- Department of Neurosurgery, Graduate School of Medical Science, Kanazawa University,
- Department of Pathology, Kanazawa University Hospital,
- Department of Neurosurgery, Asanogawa General Hospital,
- Department of Neurosurgery, Kanazawa University, Kanazawa, Japan.
Correspondence Address:
Masaaki Kobayashi, Department of Neurosurgery, Graduate School of Medical Science, Kanazawa University, Kanazawa, Japan.
DOI:10.25259/SNI_257_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Masaaki Kobayashi1, Yasuo Sasagawa1, Satoko Nakada2, Yukihiko Kohda3, Mitsutoshi Nakada4. Dural metastasis from prostate carcinoma mimicking tuberculum sellae meningioma: A case report. 09-Sep-2022;13:407
How to cite this URL: Masaaki Kobayashi1, Yasuo Sasagawa1, Satoko Nakada2, Yukihiko Kohda3, Mitsutoshi Nakada4. Dural metastasis from prostate carcinoma mimicking tuberculum sellae meningioma: A case report. 09-Sep-2022;13:407. Available from: https://surgicalneurologyint.com/surgicalint-articles/11859/
Abstract
Background: Prostate carcinoma rarely metastasizes to the central nervous system. However, when it does, a dural lesion is a common and possible misdiagnosis of meningioma. Here, we describe a case of a 77-year-old man with dural metastasis from prostate carcinoma of the tuberculum sellae.
Case Description: The patient was diagnosed with prostate carcinoma 7 years previously and was well-controlled by hormone therapy. He was incidentally found to have a suprasellar tumor and underwent endoscopic endonasal transsphenoidal surgery because of rapid tumor growth and worsening visual impairment. Since his serum prostate-specific antigen (PSA) level was within the normal range, malignant meningioma was suspected based on the magnetic resonance imaging (MRI) and the course. However, the pathological findings revealed dural metastasis from prostate carcinoma. He received radiation therapy, and the tumor disappeared on MRI. His visual impairment improved without recurrence. This case report highlights that dural metastasis of the tuberculum sellae arose despite the patient’s PSA level being within the normal range, and a single metastasis to the dura was found.
Conclusion: In patients with a history of prostate carcinoma or older men, careful follow-up considering the possibility of metastasis is required when a dural lesion is found.
Keywords: Batson’s venous plexus, Dural metastasis, Prostate carcinoma, Surgical resection, Tuberculum sellae meningioma
INTRODUCTION
Prostate carcinoma usually metastasizes to the pelvic lymph nodes and bones and its rarely to the central nervous system (CNS). CNS metastasis from metastatic prostate carcinoma was noted in 131 of 6282 (2.1%) patients with prostate carcinoma.[
CASE REPORT
A 77-year-old man diagnosed with prostate carcinoma 7 years ago received hormone therapy (gonadotropin-releasing hormone agonist) and was well-controlled. A tumor of the tuberculum sellae was observed on whole-body computed tomography in 2017 [CT;
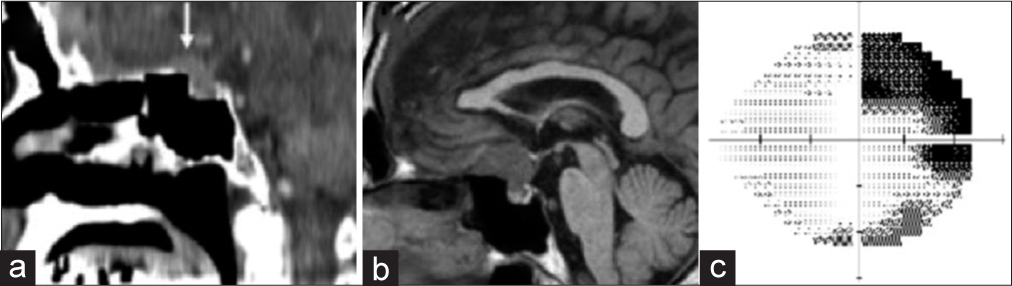
Figure 1:
(a) A high-density mass was seen on sagittal computed tomography in 2017 (white arrow). (b) T1-weighted sagittal image with iso-intensity showing the tumor located at the tuberculum sellae. (c) When the suprasellar tumor was initially found, the patient’s visual field test on the right eye showed upper temporal quadrantanopsia.
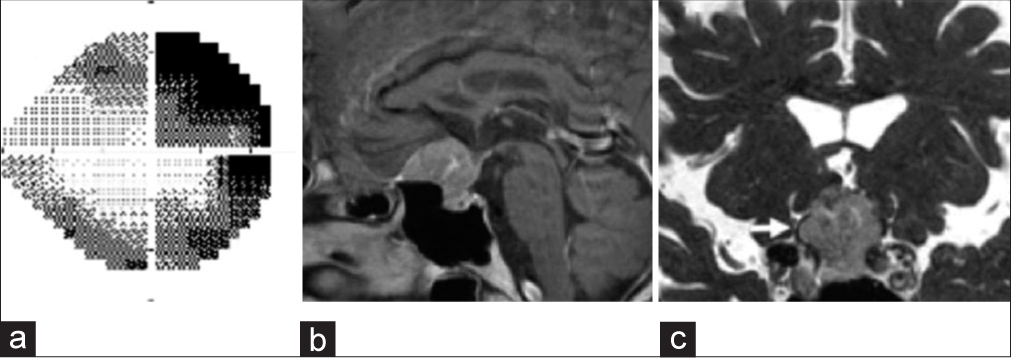
Figure 2:
(a) Two months later, his visual acuity and field were aggravated. (b) Gadolinium T1-weighted image showing tumor growth with homogeneous enhancement two months later. Dural enhancement can also be seen. (c) Fast imaging with steady-state acquisition revealed that the right optic nerve (white arrow) was compressed by the tumor.
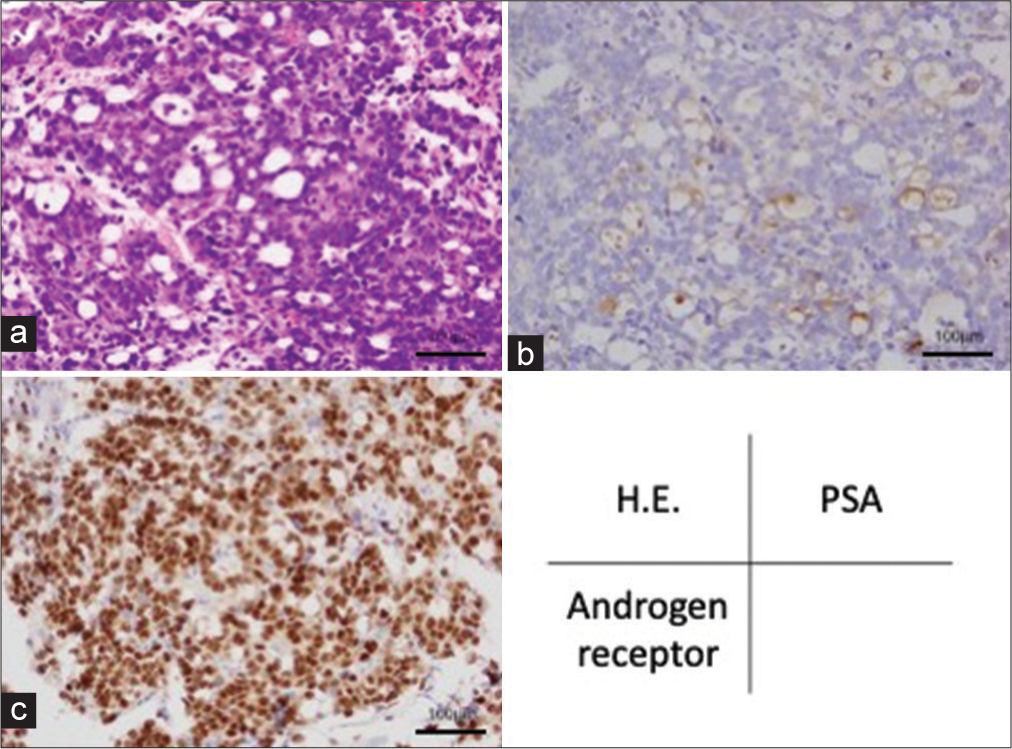
Figure 3:
(a) Atypical epithelioid cells with vacuole proliferated in a sheeting pattern and focal gland-like formation can be seen (hematoxylin-eosin stain). (b and c) Immunohistochemically, the gland-like region was positive for prostate-specific antigen, and the nuclei of the tumor cells were diffusely positive for androgen receptor.
DISCUSSION
CNS metastasis from prostate carcinoma is rare. However, prostate carcinoma is the most common cause of dural metastases. Laigle-Donadey et al. reported in a review of 198 cases of dural metastases in which the major primary tumors were in the prostate (19.5%), breasts (16.5%), and lungs (11%).[
CONCLUSION
Although rare, dural metastasis should be considered a differential diagnosis in patients with prostate cancer. Careful follow-up considering the possibility of metastasis is required when a dural lesion is found in patients with a history of prostate carcinoma or older men.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Chen TY, Lee HJ, Wu TC, Tsui YK, Wu TC. Intracranial dural metastatic prostate cancer can mimic meningioma: A report of two cases. Clin Imaging. 2011. 35: 391-4
2. Ganau M, Gallinaro P, Cebula H, Scibilia A, Todeschi J, Gubian A. Intracranial metastases from prostate carcinoma: Classification, management, and prognostication. World Neurosurg. 2020. 134: e559-65
3. Laigle-Donadey F, Taillibert S, Mokhtari K, Hildebrand J, Delattre JY. Dural metastases. J Neurooncol. 2005. 75: 57-61
4. Lath CO, Khanna PC, Gadewar S, Patkar DP. Intracranial metastasis from prostatic adenocarcinoma simulating a meningioma. Australas Radiol. 2005. 49: 497-500
5. Mitchell RA, Dimou J, Tsui A, Kavar B. Metastatic prostate adenocarcinoma invading an atypical meningioma. J Clin Neurosci. 2011. 18: 1723-5
6. Nayak L, Abrey LE, Iwamoto FM. Intracranial dural metastases. Cancer. 2009. 115: 1947-53
7. Tagle P, Villanueva P, Torrealba G, Huete I. Intracranial metastasis or meningioma? An uncommon clinical diagnostic dilemma. Surg Neurol. 2002. 58: 241-5
8. Tremont-Lukats IW, Bobustuc G, Lagos GK, Lolas K, Kyritsis AP, Puduvalli VK. Brain metastasis from prostate carcinoma: The M. D. Anderson cancer center experience. Cancer. 2003. 98: 363-8