- Department of Neurosurgery, Aga Khan University Hospital, Aga Khan University, Karachi, Sindh, Pakistan.
- Department of Radiology, Aga Khan University Hospital, Aga Khan University, Karachi, Sindh, Pakistan.
Correspondence Address:
Fatima Mubarak, Department of Radiology, Aga Khan University Hospital, Aga Khan University, Karachi, Sindh, Pakistan.
DOI:10.25259/SNI_990_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Mohammad Hamza Bajwa1, Mohammad Yousuf Ul Islam1, Fatima Mubarak2. Giant tumefactive perivascular spaces in a pediatric patient: A rare radiological entity. 14-Dec-2021;12:613
How to cite this URL: Mohammad Hamza Bajwa1, Mohammad Yousuf Ul Islam1, Fatima Mubarak2. Giant tumefactive perivascular spaces in a pediatric patient: A rare radiological entity. 14-Dec-2021;12:613. Available from: https://surgicalneurologyint.com/surgicalint-articles/11286/
Abstract
Background: Giant tumefactive perivascular spaces (TPVS) are radiological rarities and may mimic other neurological structural lesions. Fewer than 80 cases have been reported in the literature with even fewer in the pediatric population.
Case Description: The authors present an image report showcasing a 3-year-old boy presenting with uncontrolled seizures despite multiple anti-epileptic medications. His magnetic resonance imaging showed multiple, non-contrast enhancing cyst clusters within the left parieto-occipital region that was hyperintense on T2-weighted imaging, and isointense to cerebrospinal fluid. Due to a characteristic absence of perilesional edema seen on fluid-attenuated inversion recovery imaging or diffusion restriction on diffusion-weighted imaging (DWI) sequences, this was diagnosed as a giant TPVS.
Conclusion: Accurate diagnosis of these rare radiological entities is based on pathognomonic findings that can help prevent unnecessary surgery and guide management for patients, particularly in the pediatric population as seen in our case.
Keywords: Giant tumefactive perivascular space, Neuro-radiology, Pediatric, Virchow–robin space
INTRODUCTION
Perivascular (Virchow-Robin) spaces (PVSs) were initially described by Virchow in 1851 and Robin in 1859 as pial-lined fluid filled structures accompanying penetrating arteries as they enter the cerebrum.[
The rarity of these lesions is such that fewer than 80 giant (larger than 15 mm) tumefactive perivascular spaces (TPVSs) have been reported in the literature, and even fewer in the pediatric population.[
CLINICAL IMAGE
A 3-year-old boy presented to our clinic with partial clonic seizures for the past 1½ years. These lasted for 30 min and were terminated with an anticonvulsant. Every episode occurred in the same fashion and on the same side of the body. He had no prior history of loss of consciousness, intractable vomiting, fall, or trauma. Despite being on two anti-epileptic medications from his previous medical team, he had another seizure a few months before presentation. On examination, he was awake, alert, and his speech was intact. He was able to follow commands with no motor deficits. He did not exhibit any cerebellar signs or gait abnormalities. Systemic examination was also unremarkable.
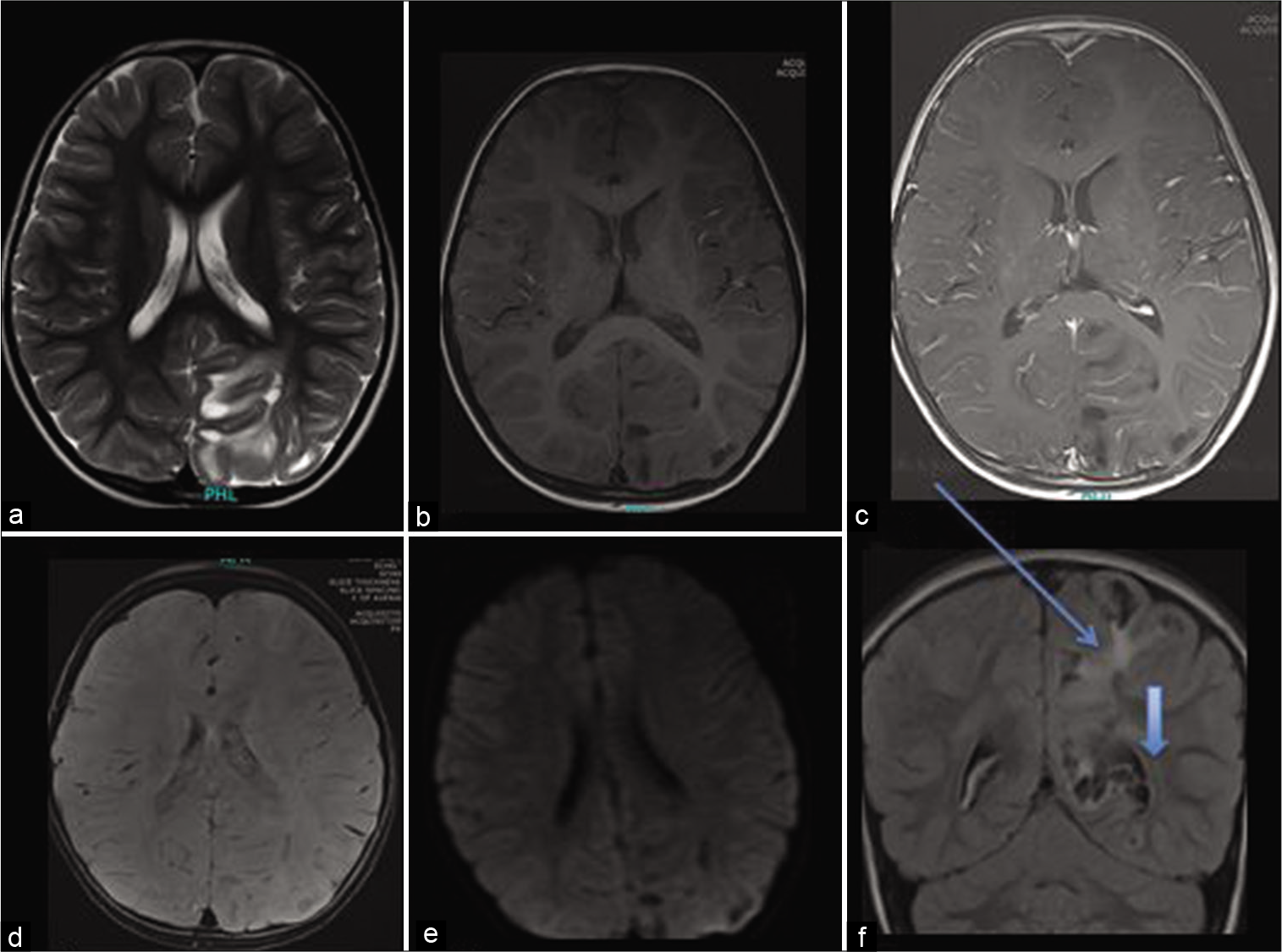
On magnetic resonance imaging (MRI) brain with contrast imaging, there was an irregular and abnormal signal intensity lesion in the left parieto-occipital region [
Figure 1:
Axial T2 (a), Axial T1 plain (b), and post-contrast (c). In addition, axial susceptibility weighted imaging (SWI) (d), diffusion (e), and coronal fluid-attenuated inversion recovery (f) show diffuse and confluent white matter hyperintensity (marked by arrow) surrounding the giant perivascular spaces (arrow head). There is associated gyral expansion over the perivascular spaces with no enhancement. The SWI shows no hemorrhage or calcifications. There is no diffusion restriction (e) or post-contrast enhancement (c).
The lesion remained stable on follow-up imaging and was labeled as giant TPVS.
DISCUSSION
In one of the largest retrospective studies of 37 patients with giant TPVSs, lesions were predominant in male patients (1.8:1) and ranged from 6 to 48 years in age.[
Commonly, perivascular spaces present near the lenticulostriate arteries.[
Due to their compressive nature, these lesions may be mistaken for cystic neoplastic conditions.[
For clinical management, dilated TPVS are subdivided into categories according to their anatomical relationship with penetrating arteries.[
CONCLUSION
Giant TPVS may be mistaken for other pathological processes, particularly due to their rarity in pediatric patients. However, pathognomonic radiological characteristics can help identify cases that may not require surgical intervention.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Bayram E, Akinci G, Topcu Y, Cakmakci H, Giray O, Ercal D. Multi-cystic white matter enlarged virchow robin spaces in a 5-year-old boy. Childs Nerv Syst. 2012. 28: 743-6
2. Fujimoto K, Kuroda JI, Hide T, Hasegawa Y, Yano S, Kuratsu JI. Giant tumefactive perivascular spaces that expanded and became symptomatic 14 years after initial surgery. Surg Neurol Int. 2012. 3: 127
3. Groeschel S, Chong WK, Surtees R, Hanefeld F. Virchow-robin spaces on magnetic resonance images: Normative data, their dilatation, and a review of the literature. Neuroradiology. 2006. 48: 745-54
4. Heier LA, Bauer C, Schwartz L, Zimmerman R, Morgello S, Deck M. Large virchow-robin spaces: MR-clinical correlation. Am J Neuroradiol. 1989. 10: 929-36
5. Kwee RM, Kwee TC. Virchow-robin spaces at MR imaging. Radiographics. 2007. 27: 1071-86
6. Mascalchi M, Salvi F, Godano U, Nistri M, Taiuti R, Tosetti M. Expanding lacunae causing triventricular hydrocephalus: Report of two cases. J Neurosurg. 1999. 91: 669-74
7. Pollock H, Hutchings M, Weller R, Zhang ET. Perivascular spaces in the basal ganglia of the human brain: Their relationship to lacunes. J Anat. 1997. 191: 337-46
8. Salzman KL, Osborn AG, House P, Jinkins JR, Ditchfield A, Cooper JA. Giant tumefactive perivascular spaces. Am J Neuroradiol. 2005. 26: 298-305
9. Sankararaman S, Velayuthan S, Ambekar S, Gonzalez-Toledo E. Giant tumefactive perivascular spaces: A further case. J Pediatr Neurosci. 2013. 8: 108-10
10. Sato N, Sze G, Awad IA, Putman CM, Shibazaki T, Endo K. Parenchymal perianeurysmal cystic changes in the brain: Report of five cases. Radiology. 2000. 215: 229-33
11. Song CJ, Kim JH, Kier EL, Bronen RA. MR imaging and histologic features of subinsular bright spots on T2-weighted MR images: Virchow-robin spaces of the extreme capsule and insular cortex. Radiology. 2000. 214: 671-7
12. Stephens T, Parmar H, Cornblath W. Giant tumefactive perivascular spaces. J Neurol Sci. 2008. 266: 171-3
13. Woo PY, Cheung E, Zhuang JT, Wong HT, Chan KY. A giant tumefactive perivascular space: A rare cause of obstructive hydrocephalus and monoparesis. Asian J Neurosurg. 2018. 13: 1295-300