- Department of Neurosurgery, Liaquat National Hospital, Karachi, Pakistan
- Department of Neurosurgery, Memon Medical Institute Hospital, Karachi, Pakistan.
Correspondence Address:
Syeda Alisha Ali Zaidi, Department of Neurosurgery, Liaquat National Hospital, Karachi, Pakistan.
DOI:10.25259/SNI_815_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Syeda Alisha Ali Zaidi1, Amanullah1, Syeda Kubra Kishwar Jafri2, Salman Sharif1. Glioblastoma multiforme at internal auditory canal. 06-Jan-2023;14:2
How to cite this URL: Syeda Alisha Ali Zaidi1, Amanullah1, Syeda Kubra Kishwar Jafri2, Salman Sharif1. Glioblastoma multiforme at internal auditory canal. 06-Jan-2023;14:2. Available from: https://surgicalneurologyint.com/surgicalint-articles/12098/
Abstract
Background: Glioblastomas are the most common adult primary brain tumor present supratentorially. The presence of true extra-axial GBM infratentorially, especially in the internal auditory canal, is extremely rare with only three cases reported previously in the literature. We report the fourth case of primary internal auditory canal/cerebellopontine angle (CPA) glioblastoma which initially mimicked vestibular schwannoma on the basis of its location and presentation.
Case Description: A 65-year-old male presented with headache, vertigo, and progressive right ear deafness for 5 months. His preoperative magnetic resonance imaging findings were consistent with vestibular schwannoma. Maximum safe resection (near total) was done. The final histopathology report showed glioblastoma multiforme.
Conclusion: As per our knowledge, this is the fourth reported case of an extra-axial VIII cranial nerve glioblastoma located in internal auditory canal. Hence, despite being very rare, they should be considered as a differential in tumors at CPA.
Keywords: Glioblastoma, Internal acoustic meatus, Cerebellopontine angle, Vestibular schwannoma
INTRODUCTION
Glioblastomas are the most common adult malignant primary brain tumors arising from the glial cells. They are characteristically present in the supratentorial cerebral hemispheres and account for almost 20% of all intracranial tumors.[
CASE PRESENTATION
This is the case of a 65-year-old male, with no known comorbid, who presented through outpatient clinic with the complaints of right-sided deafness, peripheral vertigo associated with vomiting, and progressive headache for the past 5 months. Symptoms had become severe over the past 2 months making him completely deaf in the right ear. There was no complaint of altered level of consciousness, fits, falls, no visual symptoms, or facial weakness. His neurologic examination revealed a horizontal left beating nystagmus, right-sided sensorineural hearing loss, and no other cranial nerve dysfunction, however, Romberg’s test was positive with impaired tandem walking.
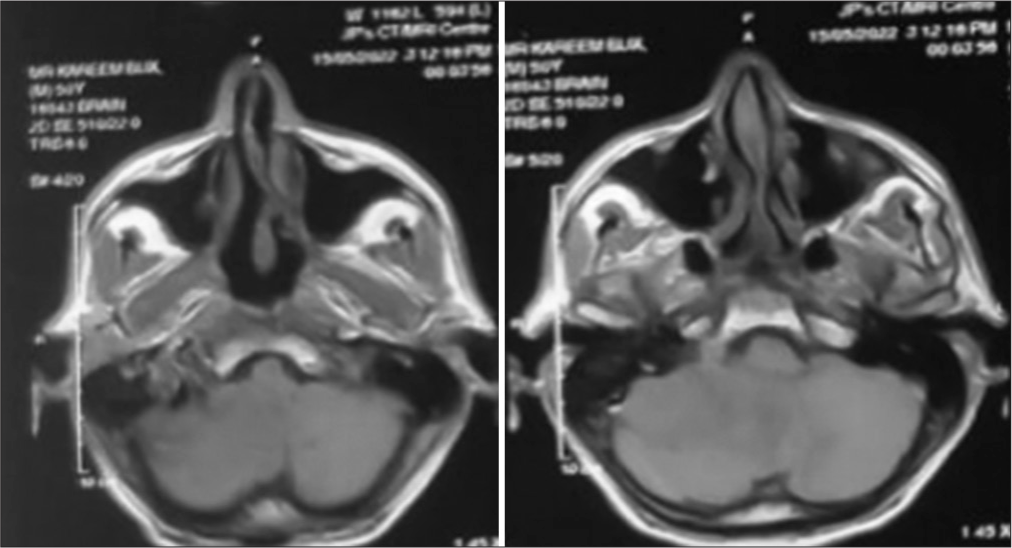
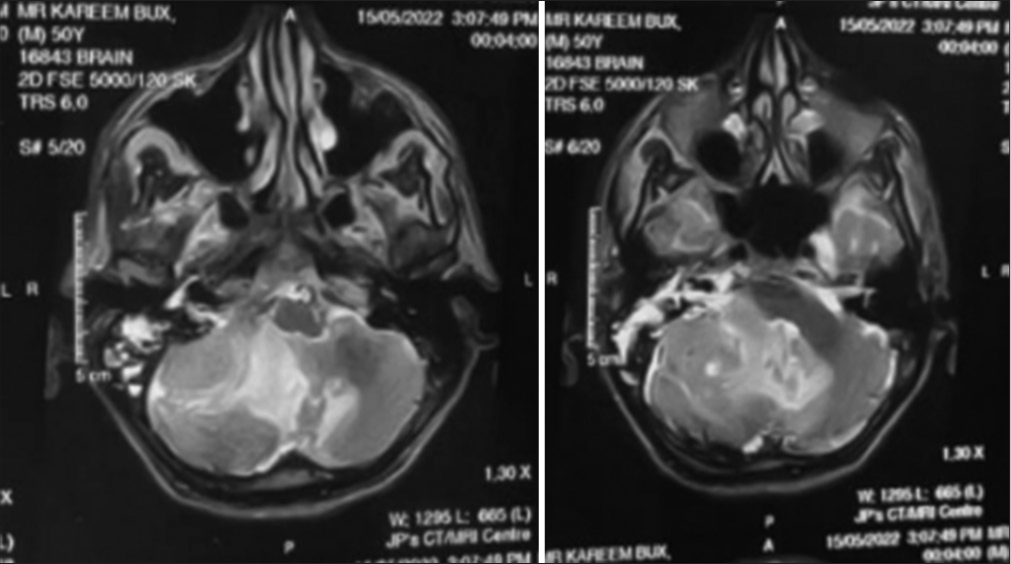
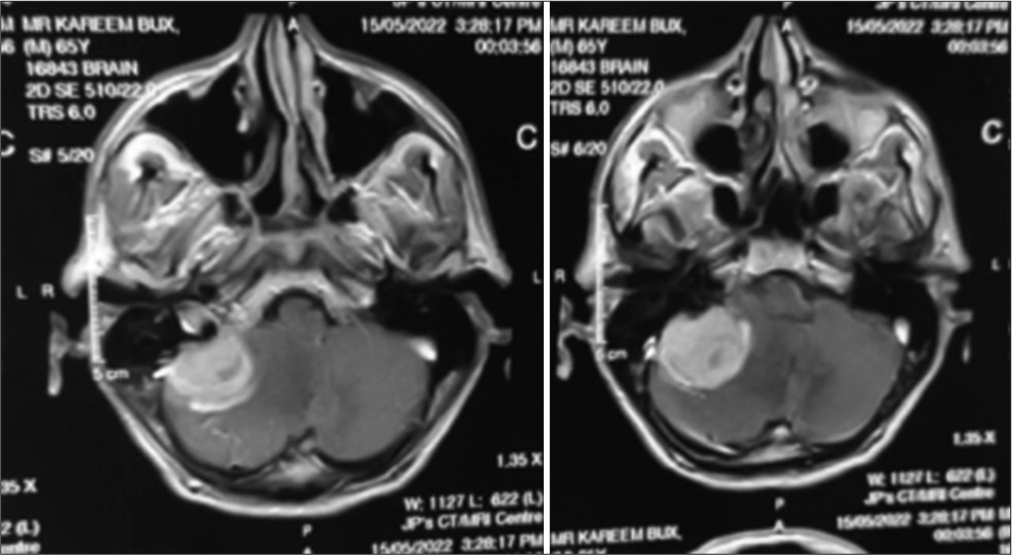
Magnetic resonance imaging (MRI) of the brain with contrast showed a lesion in the right CPA, hypointense on T1-weighted sequences [
Operative course
We positioned the patient in a park bench position with the right side facing up. After fixing the patient’s head in the Mayfield head clamp, retrosigmoid craniotomy was done and surgery progressed in a usual fashion. The cerebellomedullary cistern was opened followed by drainage of cerebrospinal fluid. Cerebellum was retracted with the help of a Duro retractor and the tumor was identified. It was encapsulated and firm in consistency. The cerebellar invasion was evident along the medial aspect along with dense adhesions between the capsule and lower cranial nerves. After taking a biopsy, intracapsular tumor debulking was done with the help of an ultrasonic aspirator and near-total resection was achieved with the possible removal of all visible tumor mass.
Postoperative course
Postoperatively, the patient remained stable with no new neurology. His central nervous system examination was the same as the preoperative examination, and hence, he was discharged and followed up as an outpatient in the clinic. Histopathology revealed fragments of neoplastic lesions showing dense cellularity composed of markedly atypical cells with abundant eosinophilic cytoplasm and pleomorphic enlarged nuclei with vesicular chromatin and prominent nucleoli. Patchy areas of palisading necrosis were also seen. Few gemistocytic cells were seen. Mitosis of approximately 3–4/10 HPF was noted.
Immunohistochemical studies were performed by DAKO envision method using the following antibodies:
GFAP patchy +ve S100 patchy +ve EMA –ve CKAE1/AE3 −ve
Ki67 approximately 8–10 % proliferative index.
All of these features favored the diagnosis of glioblastoma WHO Grade-IV.
The patient was then referred to a neuro-oncologist for further management, however, because of the poor prognosis, the patient refused further management and follow-up. On calling, after 2 months, we came to know that after remaining symptom free for about 1 month, the patient had started to have symptoms of vertigo and severe gait imbalance.
DISCUSSION AND LITERATURE REVIEW
Glioblastomas are WHO Grade IV primary intracranial tumors arising from glial cells. They commonly tend to occur in the supratentorial region, especially in frontal lobes, however, in very rare cases, gliomas (WHO Grades I–IV) may arise from other cortical areas such as brainstem cerebellum and spinal cord, in very rare instances, they arise from cranial nerves.[
Glioblastoma is classically hypointense to isointense, with a ring-enhancing pattern on postgadolinium T1 images. It is hyperintense on both T2 and FLAIR images. Magnetic resonance spectroscopy (MRS), showing larger choline, creatinine and lactate peaks, and a shallow N-acetylaspartate peak, is also used sometimes and is a more visionary imaging tool as it can aid in differentiating benign from malignant tumors.[
The current treatment of all newly diagnosed GBM patients is cytoreductive surgery (>98%), followed by the Stupp regimen (fractionated focal irradiation in daily fractions of 2 Gy given 5 days/week for 6 weeks (a total of 60 Gy), plus concomitant daily temozolomide (75 mg/m2/day, 7 days/week from the first to the last day of radiotherapy), followed by six cycles of adjuvant temozolomide (150–200 mg/m2/day for 5 days during each 28-day cycle).[
However, the current treatment options for vestibular schwannoma include observation, surgical resection, fractionated radiotherapy, and radiosurgery. Small, asymptomatic, or incidental tumors undergo a trial of observation versus radiosurgery whereas large tumors undergo surgical decompression, followed by fractionated radiotherapy or radiosurgery [
It has been reported that the overall survival of patients with newly diagnosed glioblastoma multiforme (GBM) is 17–30% at 1 year and only 3–5% at 2 years, despite access to state-of-the-art modalities of therapy. Young age and high KPS (Karnofsky Performance Scale) are the strongest positive predictors of survival.[
We additionally found a total of 14 overall reports of the tumors at a CPA out of which eight cases turned out to be low-grade gliomas, these eight cases were pilocytic and fibrillary astrocytomas mostly in children. The remaining five were tumors of unknown origin.[
Following diagnostic confirmation, through histopathology, all patients were advised to undergo adjuvant therapy. Those who underwent chemo and radiotherapy had a symptom-free span of around 6 months–1 year, while patients who refused any further management survived for around 1–2 months without recurrence of symptoms.[
CONCLUSION
There have been multiple cases reported about the presence of glioblastoma at the CPA which mimicked vestibular schwannoma or meningioma due to their location and presentation on MRI. MRS may help in differentiating between benign and malignant tumors. These tumors should be followed aggressively to detect any suspicious malignant activity to treat them early and prevent or delay the recurrence to provide maximum symptom-free survival.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Adamson C, Kanu OO, Mehta AI, Di C, Lin N, Mattox AK. Glioblastoma multiforme: A review of where we have been and where we are going. Expert Opin Investig Drugs. 2009. 18: 1061-83
2. Arnautovic KI, Husain MM, Linskey M.E. Cranial nerve root entry zone primary cerebellopontine angle gliomas: A rare and poorly recognized subset of extraparenchymal tumors. J Neurooncol. 2000. 49: 205-12
3. . Available from: https://www.radiopaedia.org/articles/vestibularschwannoma#nav_clinical-presentation [Last accessed on September 2022 Sep 04].
4. Beutler AS, Hsiang JK, Moorhouse DF, Hansen LA, Alksne JF. Pilocytic astrocytoma presenting as an extra-axial tumor in the cerebellopontine angle: Case report. Neurosurgery. 1995. 37: 125-8
5. Cushing H, editors. Tumors of the Nervus Acusticus and the Syndrome of the Cerebellopontine Angle. Philadelphia, PA: WB Saunders; 1917. p.
6. Duan H, Kitazawa K, Yako T, Ichinose S, Kobayashi S, Sudo M. Gliosarcoma in the cerebellopontine angle with rapid tumor growth and intratumoral hemorrhage. World Neurosurg. 2016. 92: 580.e17-21
7. Forton G, Verlooy J, Cras P, Parizel P, Van de Heyning P. Problems with flute playing: An otological problem? Case report of a peculiar cerebellar astrocytoma. Acta Otorhinolaryngol Belg. 1992. 46: 405-10
8. Goldbrunner R, Weller M, Regis J, Lund-Johansen M, Stavrinou P, Reuss D. EANO guideline on the diagnosis and treatment of vestibular schwannoma. Neuro Oncol. 2020. 22: 31-45
9. Hasegawa T, Kida Y, Kobayashi T, Yoshimoto M, Mori Y, Yoshida J. Long-term outcomes in patients with vestibular schwannomas treated using gamma knife surgery: 10-year follow up. J Neurosurg. 2005. 102: 10-6
10. Kasantikul V, Palmer JO, Netsky MG, Glasscock ME, Hays JW. Glioma of the acoustic nerve. Arch Otolaryngol. 1980. 106: 456-9
11. Lee JH, Kim JH, Kwon TH. Primary glioblastoma of the cerebellopontine angle: Case report and review of the literature. J Korean Neurosurg Soc. 2017. 60: 380-4
12. Matsuda M, Onuma K, Satomi K, Nakai K, Yamamoto T, Matsumura A. Exophytic cerebellar glioblastoma in the cerebellopontine angle: Case report and review of the literature. J Neurol Surg Rep. 2014. 75: e67-72
13. Mirone G, Schiabello L, Chibbaro S, Bouazza S, George B. Pediatric primary pilocytic astrocytoma of the cerebellopontine angle: A case report. Childs Nerv Syst. 2009. 25: 247-51
14. Panse R. An acoustician’s Glioma (Ein Gliom des Akustikus). Arch Ohr Heilk. 1904. 61: 251-5
15. Swaroop GR, Whittle IR. Exophytic pontine glioblastoma mimicking acoustic neuroma. J Neurosurg Sci. 1997. 41: 409-11
16. Takada Y, Ohno K, Tamaki M, Hirakawa K. Cerebellopontine angle pilocytic astrocytoma mimicking acoustic schwannoma. Neuroradiology. 1999. 41: 949-50
17. Takami H, Prummer CM, Graffeo CS, Peris-Celda M, Giannini C, Driscoll Cl. Glioblastoma of the cerebellopontine angle and internal auditory canal mimicking a peripheral nerve sheath tumor: Case report. J Neurosurg. 2019. 131: 1835-9
18. Wang Y, Zhang J, Li W, Jiang T, Qi S, Chen Z. Guideline conformity to the Stupp regimen in patients with newly diagnosed glioblastoma multiforme in China. Future Oncol. 2021. 17: 4571-82
19. Wu B, Liu W, Zhu H, Feng H, Liu J. Primary glioblastoma of the cerebellopontine angle in adults. J Neurosurg. 2011. 114: 1288-93
20. Yang DX, Jing Y, Xu ZM, Yuan F, Liu YL, Wang GS. Primary glioblastoma of the cerebellopontine angle in an adult mimicking an acoustic neuroma. World Neurosurg. 2019. 122: 48-52