- Department of Neurosurgery, Ehime Prefectural Niihama Hospital, Niihama City, Ehime, Japan,
- Department of Neurosurgery, Ehime University Graduate School of Medicine, Toon City, Ehime, Japan
Correspondence Address:
Shirabe Matsumoto, Department of Neurosurgery, Ehime Prefectural Niihama Hospital, Niihama City, Japan.
DOI:10.25259/SNI_743_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Shirabe Matsumoto1, Noriyuki Fumoto1, Masahiko Tagawa2, Hideo Tanaka1. Endovascular treatment of a ruptured blister-like aneurysm at an azygos anterior cerebral artery: A case report and review of the literature. 27-Jan-2023;14:27
How to cite this URL: Shirabe Matsumoto1, Noriyuki Fumoto1, Masahiko Tagawa2, Hideo Tanaka1. Endovascular treatment of a ruptured blister-like aneurysm at an azygos anterior cerebral artery: A case report and review of the literature. 27-Jan-2023;14:27. Available from: https://surgicalneurologyint.com/surgicalint-articles/12129/
Abstract
Background: Endovascular treatment for a ruptured blister-like aneurysm (BLA) has recently become a hopeful approach. BLAs are usually located on the dorsal wall of the internal carotid artery, whereas one located on the azygos anterior cerebral artery (ACA) is so rare, it has never been reported. We report a case of a ruptured BLA arising at the distal bifurcation of an azygos ACA treated by stent-assisted coil embolization.
Case Description: A 73-year-old woman presented with a disturbance of consciousness. Computed tomography showed diffuse subarachnoid hemorrhage, which was observed to be particularly dense in the interhemispheric fissure. Three-dimensional rotation angiography showed a tiny and conical bulge on the distal bifurcation of the azygos trunk. Follow-up digital subtraction angiography performed on day 4 showed enlargement of the aneurysm, and a BLA arising at the azygos bifurcation was diagnosed. Stent-assisted coiling (SAC) was performed using a low-profile visualized intraluminal support (LVIS) Jr. stent, which was implanted from the left pericallosal artery to the azygos trunk. Follow-up angiography showed that the aneurysm thrombosed gradually and reached complete occlusion 90 days after onset.
Conclusion: SAC for a BLA at the distal bifurcation of an azygos ACA might be an effective treatment option leading to early complete occlusion, but thrombus formation as an intraoperative complication should be noted in the BLA at the bifurcation or the peripheral artery, as in the present case.
Keywords: Blister-like aneurysm, Azygos anterior cerebral artery, Endovascular treatment
INTRODUCTION
Blister-like aneurysms (BLAs) were described for the 1st time in 1986 as rare pathologic entities, accounting for 0.3–1% of all intracranial aneurysms.[
CASE DESCRIPTION
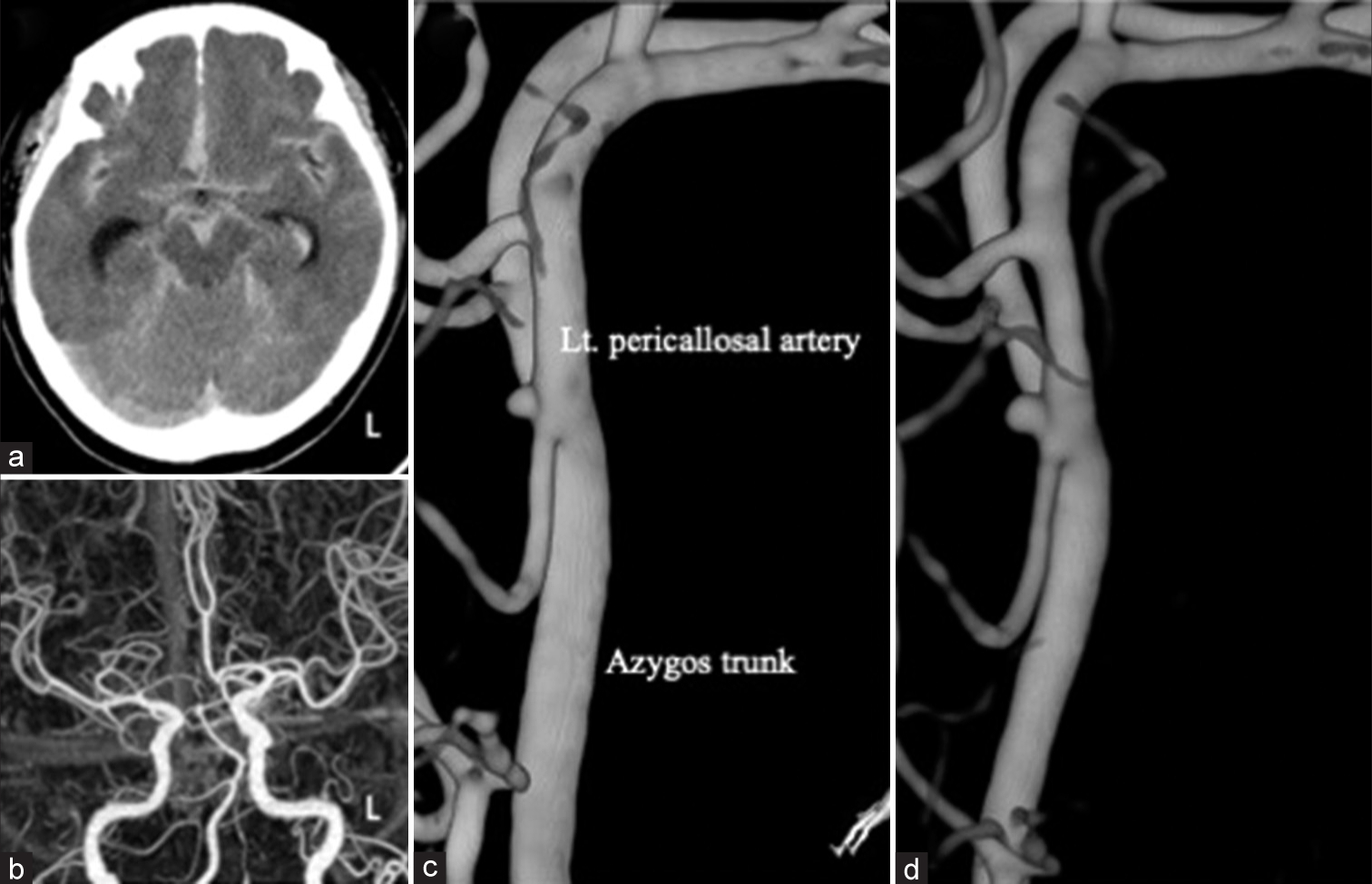
A 73-year-old woman presented with subarachnoid hemorrhage (SAH). She was found unconscious at home and transferred to the hospital. She had a history of hypertension and hyperlipidemia. Her consciousness level was Glasgow Coma Scale E3V4M5. The SAH grade was Hunt and Hess Grade 3 and WFNS Grade 4. The thickest hematoma of the SAH was noted in the interhemispheric fissure on computed tomography (CT) [
Figure 1:
(a) Axial computed tomography (CT) on the initial visit shows diffuse subarachnoid hemorrhage distributed mainly at the interhemispheric fissure. (b) CT angiography shows an azygos anterior cerebral artery (ACA) with hypoplasia of the A1 segment of the right ACA. (c) Three-dimensional rotational angiography (3D-RA) with injection from the left internal carotid artery shows a tiny bulge on the distal bifurcation of an ACA on day 0. This small aneurysm rides from the distal end of the azygos trunk to the left pericallosal artery. (d) The follow-up 3D-RA on day 4 shows obvious enlargement of the aneurysm.
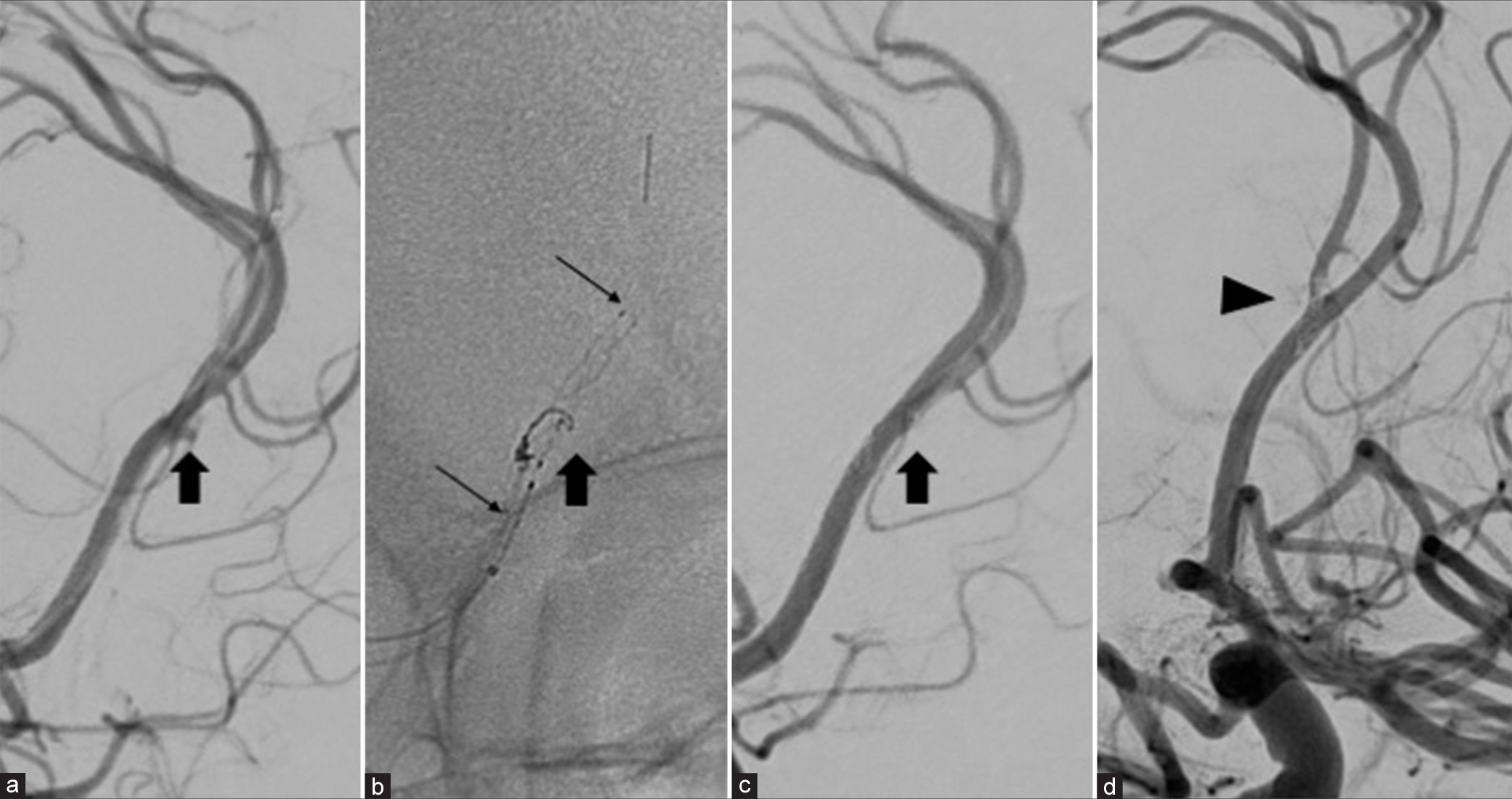
Figure 2:
Stent-assisted coiling was subsequently performed on the same day as the follow-up digital subtraction angiography (DSA). (a) Left carotid angiography at a working angle shows a blister-like aneurysm (BLA) (black arrow) at the distal bifurcation of the azygos trunk and a Headway 17 microcatheter placed on the left pericallosal artery for stenting. (b) The fluoroscopic image demonstrates two loops of a single GalaxyG3 coil inside the BLA (black arrow) and the implanted LVIS junior stent from the left pericallosal artery to the azygos trunk (long thin black arrow). (c and d) The final DSA shows immediate complete occlusion of the BLA (black arrow) and thrombus at the origin of a right pericallosal artery (black arrowhead).
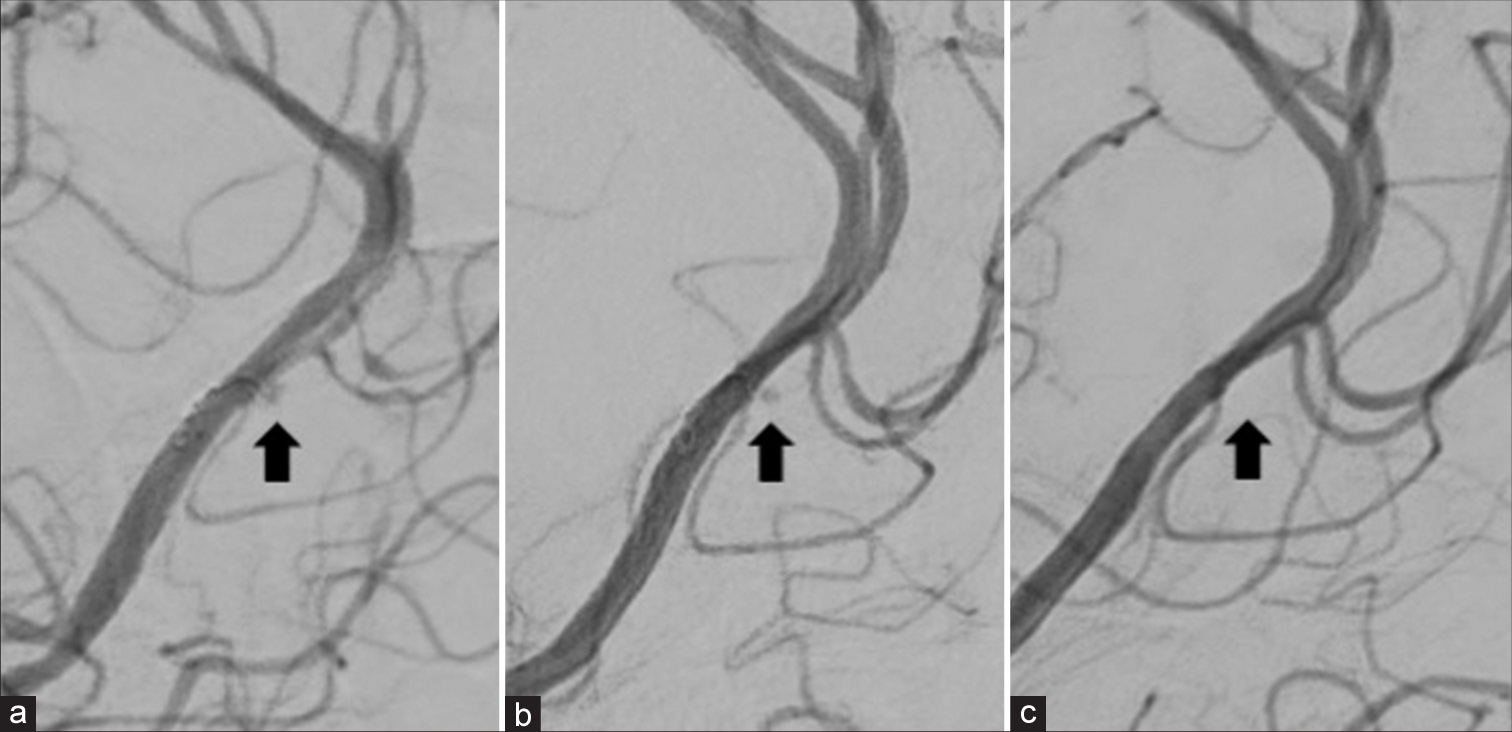
Figure 4:
The follow-up digital subtraction angiography (DSA). (a and b) The DSA 14 (a) and 30 (b) days after onset shows the residual contrast effect and gradual thrombosis within the dome of a blister-like aneurysm (black arrow in (a) and (b)). (c) The DSA at 90 days shows complete obliteration (black arrow).
DISCUSSION
A very rare case of a ruptured BLA at an azygos ACA that was successfully treated with stent-assisted coil embolization and achieved to complete occlusion was reported.
The most frequent typical site of origin of BLAs is the dorsal wall of the ICA at a non-branching site, which was first described as “chimame.”[
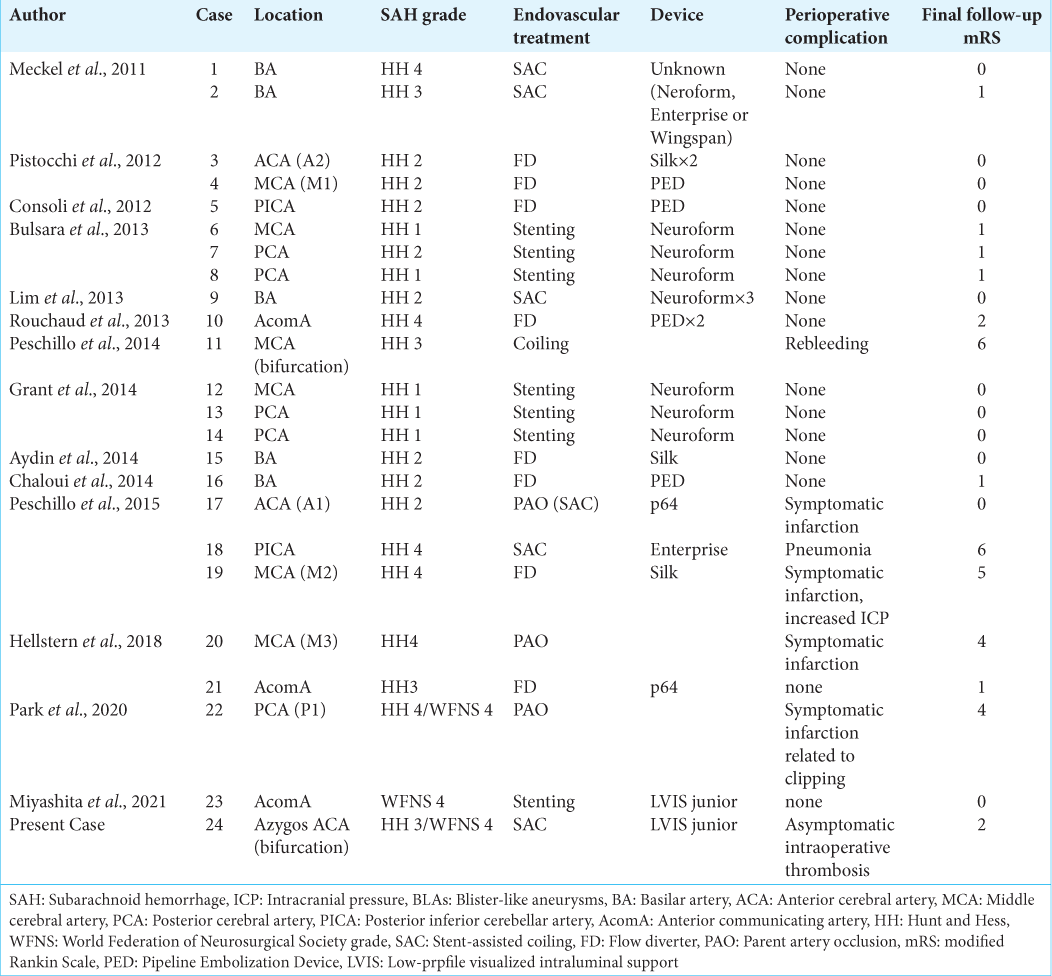
Endovascular treatments for BLAs include parent artery occlusion (PAO), coil embolization, SAC, stenting, and flow diverter (FD). A total of 23 cases of BLAs in atypical locations treated by intervention were found with the details reported; they included PAO in three cases, coiling in one case, SAC in five cases, stenting in seven cases, and FD in eight cases [
Other endovascular treatment options, SAC, stenting, and FD, have good clinical and angiographical outcomes without serious perioperative complications related to the procedure, except in one case treated by FD for an MCA BLA that developed a major stroke.[
Some studies on the treatment of BLAs using a Pipeline Embolization Device (Medtronic, Minneapolis, MN, USA) have shown good clinical and angiographical outcomes.[
Both stenting alone and SAC seemed to be good treatment options for the present case. The advantage of stenting over SAC is safety, since the risk of rupture is low during coil embolization. In retrospective multicenter study, Fang et al. sail that there were 12 (5.7%) perioperative hemorrhagic complications among 212 SAC procedures.[
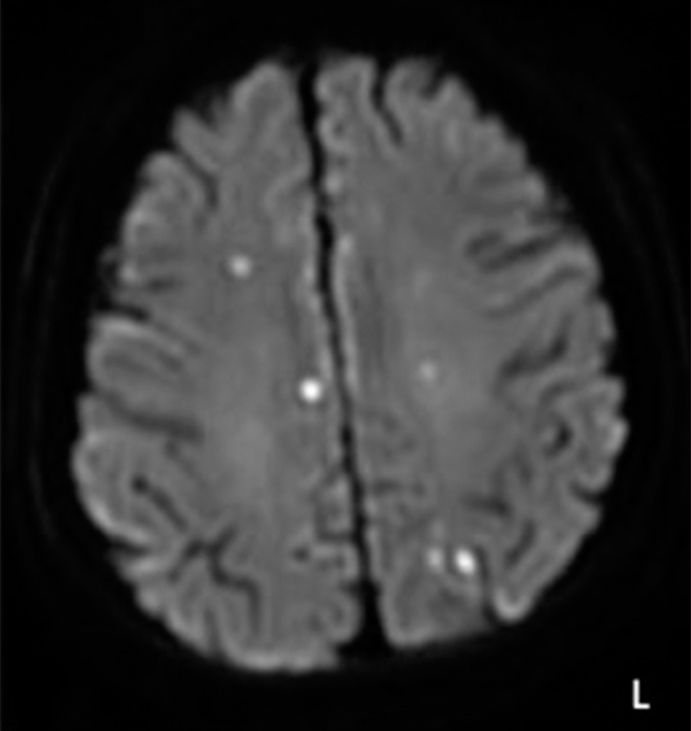
Since SAC achieved complete occlusion immediately at a higher rate than stenting and FD, it is considered to be beneficial for the treatment of BLAs. In SAC, the coil that fills in the aneurysm appears to rapidly promote thrombosis of the ruptured part of a BLA and facilitate healing of the vessel wall of the parent artery by stent placement. The opportunity to pack the coil into the aneurysm is only at the initial endovascular treatment using the stent, and it may be difficult to add coils in case of aneurysmal growth due to the size and shape of the BLA. The most important thing to avoid during SAC seems to be intraoperative rupture due to coil filling. In the present case, a microcatheter was placed in the proximal side of the aneurysm, and the coil was poured into the dome. Although only two loops of coil fit inside the aneurysm, immediate thrombosis of the dome was confirmed, and tighter coil insertion was avoided to prevent intraoperative rupture. However, a thrombus that did not cause a vessel occlusion and a symptomatic infarction was formed on the branch where the stent was not placed. It was considered to be an intraoperative complication that should be noted, in cases such as the present BLA located on the bifurcation of peripheral blood vessels, unlike a non-branching site of the ICA.
The pathophysiology of BLAs is not well understood. BLAs are said to consist of a fragile fibrin layer in a focal arterial segment, unlike true aneurysms.[
An azygos ACA is a rare anatomical anomaly with an incidence of approximately 1.1%.[
Because BLAs are presumed to be a type of vascular dissection or pseudoaneurysm, their treatment should not focus solely on the aneurysm sac but should also address the reconstruction of the wall of the affected vessel. The present aneurysm was completely occluded by placing a LVIS junior stent and only two loops of a coil in the dome. If this aneurysm was a saccular aneurysm, tighter coil embolization may have been needed to achieve complete occlusion. It was highly likely that the coil outside the aneurysm was sandwiched between the vascular walls by the stent, blocking the re-entry site of the BLA and completely eradicating the BLA. In our view, because the present aneurysm was a BLA, it had been completely occluded by the repairment of the parent artery by the stenting, even though it was not tightly packed by the coil. Based on this finding, the present aneurysm was diagnosed as a BLA at the azygos ACA.
CONCLUSION
This is the first report of a ruptured BLA arising on the bifurcation of the azygos trunk treated by SAC. The present case showed good clinical and radiological outcomes. SAC might be an effective treatment for BLAs leading to early complete occlusion, but thrombus formation as an intraoperative complication should be noted in BLAs at the bifurcation or a peripheral artery, such as the present case.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Andaluz N, Zuccarello M. Blister-like aneurysms of the anterior communicating artery: A retrospective review of diagnosis and treatment in five patients. Neurosurgery. 2008. 62: 807-11 discussion 811
2. Baptista AG. Studies on the arteries of the brain. II. The anterior cerebral artery: Some anatomic features and their clinical implications. Neurology. 1963. 13: 825-35
3. Endo S, Ogiichi TM, Kurimoto T, Nishijima M, Takaku MA. Pathological study of intracranial artery dissection with subarachnoid hemorrhage. Surg Cereb Stroke. 1997. 25: 169-76
4. Fang Y, Zhu D, Peng Y, Zhong M, Xu J, Li Q. Treatment of blood blister-like aneurysm with stent-assisted coiling: A retrospective multicenter study. World Neurosurg. 2019. 126: e486-91
5. Fujimoto Y, Yamanaka K, Nakajima Y, Yoshimura K, Yoshimine K. Ruptured aneurysm arising from the proximal end of an azygos anterior cerebral artery-case report. Neurol Med Chir. 2004. 44: 242-4
6. Hellstern V, Aguilar-Perez A, AlMatter M, Bhogal P, Henkes E, Ganslandt O. Microsurgical clipping and endovascular flow diversion of ruptured anterior circulation blood blister-like aneurysms. Interv Neuroradiol. 2018. 24: 615-23
7. Ishikawa T, Nakamura N, Houkin K, Nomura M. Pathological consideration of a “blister-like” aneurysm at the superior wall of the internal carotid artery: Case report. Neurosrugery. 1997. 40: 403-6
8. Kaspera W, Ladzinski P, Stowinski J, Kopera M, Tomalski W, Slaska-Kaspera A. Blood flow velocity in the arteries of the anterior cerebral artery complex in patients with an azygos anterior cerebral artery aneurysm: A transcranial color-coded sonography study. Clin Neurol Neurosurg. 2009. 111: 63-8
9. Linfante I, Mayich M, Sonig A, Fujimoto J, Siddiqui A, Dabus G. Flow diversion with pipeline embolic device as treatment of subarachnoid hemorrhage secondary to blister aneurysms: Dual-center experience and review of the literature. J Neurointerv Surg. 2017. 9: 29-33
10. Miyashita K, Nambu K, Shimizu Y, Tohma Y. Blister-like aneurysm of the anterior communicating artery treated with only low-profile visualized intraluminal support junior stent. Surg Neurol Int. 2021. 12: 564
11. Mohlenbruch MA, Seker F, Ozluk E, Kizilkilic O, Broussalis E, Killer-Oberpfalzer M. Treatment of ruptured blister-like aneurysms with the FRED flow diverter: A multicenter experience. AJNR Am J Neuroradiol. 2020. 41: 2280-4
12. Nakagawa F, Kobayashi S, Takemae T, Sugita K. Aneurysms protruding from the dorsal wall of the internal carotid artery. J Neurosurg. 1986. 65: 303-8
13. Park S, Miyakoshi A, Yoshida S, Arai D, Kawanabe Y, Sato T. Ruptured blister-like aneurysm arising from the proximal posterior cerebral artery. World Neurosurg. 2021. 147: 38-41
14. Peschillo S, Cannizzaro D, Caporlingua A, Missori P. A systematic review and meta-analysis of treatment and outcome of blister-like aneurysms. AJNR Am J Neuroradiol. 2016. 37: 856-61
15. Peschillo S, Miscusi M, Caporlingua A, Cannizzaro D, Santoro A, Delfini R. Blister-like aneurysms in atypical locations: A single-center experience and comprehensive literature review. World Neurosurg. 2015. 84: 1070-9
16. Peschillo S, Missori P, Piano M, Cannizzaro D, Guidetti G, Santoro A. Blister-like aneurysms of middle cerebral artery: A multicenter retrospective review of diagnosis and treatment in three patients. Neurosurg Rev. 2014. 38: 197-203
17. Pistocchi S, Blanc R, Bartolini B, Piotin M. Flow diverters at and beyond the level of the circle of Willis for the treatment of intracranial aneurysms. Stroke. 2012. 43: 1032-8
18. Regelsberger J, Matschke J, Gryzyska U, Ries T, Fiehler J, Koppen J. Blister-like aneurysms-a diagnostic and therapeutic challenge. Neurosurg Rev. 2011. 34: 409-16
19. Sarikaya B, Oksuz E, Deniz FE, Firat MM. Endovascular treatment of a ruptured aneurysm at a very rare location distal azygos anterior cerebral artery. Interv Neuroradiol. 2008. 14: 457-60
20. Scerrati A, Visani J, Flacco ME, Ricciardi L, Trungu S, Raco A. Endovascular treatment of ruptured intracranial blister aneurysms: A systematic review and meta-analysis. AJNR Am J Neuroradiol. 2021. 42: 538-45