- Department of Neurosurgery, Azienda Sanitaria Universitaria Integrata di Udine, Udine, Italy
- Department of Neuroradiology, Azienda Sanitaria Universitaria Integrata di Udine, Udine, Italy
Correspondence Address:
Gabriele Capo
Department of Neurosurgery, Azienda Sanitaria Universitaria Integrata di Udine, Udine, Italy
DOI:10.4103/sni.sni_170_17
Copyright: © 2018 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Gabriele Capo, Maria C. Vescovi, Giovanni Toniato, Benedetto Petralia, Vladimir Gavrilovic, Miran Skrap. Giant vertebral aneurysm: A case report detailing successful treatment with combined stenting and surgery. 16-Jan-2018;9:6
How to cite this URL: Gabriele Capo, Maria C. Vescovi, Giovanni Toniato, Benedetto Petralia, Vladimir Gavrilovic, Miran Skrap. Giant vertebral aneurysm: A case report detailing successful treatment with combined stenting and surgery. 16-Jan-2018;9:6. Available from: http://surgicalneurologyint.com/surgicalint-articles/giant-vertebral-aneurysm-a-case-report-detailing-successful-treatment-with-combined-stenting-and-surgery/
Abstract
Background:Giant aneurysms (>25 mm) arising from the vertebral artery (VA) often present with slow progression of symptoms and signs because of gradual brainstem and cranial nerve compression. The underlying pathophysiology is not well understood, and treatment, wherever possible, is tailored to each singular case. Endovascular management does not usually solve the problem of mass compression, whereas surgical treatment involves several complications.
Case Description:A 58-year-old woman presented with a continuously growing giant right VA aneurysm, partially thrombosed, even after endovascular treatment (placement of two diversion flow stents). Operative partial aneurysmectomy and intraoperative placement of an endovascular balloon allowed removal from circulation without significant bleeding with a good neurological outcome.
Conclusions:The variability of VA thrombosed giant aneurysms implies a customized therapeutic strategy. Combined endovascular techniques and surgical clipping allow safe and successful trapping and aneurysmectomy. This case highlights the benefits of treating similar pathologies with a combination of both techniques.
Keywords: Endovascular treatment, fenestrated clip, flow-diverter, giant aneurysm, neurovascular surgery, temporary balloon occlusion, vertebral artery
INTRODUCTION
Intracranial aneurysms (IAs) with a diameter of >25 mm arising from the vertebral artery (VA) are rare, representing 4–6% of all intracranial giant aneurysms.[
CASE DESCRIPTION
History
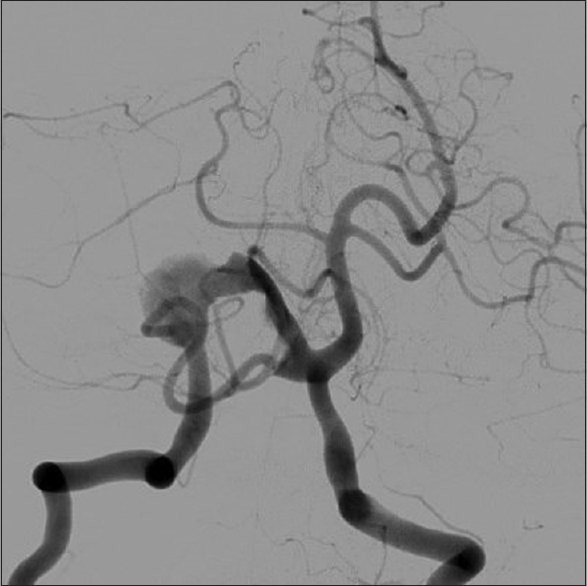
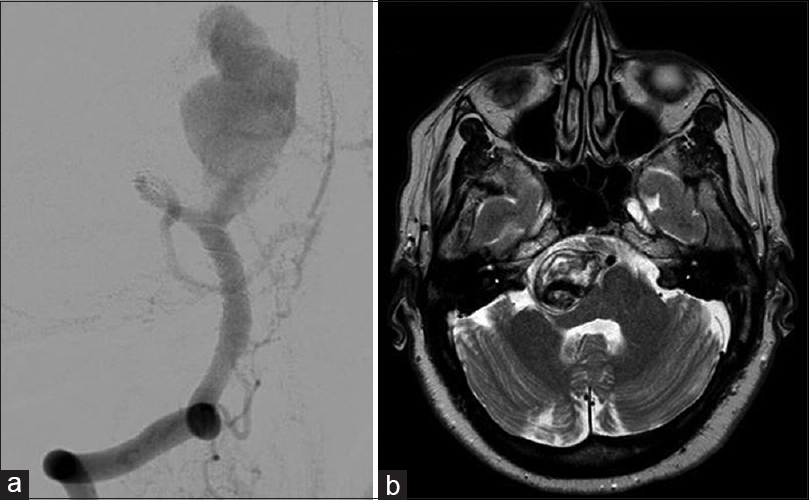
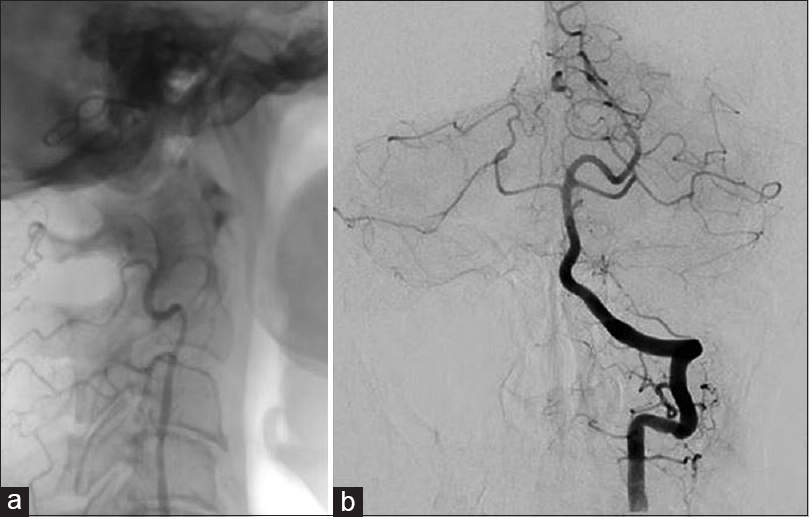
A 58-year-old woman presented in another institution with complaints of headache, dizziness, bilateral arm paresthesia, and visual disturbance. Her neurological examination showed abducens nerve palsy. A computerized tomography scan (angio-CT), and later cerebral angiography, displayed a giant partially thrombosed right VA (V4) dissecting aneurysm [
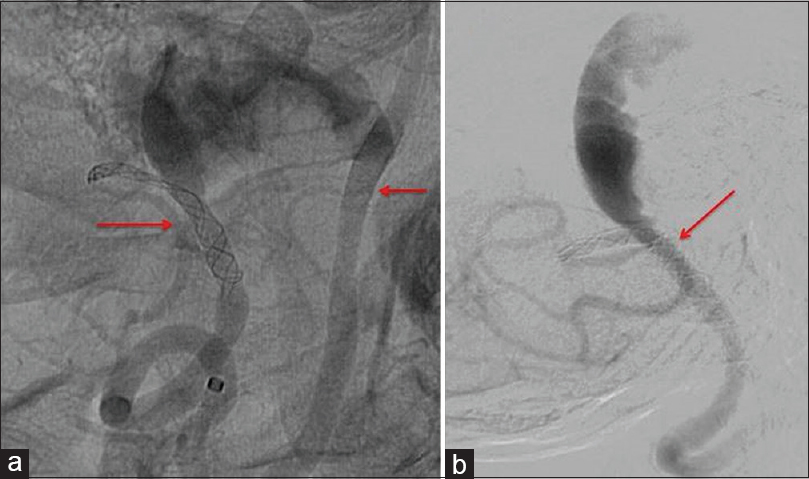
After beginning antiplatelet therapy, the patient underwent two stent insertions: the first PED 3 × 14 (Medtronic/Covidien, Irvine, CA) in the left VA-basilar junction to exclude the dissecting segment of the right VA, and the second Silk 3 × 18 in the right VA-PICA junction (Balt Extrusion, Montmorency, France). Before discharge, VA angiograms demonstrated correct placement of diversion flow stents [
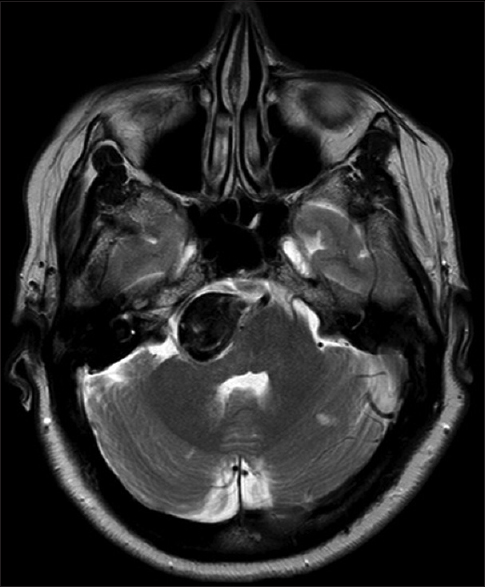
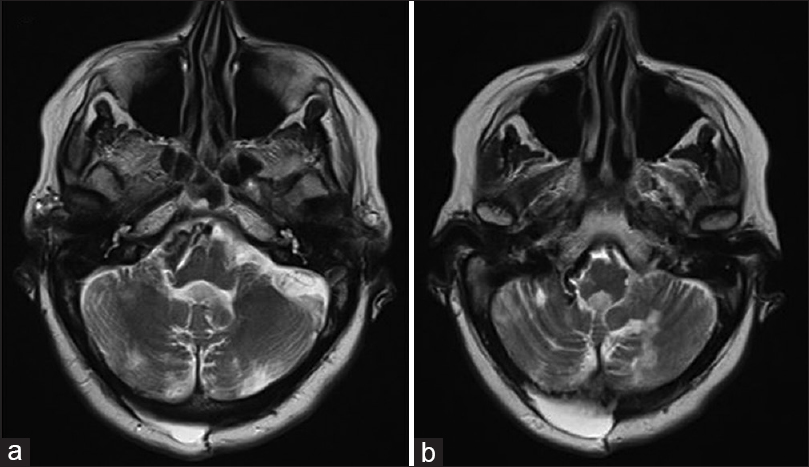
Unfortunately, a subsequent magnetic resonance imaging (MRI) and angiogram [
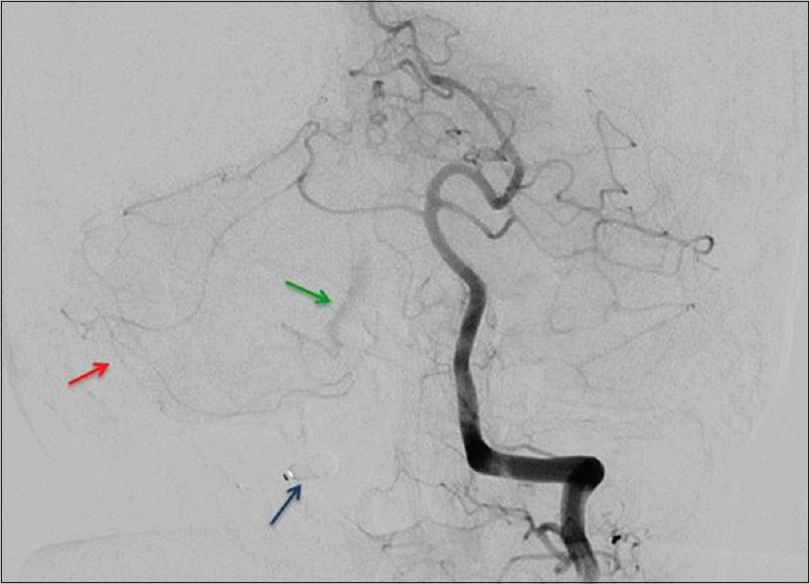
The patient was referred to our neurosurgical center 1 year after the stenting, with a 6-month history of worsening spastic paraparesis, mild swallowing disturbance, light dysphonia, and diplopia. A right and left vertebral angiogram with an occlusion test [
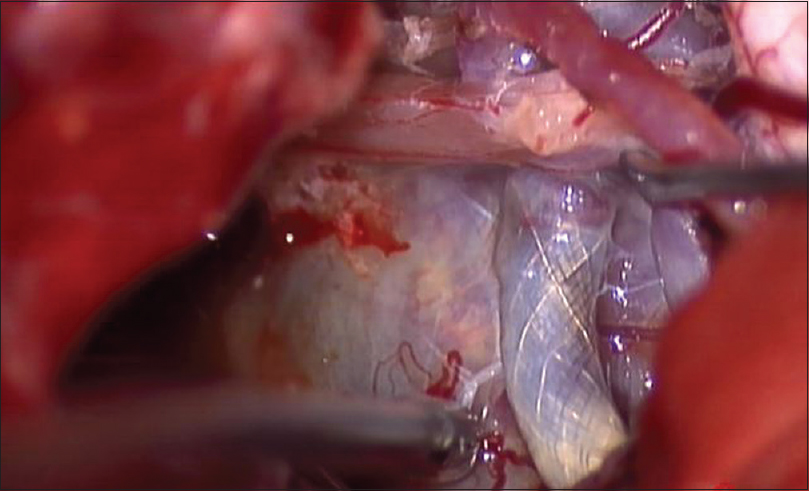
On second admission, the patient underwent a right suboccipital craniectomy followed by partial aneurysmectomy. The surgical procedure was preceded by the placement of a nondetachable flow dependent balloon catheter (Balt B1, Balt Extrusion, France) to be inflated during reduction of the intra-aneurysmal thrombosis and clip positioning. The exact position and the inflation and deflation volume of the balloon were carefully checked several times. A 3,000 unit of heparin bolus was injected at the beginning of the procedure. The catheter had been successively washed with heparinized saline solution (5,000 units/l). When we were sure of the correct position and the adequate volume of balloon inflation, we moved the patient with the complete arterial femoral system to the neurosurgical theatre.[
Operation
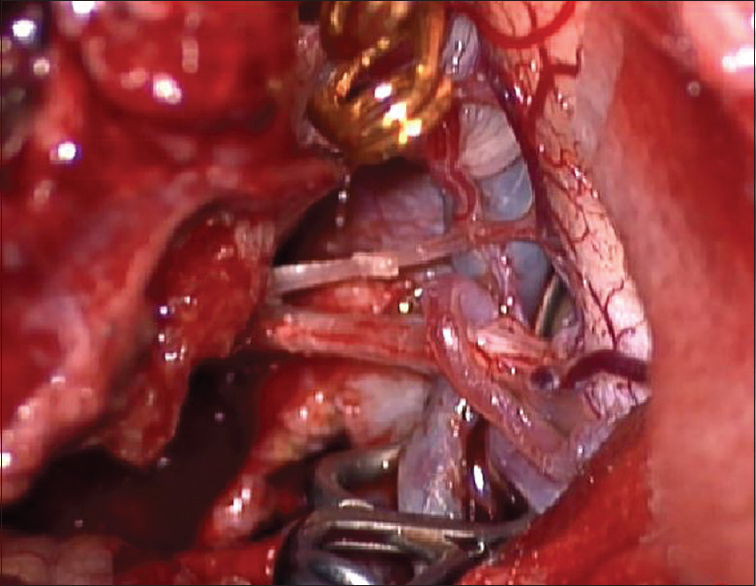
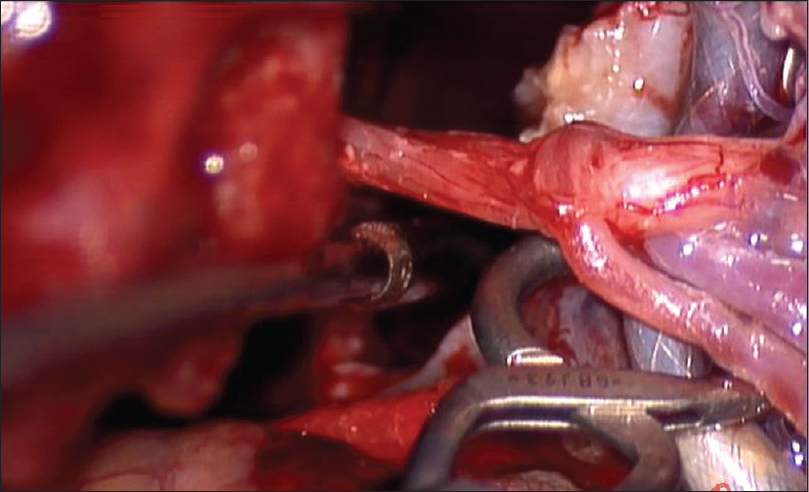
We performed a right suboccipital craniotomy with the patient under general anesthesia along with neurophysiological monitoring. An endotracheal tube with electrodes was used for monitoring the vocal cords. Upon opening the cisterna magna, the segment of VA-PICA containing the flow diversion stent was exposed. The aneurysmal sac was identified and isolated from the surrounding tissue and cranial nerves [
Postoperative course
After a transient admission to the intensive care unit, the patient was transferred to the ward. A postoperative CT scan showed no hemorrhage or ischemia in the posterior fossa. During the postoperative course, the patient experienced temporary mild worsening of her dysphonia and dysphagia, a new development of right partial palsy of the VII cranial nerve palsy (III HB) and left superior arm thermal hypoesthesia. Laryngeal endoscopy highlighted a right partial vocal cord paresis with only mild dysphagia. There was no marked effect on her gait disturbance. Speech and rehabilitation therapy was scheduled.
An angiography performed 10 days later showed complete resolution of the aneurysm patency, however, unfortunately also the PICA occlusion even if without sequelae. MRI confirmed an initial reduction of mass effect on the brainstem and cerebellum and no evidence of PICA distribution ischemia. However, the patient's condition improved gradually; she was discharged 18 days after the surgery to a rehabilitation unit.
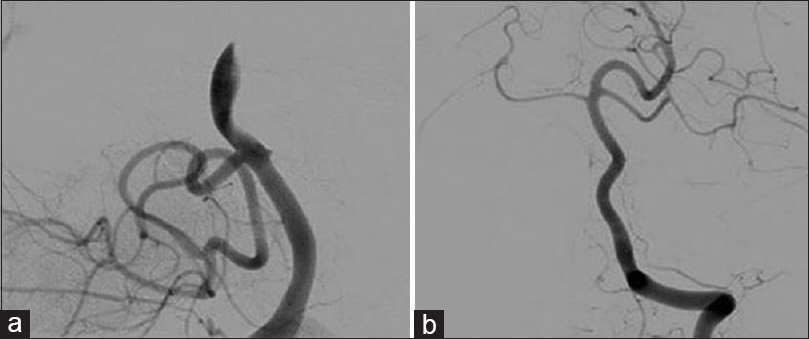
After 4 months, the patient experienced a progressive and almost complete recovery. Now she ambulates independently without assistance, has a normal diet, and has right HB 1 facial paresis. Follow-up angiography and MRI were performed at 4 months after the surgery. The angiogram confirmed the right PICA occlusion and the exclusion of the aneurysm from the circulation [
DISCUSSION
In the case of giant VA aneurysms, symptoms commonly result from the compression of neuronal structures, ischemic stroke, or aneurysm rupture. Most cases reported in the literature describe a progressive enlargement with the deterioration of symptoms, even after repeated endovascular treatments. Although this enlargement phenomenon has been extensively studied, there is no consensus regarding the mechanisms underlying the increase of almost completely thrombosed giant aneurysms presenting as mass lesions.[
The treatment of similar cases is scarcely reported in the literature with every case presenting its own unique features. Surgery may represent too high a risk, but on the other hand, embolization alone does not guarantee the reduction of the mass effect. Quite often, the opposite result is obtained, with the enlarging of the sac and it is necessary to adopt different customized approaches to solve the problem.[
Attempts to treat patients with giant vertebrobasilar junction aneurysm with a combination of flow diversion and bypass surgery have been chronicled previously.[
Balloon assistance surgery is not frequently used though it has been described since 1993.[
Our patient presented with a giant VA aneurysm that continued to grow even after proximal and distal endovascular treatment. The proximal stent maintained flow patency to PICA, but did not avoid sac-filling; conversely, the distal stent successfully excluded the back flow from the basilar artery. It was not sufficient to stop the aneurysm growing and leave the compression on the brainstem. In our case, combined use of the neuroradiological and surgical treatments overcame the problem. The availability of the two stents has proven to be very useful to the surgeon. The proximal stent on the PICA allowed safe manipulation[
An alternative treatment could be a bypass surgery. Some authors would have preferred a proximal occlusion and anastomosis. During the planning of the operation, we did not consider a bypass to the distal part of the PICA considering that the reinforced vessel would facilitate the clipping without delayed complications and because of the uneventful temporary balloon occlusion of the VA which shows collateral vessels supplying the territory of the PICA. Hence, we decided to avoid additional difficulties to this procedure and to use fenestrated clip.
Nevertheless, we did not avoid PICA occlusion. We did not check the flow with micro-Doppler Ultrasound or ICG angiography during the surgery because we were very confident of the vessel patency as it appeared along a quite long segment. However, we did not think that we would be able to significantly modify the clip position in case of flow reduction. In that situation, the only other option we would have considered was an anastomosis if feasible under the circumstances. Furthermore, we did not use antiplatelet therapy and antivasospastic agent, which could avoid this complication, considering the good postoperative course.
Indeed, the patient did not experience neurological deficits and had good collateral flow compensation as postoperative angiography showed. After 4 months, she reached an almost complete satisfactory recovery
CONCLUSION
In the case of a giant partially thrombosed aneurysm with thick wall and brain compression, aneurysmectomy should be considered as the goal. The challenging case presented here clearly illustrates the usefulness of an endovascular-neurosurgical combined strategy to treat such cases. Using only one of these approaches could increase the risks of treatment, and might not solve the problem.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient has given her consent for her images and other clinical information to be reported in the journal. The patient understands that name and initial will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Bailes JE, Deeb ZL, Wilson JA, Jungreis CA, Horton JA. Intraoperative angiography and temporary balloon occlusion of the basilar artery as an adjunct to surgical clipping: Technical note. Neurosurgery. 1992. 30: 949-53
2. Chow M, McDougall C, O’Kelly C, Ashforth R, Johnson E, Fiorella D. Delayed spontaneous rupture of a posterior inferior cerebellar artery aneurysm following treatment with flow diversion: A clinicopathologic study. Am J Neuroradiol. 2012. 33: E46-51
3. Cikla U, Uluc K, Baskaya MK. Microsurgical clipping of a giant vertebrobasilar junction aneurysm under hypothermic circulatory arrest. Neurosurg Focus. 2015. 39: V13-
4. Fox B, Humphries WE, Doss VT, Hoit D, Elijovich L, Arthur AS. Rupture of giant vertebrobasilar aneurysm following flow diversion: Mechanical stretch as a potential mechanism for early aneurysm rupture. BMJ Case Rep. 2014. 2014: 1-4
5. Hampton T, Walsh D, Tolias C, Fiorella D. Mural destabilization after aneurysm treatment with a flow-diverting device: A report of two cases. J Neurointerv Surg. 2011. 3: 167-71
6. Iihara K, Murao K, Sakai N, Soeda A, Ishibashi-Ueda H, Yutani C. Continued growth of and increased symptoms from a thrombosed giant aneurysm of the vertebral artery after complete endovascular occlusion and trapping: The role of vasa vasorum. Case report. J Neurosurg. 2003. 98: 407-13
7. J-O’Shanahan A, Noda K, Tsuboi T, Ota N, Kamiyama H, Tokuda S. Radical surgical treatment for recurrent giant fusiform thrombosed vertebral artery aneurysm previously coiled. Surg Neurol Int. 2016. 7: S237-42
8. Kalani MYS, Zabramski JM, Nakaji P, Spetzler RF. Bypass and flow reduction for complex basilar and vertebrobasilar junction aneurysms. Neurosurgery. 2013. 72: 763-75
9. Kan P, Srinivasan VM, Mbabuike N, Tawk RG, Ban VS, Welch BG. Aneurysms with persistent patency after treatment with the Pipeline Embolization Device. J Neurosurg. 2017. 126: 1894-8
10. Kimura T, Kin T, Shojima M, Morita A. Clip reconstruction of giant vertebral artery aneurysm after failed flow reduction therapy. Neurosurg Focus. 2015. 39: V5-
11. Mizoi K, Yoshimoto T, Takahashi A, Ogawa A. Direct clipping of basilar trunk aneurysms using temporary balloon occlusion. J Neurosurg. 1994. 80: 230-6
12. Mizoi K, Takahashi A, Yoshimoto T, Fujiwara S, Koshu K. Combined Endovascular and Neurosurgical Approach for Paraclinoid Internal Carotid Artery Aneurysms. Neurosurgery. 1993. 33: 986-92
13. Nagahiro S, Takada A, Goto S, Kai Y, Ushio Y. Thrombosed growing giant aneurysms of the vertebral artery: Growth mechanism and management. J Neurosurg. 1995. 82: 796-801
14. Pahl FH, Vellutini EDAS, Capel Cardoso AC, De Oliveira MF. Vasa Vasorum and the Growing of Thrombosed Giant Aneurysm of the Vertebral Artery: A Case Report. World Neurosurg. 2016. 85: 368.e1-4
15. Petralia B, Skrap M. Temporary balloon occlusion during giant aneurysm surgery-A technical description. Interv Neuroradiol. 2006. 12: 245-50
16. Report C, Skrap M, Petralia B, Toniato G. The combined treatment of stenting and surgery in a giant unruptured aneurysm of the middle cerebral artery. Surg Neurol Int. 2015. p. 6-
17. Ricci G, Ricci A, Gallucci M, Zotta D, Scogna A, Costagliola C. Combined endovascular and microsurgical approach in the treatment of giant paraclinoid and vertebrobasilar aneurysms. J Neurosurg Sci. 2005. 49: 1-6
18. Schucart WA, Kwan ES, Heilman CB. Temporary balloon occlusion of a proximal vessel as an aid to clipping aneurysms of the basilar and paraclinoid internal carotid arteries: Technical note. Neurosurgery. 1990. 27: 116-9
19. Skrap M, Petralia B, Toniato G. Temporary balloon occlusion during the surgical treatment of giant paraclinoid and vertebrobasilar aneurysms. Acta Neurochir (Wien). 2010. 152: 435-42