- Department of Anesthesiology, Advocate Illinois Masonic Medical Center, Chicago, Illinois, USA
- Ghaly Neurosurgical Associates, Aurora, Chicago, Illinois, USA
- Department of Anesthesiology, University of Illinois, Chicago, Illinois, USA
Correspondence Address:
Ramsis F. Ghaly
Department of Anesthesiology, Advocate Illinois Masonic Medical Center, Chicago, Illinois, USA
Department of Anesthesiology, University of Illinois, Chicago, Illinois, USA
DOI:10.4103/sni.sni_420_17
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Ramsis F. Ghaly, Ana Plesca, Shalini Rana, Kenneth D. Candido, Nebojsa Nick Knezevic. Gabapentin-related suicide: Myth or fact?. 23-Oct-2018;9:210
How to cite this URL: Ramsis F. Ghaly, Ana Plesca, Shalini Rana, Kenneth D. Candido, Nebojsa Nick Knezevic. Gabapentin-related suicide: Myth or fact?. 23-Oct-2018;9:210. Available from: http://surgicalneurologyint.com/surgicalint-articles/9045/
Abstract
Background:The opioid epidemic in America is real and is estimated to be the number one cause of death in adults under 50 years of age. Finding alternative analgesic medications is part of the effort to decrease the prescription of narcotics, with gabapentin being at the top of the list.
Case Description:In the present case, we discuss the side-effects of gabapentin, used as part of the multimodal treatment approach of painful spinal degenerative disease. The patient stated that he had noticed personality changes after gabapentin was initiated, and that he had become more depressed, frustrated, and aggressive. His uncontrolled pain and acute mood changes led him to attempt suicide by hanging himself. Gabapentin was discontinued and the patient's suicidal ideation completely subsided.
Conclusion:It is imperative to screen, identify, and appropriately manage patients with underlying psychiatric disorders prior to initiating pain management with gabapentin. Therefore, it is crucial to raise awareness of gabapentin as a potential cause of depression, aggressive behavior, and suicidal ideation.
Keywords: Chronic low back pain, gabapentin, suicide
INTRODUCTION
Since the beginning of the new millennium, opioid-related deaths have increased five times in women and almost four times in men. More than half a million people died from drug overdoses between 2000 and 2015. It is estimated that 91 Americans die every day from an opioid overdose.[
Gabapentin is an antiepileptic medication, and is also frequently used for off-label purposes such as acute and chronic pain management in various neurological and neurosurgical disorders. Reported off-label uses are migraines, trigeminal neuralgia, complex regional pain syndrome, drug and alcohol withdrawal seizures, and bipolar disorder.[
However, despite its popularity, gabapentin is not free of side effects. The psychiatric side-effects of gabapentin have been infrequently discussed. In this article, we will report a case of a patient who demonstrated aggressive and suicidal behavior after initiation of gabapentin.
CASE DESCRIPTION
A 28-year-old male with a history of chronic low back pain described his pain as constantly present, 4–8/10 in intensity, shooting down both legs with any movement, and with associated tingling and numbness. His exercise tolerance had decreased to walking a quarter of a mile/day over a period of 6 months, and he ended up quitting his job due to his debilitating pain.
Besides the low back pain, for which the patient frequently visited the emergency room, he also had left testicular pain. He was diagnosed with orchitis, and was started on oral antibiotics. Despite the treatment, his testicular pain worsened, and he was prescribed additional antibiotics. The infection resulted in left groin pain that extended to his mid-back, buttock, and radiated down the hamstring and lateral aspect of leg, correlating with L5-S1 dermatomes.
His electromyography (EMG) showed decreased sural nerve signals. Magnetic resonance imaging (MRI) of his lumbar spine demonstrated central canal stenosis at the L3-4-5 levels and disc herniations at L3-4 and L4-5 levels. His hip X-ray, computed tomography (CT) of the abdomen, ultrasound of the testicles, and urinalysis were within normal limits.
He was first started on nonsteroidal anti-inflammatory drugs (NSAIDs), and then acetaminophen/hydrocodone and muscle relaxants were added to his treatment regimen without any relief. The next step was physical therapy and two lumbar epidural steroid injections, which provided no relief. He refused to get a third lumbar epidural steroid injection, and was referred to a neurologist for further evaluation. He was then started on gabapentin 300 mg three times per day, a dose that was gradually increased over 4 weeks to 1600 mg/day by his orthopedic surgeon in conjunction with muscle relaxants and NSAIDs.
The patient and his fiancé stated that they noticed personality changes in him; he became more depressed and aggressive. They attributed the changes in his behavior to gabapentin. His pain and mood had progressively deteriorated to the point where he attempted to commit suicide by hanging himself by the neck using his belt and a door jam. He was immediately found by his fiancé who emergently took him to the hospital. While he was hospitalized, all his medications were stopped, including gabapentin, and he was started on mood stabilizers. He noticed that his personality had changed back to normal and his suicidal ideation had subsided, after which he was discharged home.
At this point, after being on a multimodal pain medication regimen including lumbar epidural steroid injections without significant relief, the patient visited our clinic for a second opinion about his back pain. On examination, his vital signs were normal. However, he had an antalgic gait with a forward leaning posture of 15–20 degree, and was unable to stand on his toes or heels without support. Palpation also elicited mild tenderness over the lower lumbar region. His motor strength was 5/5 in both upper extremities and 4/5 in both lower extremities. Extension of the lower extremities was limited to 0 degrees, whereas range of flexion was 70 degrees with improvement in pain. Patellar reflexes were absent bilaterally, ankle reflexes were 1(+) bilaterally, Babinski's sign was negative, and the straight leg raise test was positive, left > right, 1(+) at 60 degrees. The remainder of the examination, including cranial nerve examination, respiratory, cardiovascular and abdominal was normal. Based on the clinical picture, he was recommended conservative treatment in a tertiary center.
DISCUSSION
Since its introduction in the market in 1993, gabapentin has consistently gained in popularity. A significant proportion of this use has been for non-FDA approved pain conditions. Besides its use in treating refractory partial seizures, the FDA also approved it in 2002 for the treatment of postherpetic neuralgia. Gabapentin is a structural analogue of the neurotransmitter γ-aminobutyric acid (GABA), but it does not bind to GABA receptors nor does it block uptake or metabolism of GABA. Although the mechanism of action of gabapentin is unclear, it does block voltage-gated Ca++ channels by binding to the α2-delta subunit in the dorsal horn, producing a reduced calcium influx, thereby reducing release of glutamate and substancePfrom primary nociceptive afferents and diminishing propagation of pain signals.[
The recommended starting dose for neuropathic pain is 300 mg on day 1, followed by 300 mg twice daily on day 2, and then 300 mg three times a day thereafter. This dose can be gradually increased to 1800 mg/day as 300 mg oftentimes is not sufficient. The most common side effects of gabapentin in therapeutic doses are drowsiness, somnolence, dizziness, movement disorders, diarrhea, and weight gain.[
In addition, cases have been reported of gabapentin-related suicidal acts and aggressive behavior [
The mechanism of suicide linked to antiepileptic drugs (AEDs) is not very well understood, however, there is a possibility that AEDs lower the threshold for manifesting psychiatric symptoms in individuals who are susceptible because of genetic or historical predisposition to psychiatric disorders.[
One of the factors that could impact patient's suicide intent is their chronic pain. Suicide rate among chronic pain patients was found to be significantly greater than the general population. Literature review concludes that suicidal ideation, suicide attempts, and suicide completions appeared to be common in chronic pain patients. As a result of research conducted in 2003, the American Psychiatric Association included chronic pain as a risk factor for suicidal ideation in their guidelines for the assessment and treatment of patients with suicidal behavior. Two approaches have been recommended – routinely measuring depression and suicidality in any chronic pain patient with tools such as the Beck Depression Inventory or monitoring pain and depression with the Brief Pain Inventory at every visit. High scores on pain or depression rating scales should stimulate the physician to inquire about suicidality.[
Based on the most recent systematic review, gabapentin was tested in nine different chronic pain conditions considered to be neuropathic in origin. Numbers needed to treat for an additional beneficial outcome were between 5 and 7 in postherpetic neuralgia and peripheral diabetic neuropathy. Evidence for other types of neuropathic pain was very limited. More than half of the patients treated with gabapentin will not have a justifiable pain relief.[
As part of the program for decreasing opioid prescriptions, the U.S. Centers for Disease Control and Prevention lists gabapentin as an appropriate nonopioid treatment for chronic pain, recommending the drug as a first-line agent for neuropathic pain. More recently, the American Pain Society recommended that gabapentin be included in the Clinical Practice Guideline in the management of postoperative pain.[
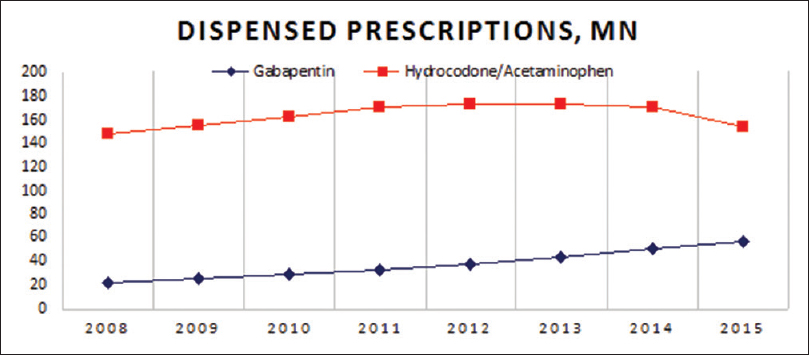
We noticed a 23% decrease in opioid dispensed prescriptions in the last 7 years while concomitantly witnessing a total of 153% increase in gabapentin prescriptions over the same period of time [
Figure 1
IMS Health data, Gabapentin dispensed prescriptions, in millions[
While it may be legal for medical practitioners to prescribe medications for off-label uses, in the United States it is illegal for manufacturers to market medications for off-label uses, which is one reason why Pfizer was fined $420 million after it was acquired from Warner-Lambert.[
After a recent survey in the USA, gabapentin had the highest proportion of off-label prescriptions (83%) among 160 commonly prescribed drugs; and only 20% of its off-label use had strong scientific evidence of clinical efficacy. This accounted for more than 90% of its sales.[
Numerous cases have been reported supporting the evidence of the abuse potential of gabapentin, in which individuals with a history of opioid abuse were the most susceptible. Prescribers should be aware of high-risk populations and monitor for signs of abuse because gabapentin is often co-prescribed with opioids and pain patients often receive prescription for benzodiazepines due to concomitant anxiety and/or difficulty sleeping.[
In a recent study performed in Central Appalachian region, a near 3,000% increase was found in the use of gabapentin “to get high” from 2008 to 2014 in a cohort of 503 prescription drug users. When compared to the previous year reports, a 165% increase was observed. The major sources of gabapentin were physicians (52%) and drug dealers (36%).[
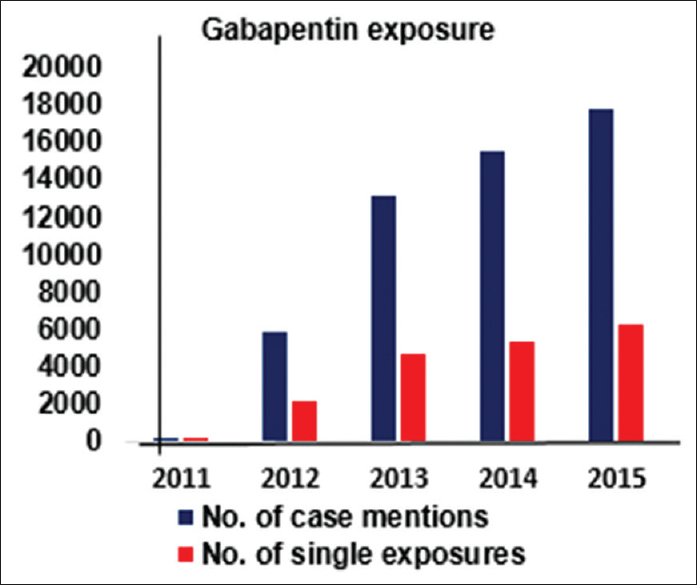
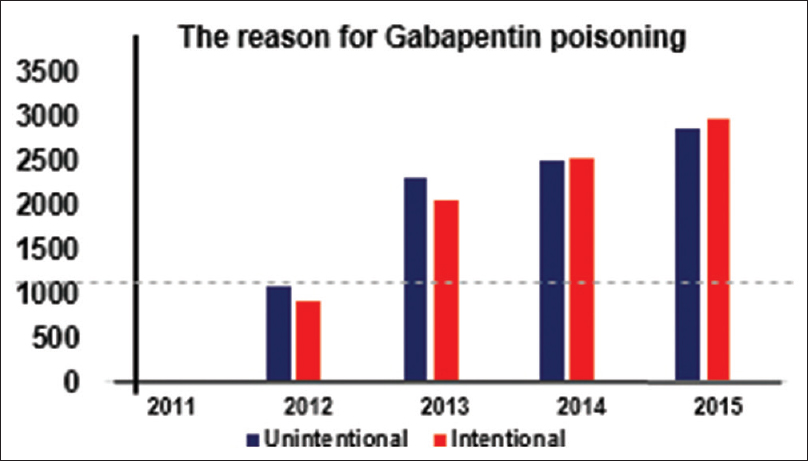
From the data presented in Figures
Figure 2
Clinical Toxicology report for Gabapentin exposure. American Association of Poison Control Centers Annual reports[
Figure 3
The reason for Gabapentin poisoning. American Association of Poison Control Centres AAPCC[
Antiepileptic drugs and suicidality
Numerous studies have been published to confirm or deny the role that antiepileptic drugs play in increasing the risk of suicidal behavior; however, so far they lack concordance and are limited by various methodological limitations.
The FDA issued a warning regarding the increased risk of suicide in patients taking AEDs based on a study performed among 28,000 people taking AEDs and 16,000 people taking placebo. Four completed suicides were registered in the group that was taking AED and none in the placebo group. The odds ratio for the association between AEDs and suicidal behavior or ideation was 1.8 (95% CI, 1.24–2.66) and 1.45 (95% CI, 0.93–2.3) for ideation alone, which did not reach statistical significance.[
The validity of this meta-analysis was questioned after careful review of the data based on several methodological problems, including the source of outcome data on suicidality, statistical methodologies used, heterogeneity of the trials, and the difference in mechanism of action of the 11 AEDs evaluated.[
The suicide attempt rates before and after gabapentin was prescribed were investigated in a cohort of 131,178 patients. The investigators found no significant differences in suicide attempt rates before (3.45/1000 patient years) versus after gabapentin prescription (3.45/1000 patient years).
While no differences were identified before and after gabapentin in the nonpsychiatric population, up to a 5 times post-prescription reduction in suicide-attempts rate were seen for bipolar disorder, major depressive disorder, and other psychiatric conditions. The pre-prescription suicide-attempts rate for patients with bipolar disorder was 48 per 1000 person years (PY); following prescription, the SAs rate decreased to 32 per 1000 PY (a 33% decrease). In patients with pain disorders, the SAs rate was 3 per 1000 PY before gabapentin prescription, and remained unchanged after gabapentin prescription. These findings suggest that gabapentin may have a protective effect among patients at an increased risk of SAs. The absence of a significant association between gabapentin and SAs in patients receiving a prescription for a pain disorder suggests that having comorbid psychiatric and pain disorders may significantly increase SA risk, which greatly decreases if the pain disorder is successfully treated with gabapentin. Their investigation also showed that patients who are on gabapentin monotherapy for pain only are at a lower risk of suicide in comparison to patients who are on multiple medications. The rationale behind this is that patients who are on multiple medications have multiple comorbidities that are difficult to treat and are already at an increased risk for suicide at baseline.[
CONCLUSION
With the increase in opioid misuse and overdose-related deaths nationwide, health care providers are encouraged to find a nonopioid analgesic. Application of gabapentin for treatment is not without side-effects, especially in patients with an undiagnosed psychiatric disorder or who are at risk of suicidality. It is imperative to screen, identify, and appropriately manage patients with underlying psychiatric disorders prior to initiating pain management with gabapentin. The recommended strategy is to perform more studies to identify the group at high risk of suicide between patients taking gabapentin and those who are not. Therefore, it is crucial to raise awareness of gabapentin as a potential cause of depression, aggressive behavior, and suicidal ideation.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ajori L, Nazari L, Mazloomfard MM, Amiri Z. Effects of gabapentin on postoperative pain, nausea and vomiting after abdominal hysterectomy: A double blind randomized clinical trial. Arch Gynecol Obstet. 2012. 285: 677-82
2. Cantrell FL, Mena O, Gary RD, McIntyre IM. An acute gabapentin fatality: A case report with postmortem concentrations. Int J Legal Med. 2015. 129: 771-5
3. .editorsCDC. Wide-ranging online data for epidemiologic research (WONDER). Atlanta, GA: CDC, National Center for Health Statistics; 2016. p.
4. Chang CY, Challa CK, Shah J, Eloy JD. Gabapentin in acute postoperative pain management. Biomed Res Int. 2014. 2014: 631756-
5. Chou R, Gordon DB, Leon-Casasola OA, Rosenberg JM, Bickler S, Brennan T. Management of Postoperative Pain: A Clinical Practice Guideline From the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016. 17: 131-57
6. Evoy KE, Morrison MD, Saklad SR. Abuse and Misuse of Pregabalin and Gabapentin. Drugs. 2017. 77: 403-26
7. Fink K, Meder W, Dooley DJ, Gothert M. Inhibition of neuronal Ca(2+) influx by gabapentin and subsequent reduction of neurotransmitter release from rat neocortical slices. Br J Pharmacol. 2000. 130: 900-6
8. Fishbain DA, Lewis JE, Gao J. The pain suicidality association: A narrative review. Pain Med. 2014. 15: 1835-49
9. Gibbons RD, Hur K, Brown CH, Mann JJ. Gabapentin and suicide attempts. Pharmacoepidemiol Drug Saf. 2010. 19: 1241-7
10. Gugelmann H, Shofer FS, Meisel ZF, Perrone J. Multidisciplinary intervention decreases the use of opioid medication discharge packs from 2 urban EDs. Am J Emerg Med. 2013. 31: 1343-8
11. Hesdorffer DC, Berg AT, Kanner AM. An update on antiepileptic drugs and suicide: Are there definitive answers yet?. Epilepsy Curr. 2010. 10: 137-45
12. Kanner AM. Are antiepileptic drugs used in the treatment of migraine associated with an increased risk of suicidality?. Curr Pain Headache Rep. 2011. 15: 164-9
13. Mao J, Chen LL. Gabapentin in pain management. Anesth Analg. 2000. 91: 680-7
14. Mack A. Examination of the evidence for off-label use of gabapentin. J Manag Care Pharm. 2003. 9: 559-68
15. Markowitz JS, Finkenbine R, Myrick H, King L, Carson WH. Gabapentin abuse in a cocaine user: Implications for treatment?. J Clin Psychopharmacol. 1997. 17: 423-4
16. Mersfelder TL, Nichols WH. Gabapentin: Abuse, Dependence, and Withdrawal. Ann Pharmacother. 2016. 50: 229-33
17. Middleton O. Suicide by gabapentin overdose. J Forensic Sci. 2011. 56: 1373-5
18. Misra S, Parthasarathi G, Vilanilam GC. The effect of gabapentin premedication on postoperative nausea, vomiting, and pain in patients on preoperative dexamethasone undergoing craniotomy for intracranial tumors. J Neurosurg Anesthesiol. 2013. 25: 386-91
19. Newman M. Bitter pills for drug companies. BMJ. 2010. 341: 5095-
20. Pinninti NR, Mahajan DS. Gabapentin-associated aggression. J Neuropsychiatry Clin Neurosci. 2001. 13: 424-
21. Radley DC, Finkelstein SN, Stafford RS. Off-label prescribing among office-based physicians. Arch Intern Med. 2006. 166: 1021-6
22. Reccoppa L, Malcolm R, Ware M. Gabapentin abuse in inmates with prior history of cocaine dependence. Am J Addict. 2004. 13: 321-3
23. Reeves RR, Burke RS. Abuse of combinations of gabapentin and quetiapine. Prim Care Companion CNS Disord. 2014. 16:
24. Reimann W. Inhibition by GABA, baclofen and gabapentin of dopamine release from rabbit caudate nucleus: Are there common or different sites of action?. Eur J Pharmacol. 1983. 94: 341-4
25. Smith RV, Havens JR, Walsh SL. Gabapentin misuse, abuse and diversion: A systematic review. Addiction. 2016. 111: 1160-74
26. Smith RV, Lofwall MR, Havens JR. Abuse and diversion of gabapentin among nonmedical prescription opioid users in Appalachian Kentucky. Am J Psychiatry. 2015. 172: 487-8
27. Vollmer KO, von Hodenberg A, Kolle EU. Pharmacokinetics and metabolism of gabapentin in rat, dog and man. Arzneimittelforschung. 1986. 36: 830-9
28. Wamil AW, McLean MJ. Limitation by gabapentin of high frequency action potential firing by mouse central neurons in cell culture. Epilepsy Res. 1994. 17: 1-11
29. Wiffen PJ, Derry S, Bell RF, Rice AS, Tolle TR, Phillips T. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2017. 6: CD007938-
30. Last accessed on 2017 Nov 15. http://www.aapcc.org/annual-reports.
31. Last accessed on 2017 Nov 15. https://www.fda.gov/Drugs/DrugSafety/PostmarketDrug Safety InformationforPatientsandProviders/ucm100197.htm.
32. Last accessed on 2017 Nov 15. http://www.huffingtonpost.com/martha-rosenberg/pfizer-neurontin-suicide_b_875603.html.
33. Last accessed on 2017 Nov 15. http://www.imshealth.com/files/web/IMSH%20Institute/Reports/Medicines-Use-and-Spending-in-the-US-2015-to-2020/IMS_Institute_Medicines_Use_and_Spending_in_2015.pdf.