- Departement of Pediatric Neurosurgery, Craniofacial Unit, Necker Enfants Malades Hospital, APHP, Paris, France
- Operative Unit of Neurosurgery, Bellaria Hospital, Bologna, Italy
- Department of Anesthesia, Necker Enfants Malades Hospital, APHP, Paris, France
- Department of Neuroradiology, Necker Enfants Malades Hospital, APHP, Paris, France
Correspondence Address:
Federico Di Rocco
Departement of Pediatric Neurosurgery, Craniofacial Unit, Necker Enfants Malades Hospital, APHP, Paris, France
DOI:10.4103/sni.sni_13_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Francesca Nicolini, Eric Arnaud, Kenichi Usami, Antonio Vecchione, Francis Brunelle, Federico Di Rocco. Impact of extra-axial cerebrospinal fluid collection in frontal morphology after surgical treatment of scaphocephaly. 30-Oct-2018;9:215
How to cite this URL: Francesca Nicolini, Eric Arnaud, Kenichi Usami, Antonio Vecchione, Francis Brunelle, Federico Di Rocco. Impact of extra-axial cerebrospinal fluid collection in frontal morphology after surgical treatment of scaphocephaly. 30-Oct-2018;9:215. Available from: http://surgicalneurologyint.com/surgicalint-articles/9050/
Abstract
Background:Infants with sagittal suture synostosis often present a pathologic dilatation of subarachnoid spaces. The impact of such subarachnoid spaces’ enlargement in the morphology of the skull, especially on the forehead and on the surgical outcome, was analyzed.
Methods:Children less than 6 months of age undergoing a surgical correction of the scaphocephaly with Renier's H technique between 2003 and 2008 were included in the study. In these patients, preoperative and postoperative fronto-occipital diameter (FOD), biparietal diameter (BPD), temporal width (TW), and naso-frontal angle (NFA) were measured. Cranial index (CI) and the difference between preoperative and postoperative CI (ΔCI) were calculated. Preoperative cranio-cortical width (CCW) was measured to analyze the subarachnoid spaces’ volumes. The children here considered were then divided into two groups: Group 1 with CCW within normal estimated value corrected for age and Group 2 with CCW larger than estimated normal value.
Results:About 159 children were enrolled (72.3% male). CCW was larger than expected in 95 children (59.8%). A positive correlation between CCW and BPD (P ≤ 0.001) and a negative correlation between CCW and NFA (P ≤ 0.001) were found. When comparing the two groups, the mean age at preoperative computed tomography (CT) scan was 121 days in Group 1 and 110 days in Group 2. The mean age at operation was 130 days in Group 1 and 123 in Group 2. The mean age at postoperative examination (RX or CT scan) was 53.4 months in Group 1 and 51.8 months in Group 2. Preoperatively, the mean BPD, TW, and CI were significantly larger in Group 2 (P ≤ 0.01), whereas the NFA was significantly narrower (P = 0.03). Postoperative analysis showed that ΔCI was statistically different between the two groups (Group 1: 10%, Group 2: 7%; P
Conclusion:Two main subtypes of forehead of infants with scaphocephaly may be distinguished. Indeed, the morphology of the forehead differs when a pathologic subarachnoid spaces’ enlargement is present preoperatively and it also affects the postoperative evolution. Such observation highlights the importance of evaluating whether subarachnoid spaces are enlarged when planning a surgical correction in isolated sagittal suture synostosis.
Keywords: Craniostenosis, isolated, nonsyndromic, outcome, prognosis, sagittal suture, surgical correction, results
INTRODUCTION
Scaphocephaly by sagittal suture synostosis is the most common form of isolated craniosynostosis, accounting for 40%–60% of cases of craniosynostosis.[
The deformation may raise an aesthetic concern, and in some of patients an increased intracranial pressure (ICP) may be observed.[
There is a strong consensus on the time of surgery, with best functional and cosmetic results achieved within the first 6 months of life.[
When performed, neuroimaging investigations may find some associated features such as copper beaten skull, fingerprinting, and altered anatomy in venous drainage.[
To answer these questions, we analyzed the surgical results obtained in the surgical management of scaphocephaly at the pediatric Neurosurgical Department at Necker Enfants Malades in Paris.
PATIENTS AND METHODS
We retrospectively reviewed the medical files and imaging studies of patients with isolated sagittal synostosis treated at the Hôpital Necker Enfants Malades between 2003 and 2008 operated on with the same surgical technique (“Renier's H technique”)[
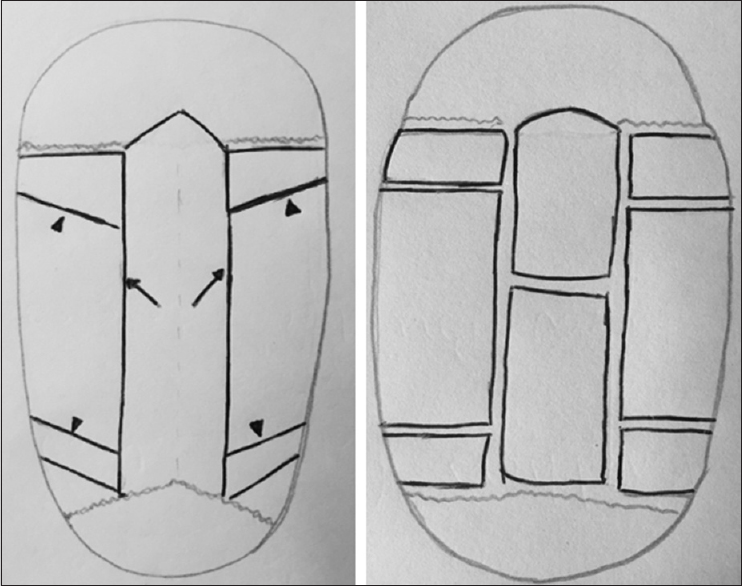
Figure 1
Left. Osteotomies in Renier's H technique. 4 cm-sagittal strip on the midline is generally performed (arrow) with four triangular retrocoronal wedges (two retrocoronal and two prelambdoid-head of arrows). Right. sagittal strip is removed and then shortened to adapt to new fronto-occipital diameter. Lateral bone flaps are “green-stick” fractured at the base
The following selection criteria were adopted: toddlers younger than 6 months of life and isolated sagittal suture synostosis, without intracranial anomalies other than an enlargement of subarachnoid spaces. Children with other cranio-facial malformations, multisutural synostosis, or brain anomalies were excluded. All patients operated on with a different surgical technique were excluded as well.
Data collection
For each patient, CT scan and cranial X-rays were collected. The following pre- and postoperative parameters were measured: naso-frontal angle (NFA), fronto-occipital diameter (FOD), biparietal diameter (BPD), and temporal width (TW) as a bipterional diameter (TW). All these parameters were calculated with Carestream Software® on preoperative CT scan.
FOD, BPD, and TW were measured in axial slices from one inner table to the other inner table and the NFA in the sagittal reconstruction of CT [
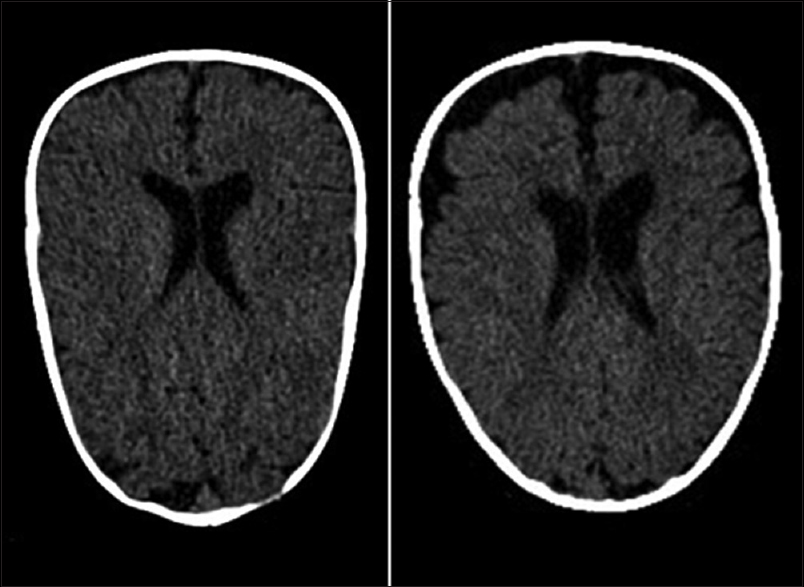
The dimension of subarachnoid spaces was calculated as cranio-cortical width (CCW), which is the widest vertical distance between brain surface and calvarium. The classical threshold of 4 mm adopted as physiological parameter according to Libicher et al.[
The patients were then divided into two groups: the first group (Group 1) was composed of infants with an enlargement of subarachnoid spaces that are smaller or equal than the threshold corrected for age (“physiological” enlargement) and the second group (Group 2) was composed of infants with a greater enlargement (“pathological” enlargement).
Statistical analysis
Data were collected using Microsoft Excel 2010®, and statistical analysis was performed with SPSS® and JMPPro 9 ® software. The t-test was used to compare the preoperative and postoperative measurements between groups. Kruskal–Wallis test was used to compare ΔCI. A significant level of P < 0.05 was considered for all analyses.
RESULTS
Data are summarized in
Population
Seventeen among the 176 patients who met the selection criteria had some incomplete imaging studies or were lost at follow-up; thus, finally 159 children were considered in this study.
There was a clear male predominance (115 patients, 72.3%) as expected. The median age at CT scan was 115 days (min. 82 days, max. 164 days). The median age at operation was 125.5 days (min. 82 days, max. 177 days). No perioperative differences were found during surgery within the two groups. In particular, no dural tears were recorded in this series in either group. The follow-up period ranged between 19 and 129 months.
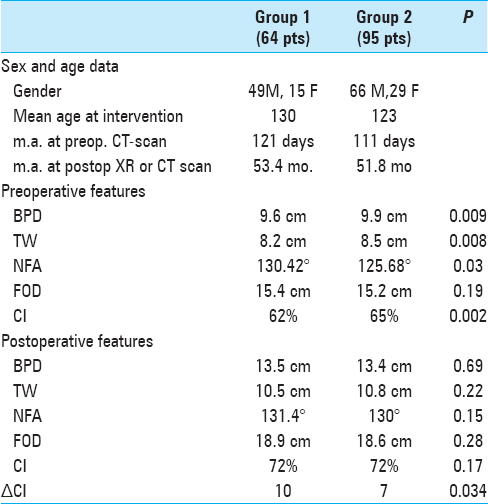
According to the enlargement of subarachnoid spaces, 64 patients (40.2%) were in Group 1 (“physiological”) and 95 patients (59.8%) in Group 2 (pathological) [
The two groups were comparable in terms of age at surgery and age at CT examination as well as age at postoperative control. The mean age at intervention was 130 days in Group 1 and 123 days in Group 2. The mean age at preoperative CT scan was 121 days in Group 1 and 111 days in Group 2. The mean age at postoperative RX or CT scan was 53.4 and 51.8 months for Groups 1 and 2, respectively.
Measurements
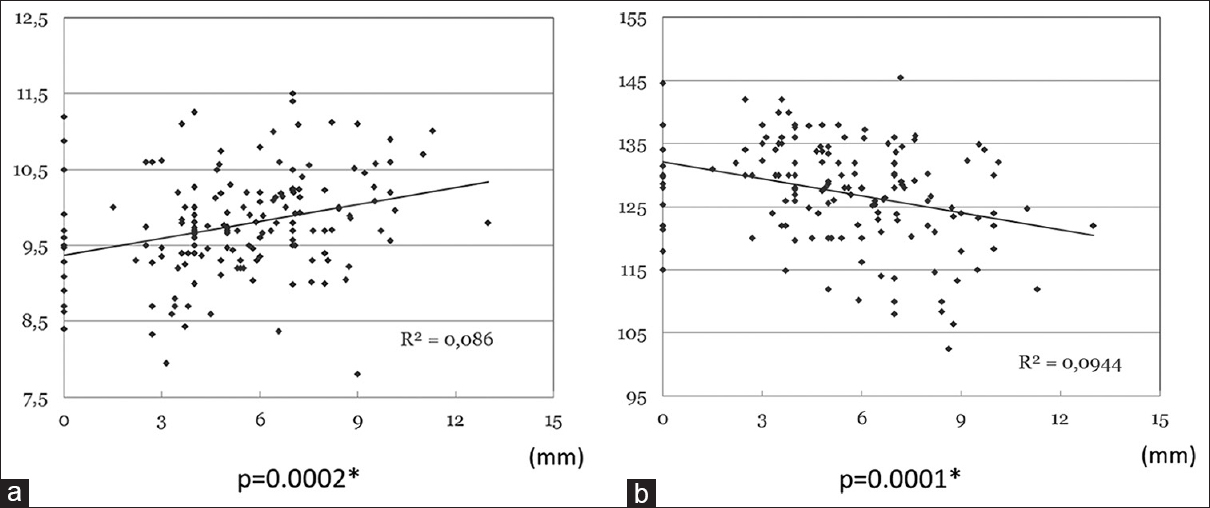
When considering the overall series, a positive correlation between CCW and BPD was found, R2 = 0.086, P = 0.0002) [
When distinguishing the two groups, the mean preoperative BPD and the TW were significantly larger in Group 2 (9.9 vs 9.6 mm, P = 0.009 for BPD and 8.5 vs 8.2 mm, P = 0.008 for TW, for Groups 1 and 2, respectively). The mean NFA was significantly narrower in children of Group 2 (130.42° vs 125.68°, P = 0.03).
Group 1 exhibited larger FOD, but this result was not statistically significant (15.4 vs 15.2 mm, P = 0.19). CI was 62% in Group 1 and 65% in Group 2 (P = 0.002).
No differences between all postoperative measurements were observed between the two groups (13.5 vs 13.4 mm for BPD, 18.9 vs 18.6 mm for FOD, 131.4° vs 130° for NFA, 72% vs 72% for CI, each P > 0.1). ΔCI in Group 1 was 10, and in Group 2 it was 7. The difference between ΔCI was statistically significant (P = 0.034).
DISCUSSION
Subarachnoid spaces vary widely during the first 2 years of life. An enlargement of subarachnoid spaces is considered as physiologic in the first months of life. It increases from birth up to about 7 months of age;[
Subarachnoid spaces can be measured as CCW – the vertical distance between brain surface and calvarium, inter-hemispheric width (IHW), and sinocortical width (SCW) on a coronal plane.[
A pathologic enlargement of subarachnoid spaces is considered as common in sagittal craniosynostosis. Our findings are consistent with the literature where up to 70% of sagittal synostosis presents this abnormal condition.[
A mechanism of venous compression has been postulated in the literature concerning such enlargement of subarachnoid spaces: the compression of sagittal sinus would result in a consequent alteration of cerebrospinal fluid (CSF) absorption.[
Our analysis shows that the presence of an enlargement of the subarachnoid spaces is associated with a modification of the shape of the forehead. Correction of the shape and bulging of the forehead is indeed one of the most challenging problems in surgical management of scaphocephaly. The amelioration of forehead aspect can be obtained with the sole growing brain potential within sixth month of life,[
In this series, all the patients in both groups were operated on with the same surgical technique, that is, “Renier's H technique,” that is, a 4-cm strip craniectomy from bregma to lambda and temporal/parietal bone flap osteotomies.[
The homogeneity of population is also demonstrated by the mean age at intervention (123 in the second group and 130 in the first group). These elements also minimize the physiological difference in enlargement of subarachnoid spaces related to age.
Given the uniformity of population, two types of foreheads could be distinguished in scaphocephaly.
In the first group of sagittal synostosis, there is a predominant narrow bitemporal and biparietal width, a long FOD, and a large NFA. In this group, the CI is particularly low and the form can be defined cosmetically “severe.” Subarachnoid spaces are virtual or comparable to normal children.
Conversely, children in the second group present a large and prominent frontal bossing, with a narrow NFA. These features are associated with dilated subarachnoid spaces. In these infants, the CI is milder than in the first group.
These findings suggest that despite a common anatomical closure of the sagittal suture, the so-called “compensatory growth” of the skull differs with two main forms: one with large subarachnoid spaces and prominent forehead and another with small subarachnoid spaces and severe alteration in CI, thus the brain may be differently affected in children with scaphocephaly according to these two subtypes.
These two subtypes respond differently to the same surgery. Patients without a pathologic enlargement of subarachnoid spaces (Group 1) exhibit only a minor improvement of the forehead with a non-significative amelioration in the postoperative NFA, whereas patients in Group 2 show a major modification in NFA. These data also suggest that it is possible to try and “foresee” how the forehead might respond to a craniofacial retrocoronal procedure and to determine in which subgroup of patients with isolated sagittal suture synostosis the aspect of the forehead might not change significantly. However, the final measurements are similar in the two groups showing that H technique can be used in both subtypes.
The main limit of this study remains the analysis of the evolution of the subarachnoid spaces. No CT or magnetic resonance imaging was performed routinely in the postoperative period in the population herein studied; for such a reason, it is not possible to assess the evolution of the enlargement of subarachnoid spaces after surgery. Because no ICP recording was systematically performed, it is not possible to associate one or other CSF pattern with an actual risk of developing a raised ICP.
Further prospective studies are needed to analyze the impact of surgical correction on subarachnoid spaces.
CONCLUSION
A pathologic subarachnoid space enlargement is found in two-thirds of scaphocephalies. The presence of such extra-axial CSF collection in these patients has a significant cosmetic impact on the preoperative morphology of the forehead and on the response to surgical correction.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
We are thankful to Salima Madini and the whole staff of medical files archive for their help in collecting patient's data and medical reports.
References
1. Arnaud E, Capon-Degardin N, Michienzi J, Di Rocco F, Renier D. Scaphocephaly part II: Secondary coronal synostosis after scaphocephalic surgical correction. J Craniofac Surg. 2009. 20: 1843-50
2. Arnaud E, Renier D, Marchac D. Prognosis for mental function in scaphocephaly. J Neurosurg. 1995. 83: 476-9
3. Badve CA, K MM, Ishak GE, Khanna PC. Craniosynostosis: Imaging review and primer on computed tomography. Pediatr Radiol. 2013. 43: 728-42
4. Becker DB, Petersen JD, Kane AA, Cradock MM, Pilgram TK, Marsh JL. Speech, cognitive, and behavioral outcomes in nonsyndromic craniosynostosis. Plast Reconstr Surg. 2005. 116: 400-7
5. Boulos PT, Lyn KY, Jane JA, Jane JA. Correction of sagittal synostosis using a modified Pi method. Clin Plast Surg. 2004. 31: 489-98
6. Cartwright CC, Jimenez DF, Barone CM, Baker L. Endoscopic strip craniectomy: A minimally invasive treatment for early correction of craniosynostosis. J Neurosci Nurs. 2003. 35: 130-8
7. Chadduck WM, Chadduck JB, Boop FA. The Subarachnoid Spaces in Craniosynostosis. Neurosurgery. 1992. 30: 867-71
8. Di Rocco F, Arnaud E, Meyer P, Sainte-Rose C, Renier D. Focus session on the changing “epidemiology” of craniosynostosis (comparing two quinquennia: 1985–1989 and 2003–2007) and its impact on the daily clinical practice: A review from Necker Enfants Malades. Childs Nerv Syst. 2009. 25: 807-11
9. Di Rocco F, Arnaud E, Renier D. Evolution in the frequency of nonsyndromic craniosynostosis. J Neurosurg Pediatr. 2009. 4: 21-5
10. Di Rocco F, Ben Gbulie U, Meyer P, Arnaud E. Current techniques and protocols in the surgical management of scaphocephaly in young infants. J Craniofac Surg. 2014. 25: 39-41
11. Di Rocco F, Knoll I, Arnaud E, Blanot S, Meyer P, Cuttarree H. Scaphocephaly correction with retrocoronal and prelambdoid craniotomies (Renier's “H” technique). Childs Nerv Syst. 2012. 28: 1327-32
12. Fessell DP, Frankel DA, Wolfson WP. Sonography of extraaxial fluid in neurologically normal infants with head circumference greater than or equal to the 95th percentile for age. J Ultrasound Med. 2000. 19: 443-7
13. Florisson JM, van Veelen ML, Bannink N, van Adrichem LN, van der Meulen JJ, Bartels MC. Papiledema in isolated single-suture craniosynostosis: Prevalence and predictive factors. J Craniofac Surg. 2010. 21: 20-4
14. Guimarãsses-Ferreira J, Gewalli F, David L, Darvann TA, Hermann NW, Kreiborg S. Sagittal synostosis: I. Preoperative morphology of the skull. Scand J Plast Reconstr Surg Hand Surg. 2006. 40: 193-9
15. Hassler W, Zentner J. Radical osteoclastic craniectomy in sagittal synostosis. Neurosurgery. 1990. 27: 539-43
16. Jane JA, Edgerton MT, Futrell JW, Park TS. Immediate correction of sagittal synostosis. J Neurosurg. 1978. 49: 705-10
17. Johnston SA. Calvarial vault remodeling for sagittal synostosis. AORNJ. 2001. 74: 632-47
18. Kleinman PK, Zito JL, Davidson RI, Raptopoulos V. The subarachnoid spaces in children: normal variations in size. Radiology. 1983. 147: 455-7
19. Kotrikova B, Krempien R, Freier K, Mühling J. Diagnostic imaging in the management of craniosynostoses. Eur Radiol. 2007. 17: 1968-78
20. Lam WW, Ai VH, Wong V, Leong LL. Ultrasonographic measurement of subarachnoid space in normal infants and children. Pediatr Neurol. 2001. 25: 380-4
21. Libicher M, Tröger J. US measurement of the subarachnoid space in infants: Normal values. Radiology. 1992. 184: 749-51
22. Magge KT, Magge SN, Keating RF, Myseros JS, Boyajian MJ, Sauerhammer TM. Incidental findings on preoperative computed tomography for nonsyndromic single suture craniosynostosis. J Craniofac Surg. 2014. 25: 1327-30
23. Marchac D, Renier D, Broumand S. Timing of treatment for craniosynostosis and facio-craniosynostosis: A 20-year experience. Br J Plast Surg. 1994. 47: 211-22
24. McLaurin LR, Matson DD. Importance of early surgical treatment of craniosynostosis; review of 36 cases treated during the first six months of life. Pediatrics. 1952. 10: 637-52
25. Murray DJ, Kelleher MO, McGillivary A, Allcutt D, Earley MJ. Sagittal synostosis: A review of 53 cases of sagittal suturectomy in one unit. J Plast Reconstr Aesthet Surg. 2007. 60: 991-7
26. Nagaraja S, Anslow P, Winter B. Craniosynostosis. Clini Radiol. 2013. 68: 284-92
27. Pensler JM, Ciletti SJ, Tomita T. Late correction of sagittal synostosis in children. Plast Reconstr Surg. 1996. 97: 1362-7
28. Persing J, Babler W, Winn HR, Jane J, Rodeheaver G. Age as a critical factor in the success of surgical correction of craniosynostosis. J Neurosurg. 1981. 54: 601-6
29. Shillito J, Matson DD. Craniosynostosis: A review of 519 surgical patients. Pediatrics. 1968. 41: 829-53
30. Sutton LN, Barlett SP, Duhaime AC, Markakis D. Total cranial vault reconstruction for the older child with scaphocephaly. Pediatr Neurosurg. 1993. 19: 63-72
31. van Veelen ML, Eelkman Rooda OH, de Jong T, Dammers R, van Adrichem LN, Mathijssen IM. Results of early surgery for sagittal suture synostosis: Long-term follow-up and the occurrence of raised intracranial pressure. Childs Nerv Syst. 2013. 29: 997-1005
32. Vollmer DG, Jane JA, Parks TS, Persing JA. Variant of sagittal synostosis strategies for surgical correction. J Neurosurg. 1984. 61: 557-62
33. Windth P, Davis C, Sanger C, Sahlin P, Lauritzen C. Spring-Assisted Cranioplasty vs pi-plasty for sagittal synostosis - a long term follow-up study. J Craniofac Surg. 2008. 19: 59-64
34. Zahl SM, Egge A, Helseth E, Wester K. Benign external hydrocephalus: A review, with emphasis on management. Neurosurg Rev. 2011. 34: 417-32