myeloma (MM). A postoperative CT scan revealed GTR [
Video 1
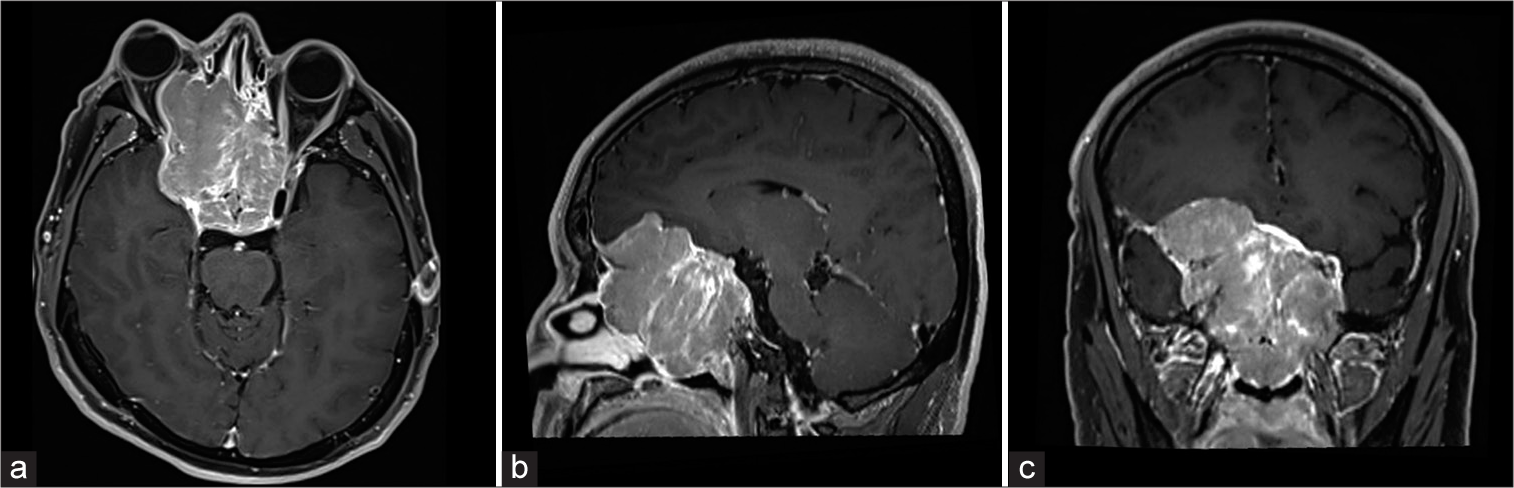
Four months later, the patient consulted due to bilateral loss of vision. A magnetic resonance imaging (MRI) was ordered showing the recurrence of the sellar lesion [
DISCUSSION
Solitary PP shows a slight male predominance, typically manifesting with mass effect and elevated intracranial pressure symptoms rather than endocrine irregularities. To the best of our knowledge, 11 cases of PP have been reported, with series including both sellar and parasellar lesions making up almost 80 cases.[
Surgical approach
Plasmacytomas exhibit a propensity for infiltrating the cavernous sinus and inducing erosion of parasellar bones at a notably higher frequency in comparison to adenomas. This proclivity poses a challenge for surgeons in achieving GTR of the lesion, with an attainment rate as low as 22%. The extent of bone invasion is directly associated with the severity of CNP and diplopia while inversely impacting the incidence of visual disturbances.[
The EEA has remained the preferred surgical modality for lesions within the sellar compartment due to its proven effectiveness and safety profile.[
Recurrent lesions or progression to systemic dyscrasias
Most patients with SEP can undergo remission, with <30% of cases progressing to MM or multiple tumors in a 10-year risk rate of progression, facing worse outcomes in mortality and morbidity in contrast to an SP, foregrounding the importance of determining if the disease is either localized or a systemic plasma cell dyscrasia.[
CONCLUSION
PP remains a rare entity in the intracranial compartment and, when present, often masquerades as pituitary neoplasms. Intrinsic characteristics of the tumor may prompt the physician to consider the possibility of a SEP. Surgical treatment followed by radiotherapy remains the mainstay course of treatment in such cases.
Ethical approval
Institutional Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropiate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Videos available on
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
Acknowledgments
This paper and the research behind it would not have been possible without the exceptional sustenance of the Department of Pathology of this institute.
References
1. Bellan LD, Cox TA, Gascoyne RD. Parasellar syndrome caused by plasma cell leukemia. Can J Ophthalmol. 1989. 24: 331-4
2. Caers J, Paiva B, Zamagni E, Leleu X, Bladé J, Kristinsson SY. Diagnosis, treatment, and response assessment in solitary plasmacytoma: Updated recommendations from a European Expert Panel. J Hematol Oncol. 2018. 11: 10
3. Çakir E, Karaarslan G, Usul H, Baykal S, Arslan E. Solitary plasmacytoma with intracranial intraorbital and, paranasal sinus extension. J Clin Neurosci. 2003. 10: 266-8
4. D’Aguillo C, Soni RS, Gordhan C, Liu JK, Baredes S, Eloy JA. Sinonasal extramedullary plasmacytoma: A systematic review of 175 patients. Int Forum Allergy Rhinol. 2014. 4: 156-63
5. Dhandapani S, Narayanan R, Jayant SS, Sahoo SK, Dutta P, Walia R. Endonasal endoscopic versus microscopic transsphenoidal surgery in pituitary tumors among the young: A comparative study and meta-analysis. Clin Neurol Neurosurg. 2021. 200: 106411
6. Di Somma A, Andaluz N, Cavallo LM, Keller JT, Solari D, Zimmer LA. Supraorbital vs endo-orbital routes to the lateral skull base: A quantitative and qualitative anatomic study. Oper Neurosurg. 2018. 15: 567-76
7. Di Somma A, Guizzardi G, Valls Cusiné C, Hoyos J, Ferres A, Topczewski TE. Combined endoscopic endonasal and transorbital approach to skull base tumors: A systematic literature review. J Neurosurg Sci. 2022. 66: 406-12
8. DiDomenico J, Ampie L, Choy W, Lamano JB, Oyon DE, Kesavabhotla K. Sellar plasmacytomas masquerading as pituitary adenomas: A systematic review. J Clin Neurosci. 2018. 50: 20-3
9. Dimopoulos M, Terpos E, Comenzo RL, Tosi P, Beksac M, Sezer O. International myeloma working group consensus statement and guidelines regarding the current role of imaging techniques in the diagnosis and monitoring of multiple Myeloma. Leukemia. 2009. 23: 1545-56
10. Dimopoulos MA, Kiamouris C, Moulopoulos LA. Solitary pasmacytoma of bone and extramedullary plasmacytoma. Hematol Oncol Clin North Am. 1999. 13: 1249-57
11. International Myeloma Working Gro. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: A report of the International Myeloma Working Group. Br J Haematol. 2003. 121: 749-57
12. Jin L, Gui S, Li C, Bai J, Cao L, Liu C. Differential diagnosis and treatment modality of parasellar plasmacytoma: Clinical series and literature review. World Neurosurg. 2019. 122: e978-88
13. Knobel D, Zouhair A, Tsang RW, Poortmans P, Belkacémi Y, Bolla M. Prognostic factors in solitary plasmacytoma of the bone: a multicenter Rare Cancer Network study. BMC Cancer. 2006. 6: 118
14. Kobayashi N, Oshitari T, Kobayashi K, Onoda T, Ikeda H, Adachi-Usami E. Optic neuritis caused by Rathke’s Cleft cyst in young adult. Case Rep Ophthalmol Med. 2014. 2014: 204762
15. Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. Endoscopic endonasal compared with microscopic transsphenoidal and open transcranial resection of giant pituitary adenomas. Pituitary. 2012. 15: 150-9
16. Kong DS, Young SM, Hong CK, Kim YD, Hong SD, Choi JW. Clinical and ophthalmological outcome of endoscopic transorbital surgery for cranioorbital tumors. J Neurosurg. 2019. 131: 667-75
17. Lee J, Kulubya E, Pressman BD, Mamelak A, Bannykh S, Zada G. Sellar and clival plasmacytomas: Case series of 5 patients with systematic review of 65 published cases. Pituitary. 2017. 20: 381-92
18. Liebross RH, Ha CS, Cox JD, Weber D, Delasalle K, Alexanian R. Clinical course of solitary extramedullary plasmacytoma. Radiother Oncol. 1999. 52: 245-9
19. Moghadasi AN, Aghakhani R, Owji M, Togha M. Pituitary apoplexy presented with optic neuritis. Iran J Neurol. 2013. 12: 117-8
20. Phan K, Xu J, Reddy R, Kalakoti P, Nanda A, Fairhall J. Endoscopic endonasal versus microsurgical transsphenoidal approach for growth hormone–secreting pituitary adenomas-systematic review and meta-analysis. World Neurosurg. 2017. 97: 398-406
21. Rosiñol L, Beksac M, Zamagni E, Van De Donk NW, Anderson KC, Badros A. Expert review on soft-tissue plasmacytomas in multiple myeloma: Definition, disease assessment and treatment considerations. Br J Haematol. 2021. 194: 496-507
22. Shen X, Liu S, Wu C, Wang J, Li J, Chen L. Survival trends and prognostic factors in patients with solitary plasmacytoma of bone: A population-based study. Cancer Med. 2021. 10: 462-70
23. Wiltshaw E. The natural history of extramedullary plasmacytoma and its relation to solitary myeloma of bone and myelomatosis. Medicine (Baltimore). 1976. 55: 217-38
24. Zoli M, Sollini G, Rustici A, Guaraldi F, Asioli S, Altavilla MV. Endoscopic transorbital approach for spheno-orbital tumors: Case series and systematic review of literature. World Neurosurg. 2023. 177: e239-53