- Department of Neurosurgery, La Princesa University Hospital, Madrid, Spain
- Department of Ophthalmology, La Princesa University Hospital, Madrid, Spain
Correspondence Address:
Ricardo Gil-Simoes
Department of Neurosurgery, La Princesa University Hospital, Madrid, Spain
DOI:10.4103/sni.sni_292_18
Copyright: © 2019 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Ricardo Gil-Simoes, José M. Pascual, Andrés P. Casas, Rafael G. de Sola. Intrachiasmatic craniopharyngioma: Assessment of visual outcome with optical coherence tomography after complete surgical removal. 21-Jan-2019;10:7
How to cite this URL: Ricardo Gil-Simoes, José M. Pascual, Andrés P. Casas, Rafael G. de Sola. Intrachiasmatic craniopharyngioma: Assessment of visual outcome with optical coherence tomography after complete surgical removal. 21-Jan-2019;10:7. Available from: http://surgicalneurologyint.com/surgicalint-articles/9163/
Abstract
Background:Optic chiasm invasion by a craniopharyngioma (CP) is exceptional. Surgical treatment of intrachiasmatic CPs associates a high risk of chiasm injury, which should be properly addressed before surgery.
Case Description:We present a 46-year-old woman admitted to the hospital with low visual acuity (0.1 in the right eye and 0.5 in the left) and a severe defect in her visual fields, in addition to headaches, diabetes insipidus, and a long-term depressive disorder. Her visual deficit progressed from a right homonymous temporal inferior quadrantanopia to an almost complete loss of vision in both eyes that only spared the upper nasal quadrants. Brain MRI showed a rounded third ventricle tumor with a potbelly expansion of the optic chiasm, suggesting chiasm invasion by the tumor. Optical coherence tomography (OCT) showed the thinning of the retinal nerve fiber layer (RNFL) in the superior and temporal wedges of the right eye and in the temporal wedge of the left one. The tumor was completely removed by employing a frontotemporal craniotomy and a translamina terminalis approach. Histological analysis showed a squamous-papillary CP. Postoperatively, a significant worsening of the visual defect was evidenced on the perimetry, which was related to a marked RNFL atrophy measured with OCT, as compared to the preoperative study. The poor long-term visual outcome in this patient correlated well with the results of postoperative OCT.
Conclusions:Preoperative analysis of retinal atrophy with optic coherence tomography allows a reliable assessment of the patient's visual outcome in CPs involving the optic chiasm.
Keywords: Craniopharyngioma, optic chiasm, optical coherence tomography, third ventricle, visual outcome
INTRODUCTION
Craniopharyngiomas (CPs) represent a complex group of epithelial benign (WHO I) tumors presumably originated from cell remnants of the Rathke's pouch.[
In the present report, we describe a new case of an intrachiasmatic CP diagnosed in an adult woman who showed a progressive worsening of her vision. The tumor was totally removed using a pterional translamina terminalis approach. This study presents, in a comprehensive way, the correlation between the patient's postoperative visual outcome and the structural injury caused to her optic apparatus by this intrachiasmatic CP, evaluated through optical coherence tomography (OCT), a useful tool for quantitative assessment of the structural damage of the retinal nerve fiber layer (RNFL).
CASE DESCRIPTION
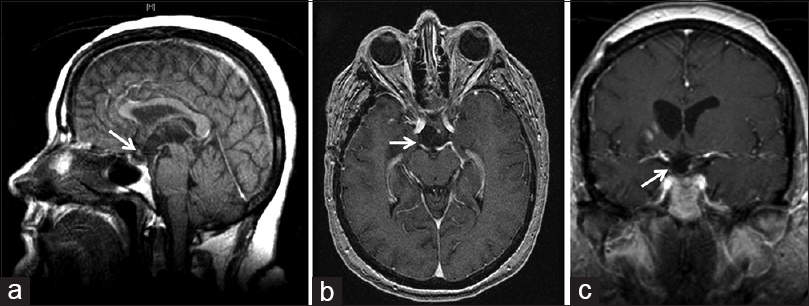
This 46-year-old woman was initially admitted to our hospital with headache, progressive decrease in visual acuity, and a fever above 38.5°C for the last month, in addition to polyuria and polydipsia for the last 2 weeks. Upon admission, her neurological exploration was unremarkable. Visual exploration disclosed a reduced visual acuity of both eyes, 20/200 in the right eye and 20/40 in the left one. The initial perimetry evidenced a homonymous left inferior quadrantanopia, whereas the funduscopic exam did not reveal any abnormality. All her baseline endocrine tests were also normal at this time. The brain MRI displayed a 1.5-cm mixed solid-cystic lesion occupying the lower 3V and adjacent hypothalamic–chiasmatic region [Figure
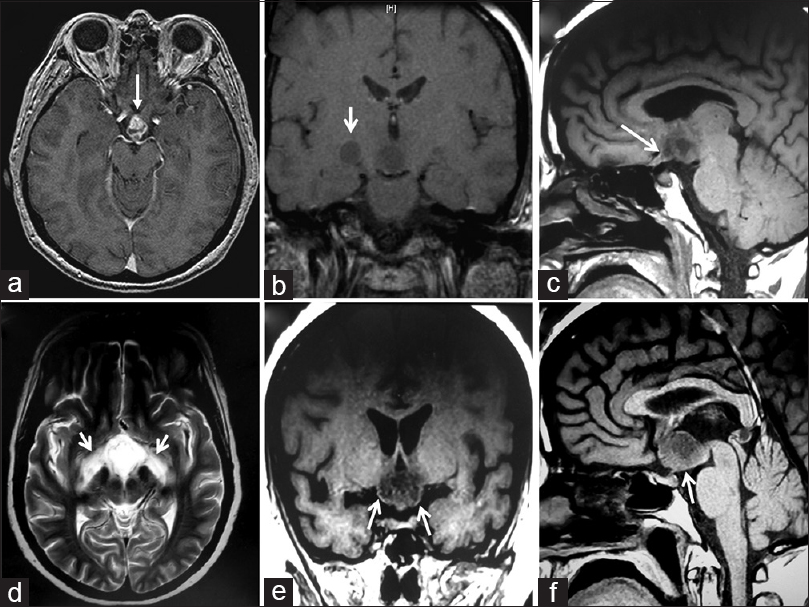
Figure 1
Preoperative magnetic resonance imaging (MRI) studies. (a, b) Initial MRI study, axial (a) and coronal (b) T1-weighted scans (1/25/2012), showing a heterogeneous solid-cystic lesion centered at the chiasmatic recess of the third ventricle. Note the anterior tongue-like tumor protrusion into the optic chiasm (white arrow). In the coronal image, there is a round hypointense area of similar low intensity as the tumor is visible over the trajectory of the right optic tract (white arrow). (c-f) Preoperative MRI study obtained 6 months later, just prior to the surgical procedure (7/13/2012), showing enlargement of the tumor. The finger-like tumor tissue projecting into the optic chiasm-right optic nerve can be identified (white arrow). (d) Axial T2-weighted preoperative MRI scan, showing hyperintense symmetrical signals following the course of both optic tracts, a sign known as “moustache sign,” which is believed to correspond to optic tract edema (white arrows). (e, f) Coronal and midsagittal T1-weighted scans demonstrating the strictly third ventricle topography of this craniopharyngioma, which has developed above an intact third ventricle floor/pituitary stalk (white arrows)
Six months later, the patient was admitted again due to visual deterioration and symptoms of panhypopituitarism and diabetes insipidus. She was treated with hormone replacement therapy and also received psychiatric assessment for a depressive disorder which had worsened in the last year.
Preoperative neuro-ophthalmological examination
At this point, a thorough neuro-ophthalmological and neuroradiological evaluation was performed. The patient could only see light with her right eye and could distinguish hand movements from 2 m distance with the left one. Visual fields were impaired severely in both eyes with a generalized loss of sensitivity in the right one and sparing of the upper nasal quadrant of the left eye (
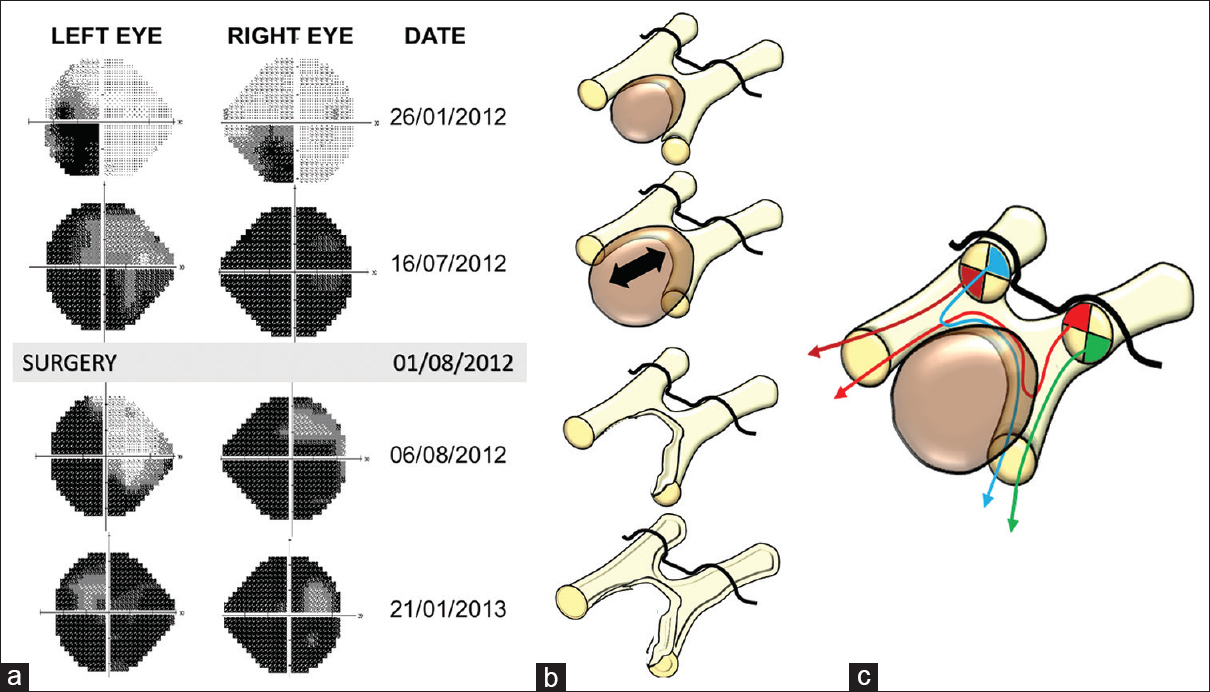
Figure 2
Visual field exam. Correlation between visual field defects and the anatomical injury to the visual pathways caused by the tumor and surgical procedure. (a) Chronological evolution of the visual field defects identified in the patient through consecutive automated perimetry exams. (b) Illustrative schemes showing the sequence of anatomical injuries to the optic chiasm and optic tracts, presumably caused by the tumor and the surgical procedure, consistent with the visual defects shown in the corresponding perimetry studies on the left panel. (c). Artistic illustration showing the spatial antero-posterior segregation of retinal fibers within the optic chiasm that accounts for the visual defects associated with intrachiasmatic craniopharyngiomas (CPs). The crossed fibers from the retina's upper quadrants follow their course at a more caudal (posterior) position within the chiasm, whereas those from the retina's lower quadrants tend to cross at a more rostral or anterior position within the chiasm. At the initial exam, the patient presented a homonymous left inferior quadrantanopia with worse visual acuity in the right eye (perimetry on 1/26/2012), a result congruent with the invasion of the chiasm's posterior aspect and right optic tract's medial aspect (top scheme in B). As the tumor enlarges, it encroaches upon a wider area of the posterior and central chiasm, including both optic tracts, thus damage resulting in a severe visual deficit (second perimetry on 07/16/2012) that only spared the uncrossed temporal inferior fibers from the left retina along the chiasm's left edge (second scheme from the top in (b) and brown tract of fibers in (c)). After removal of the CP, the persistence of the visual deficit in the lower quadrants of the right eye and in the nasal quadrants of the left eye (immediate postoperative perimetry on 8/6/2012) was congruent with irreversible injury to the chiasm's posterior aspect and the medial aspect of the right optic tract (third scheme from the top in B and blue tract of fibers in C). The late postoperative perimetry exam (postoperative perimetry on 01/21/2013) showed a significantly worsening of the visual defect, probably due to degenerative changes in retinal fibers associated with gliotic scarring (bottom scheme in B)
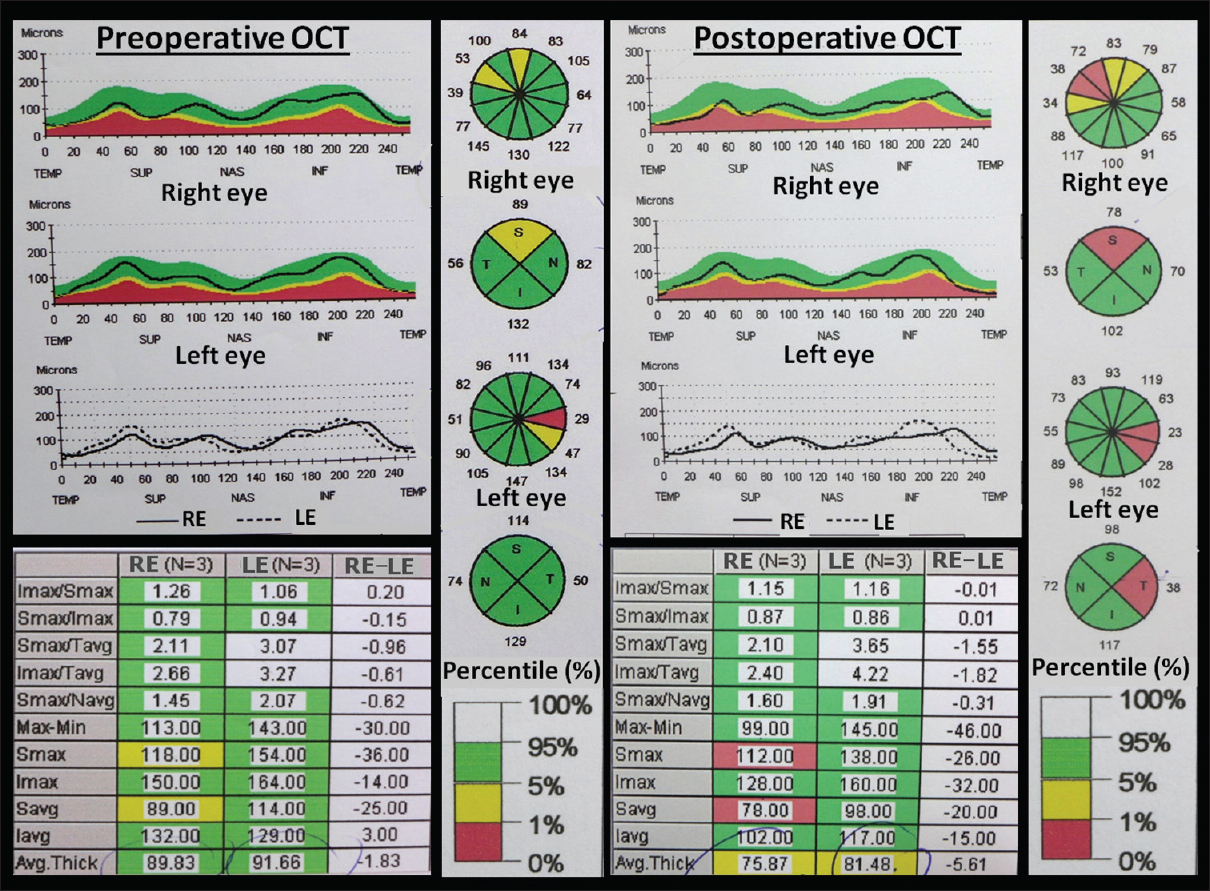
Figure 3
Optical coherence tomography (OCT) exams. Analysis of the changes in the retinal nerve fiber layer (RNFL) thickness that occurred as the result of the chiasmal damage associated with this intrachiasmatic craniopharyngioma and the surgical procedure for its removal. Left panel: Preoperative OCT confirmed the presence of RNFL thinning involving the superior and temporal wedges (or quadrants) of both eyes, more severe in the upper retina of the right eye (yellow wedge). Right panel: Postoperative OCT demonstrated further thinning of the RNFL in the upper retina of the right eye as well as in the temporal wedge of the left retina (orange wedges). Such a thinning reflected the irreversible damage of the axonal fibers within the area of the optic chiasm and optic tracts infiltrated by the tumor, a result indicative of a worse prognosis for recovery following the surgery
Surgical procedure
The tumor was approached through a right frontotemporal craniotomy. After opening the Sylvian fissure, the optic chiasm was exposed. It was displaced against the tuberculum sellae and presented a quite swollen and shortened appearance [
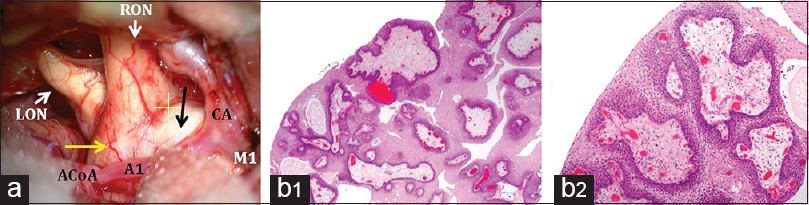
Figure 4
(a) Intraoperative view and histological diagnosis. Surgical view through the right frontal-temporal (pterional) approach showing the anatomical deformation of the optic chiasm under microscopic magnification. A prefixed, swollen optic chiasm is observed. Note the bulging of the lamina terminalis (yellow arrow) and the whitish expansion of the right optic tract, a sign of tumor infiltration. A1: A1 segment of the right anterior cerebral artery; ACoA: anterior communicating artery; CA: carotid artery; M1: M1 proximal segment of the right median artery; LON: left optic nerve; RON: right optic nerve; b1–b2: histopathological sections showing the distinctive features of this squamous-papillary craniopharyngioma. Solid areas of well-differentiated stratified squamous epithelium with basal palisading and pseudopapillae formation around cores of loose fibrovascular stroma are typical of this variant. H and E, original magnification × 16 (b1) and × 40 (b2)
Postoperative course and visual outcome
The patient recovered successfully from the surgical procedure, although her panhypopituitarism, diabetes insipidus, and subjective visual disturbance remained unchanged. Complete removal of the lesion was confirmed on postoperative MRI [
Figure 5
Postoperative MRI studies. (a) Midsagittal T1-weighted MRI scan after total removal of this intrachiasmatic craniopharyngioma. The outline of the prefixed, downward displaced optic chiasm (white arrow) is observed next to the defective or “breached” third ventricle floor, as the atrophic tuber cinereum was removed along with the tumor. (b, c) Axial and coronal T1-weighted scans through the level of the optic apparatus. Notice the anatomical injury to the optic chiasm-right optic tract junction after removing the lesion (white arrows)
Follow-up neuro-ophthalmological assessment 1 week after surgery showed some improvement in the patient's visual acuity and visual fields. Visual acuity returned to its prior initial values, from only light perception to 20/60 in the right eye and 20/40 in the left one. A marked improvement of vision in the nasal hemi-field in the left eye was observed as well, although only a partial recovery at the temporal-superior quadrant of the right eye occurred (
DISCUSSION
Intrachiasmatic CPs: An intraventricular topographical variant
CPs with either a primary intrachiasmatic development or true anatomical invasion of the optic chiasm represent a quite exceptional topographical variant, with very few cases recorded in the literature.[
The intrachiasmatic development of a CP can be explained following the embryonic theory for the origin of CPs, originally proposed by Jakob Erdheim in 1904.[
MRI findings defining the intrachiasmatic occupation by a CP
The optic chiasm displays an oval or elliptical morphology on midsagittal MRI scans in healthy individuals. In contrast, a downward displacement of this structure with a crescent-moon shape on midsagittal MRI represents the fundamental deformation produced by CPs developed within the 3V.[
Chiasm pathways injured by intrachiasmatic CPs: Correlation with visual field deficits
Over 2 million nerve fibers pass through the optic chiasm, in which a spatial segregation of crossed and uncrossed fibers can be evidenced.[
Visual disturbances associated with CPs are mainly related to the particular type of anatomical distortion to the optic chiasm caused by the tumor.[
The sequential stages of visual field impairment in our patient were congruent with the topography and the anatomical region of the chiasm injured by the tumor. At the initial perimetry, the patient presented a homonymous left inferior quadrantanopia, worse in the right eye, a deficit explained by a damage to the posterior edge of the optic chiasm, and the medial aspect of the right optic tract [
The rapid worsening of patient's visual deficit over the following 6 months [
Surgical removal and visual outcome of intrachiasmatic CPs: The predictive value of OCT
Intrachiasmatic CPs essentially represent a particular variant of 3V CPs which are infiltrating the optic chiasm.[
One week after the procedure, the first postoperative perimetry examination showed a significant improvement in visual acuity in the nasal field of the left eye but only a partial recovery of vision in the temporal superior quadrant of the right eye (
No conclusive explanations regarding the initial incongruence between the early postoperative visual improvement but worsening OCT results in this patient can be stated. The greater atrophy in the RNFL immediately following the surgical procedure would logically reflect the injury to the chiasm associated with the surgical maneuvers employed to dissect the intrachiasmatic tumor attachment. RNFL thinning reflects irreversible retinal ganglion cell damage, which usually occurs after prolonged compression of the visual pathways.[
The final question to consider is the degree of removal to be attempted when dealing with a CP that infiltrates the optic chiasm. A generalized surgical attitude cannot be recommended, and the most appropriate, judicious decision should be established on an individual basis. The high risk of irreversible chiasm injury associated with radical removal of a CP must be pondered with the rate of tumor recurrence, taking into consideration the preoperative visual status, the patient's age, and, above all, the extent and strength of tumor attachment. The impossibility of applying any type of radiosurgery on the optic chiasm for the treatment of CP remnants adds an extra level of complexity to this issue. Preoperative OCT assessment of the structural damage to the visual pathways may help to predict the potential reversibility of visual impairment and provides helpful insight to guide the intraoperative surgical decisions.
CONCLUSIONS
Intrachiasmatic CPs represent an exceptional variant of 3V CPs characterized by the invasion of the posterocentral region of the optic chiasm. A characteristic potbelly deformation of the optic chiasm visible on preoperative MRI points to CP infiltration of this structure. Tumor encroachment of the chiasm's posterior aspect and adjacent optic tracts usually cause a visual deficit involving initially the temporal lower quadrants. The pterional approach combined with the opening of the lamina terminalis provides an optimal exposure of any CP extension into the optic chiasm. Preoperative analysis of retinal atrophy with OCT allows a reliable assessment of the patient's visual outcome and it may help to decide about the degree of CP removal to be performed.
Abbreviations
CP: Craniopharyngioma; MRI: Magnetic resonance imaging; OCT: Optical coherence tomography; RNFL: Retinal nerve fiber layer. 3V: Third ventricle; 3VF: Third ventricle floor.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest
Acknowledgments
The authors wish to express their gratitude to George Hamilton for his critical review of the language and style of the manuscript.
References
1. Bialer OY, Goldenberg-Cohen N, Toledano H, Snir M, Michowiz S. Retinal NFL thinning on OCT correlates with visual field loss in pediatric craniopharyngioma. Can J Ophthalmol. 2013. 48: 494-9
2. Brodsky MC, Hoyt WF, Barnwell SL, Wilson CB. Intrachiasmatic craniopharyngioma: A rare cause of chiasmal thickening. J Neurosurg. 1988. 68: 300-2
3. Ciric IS, Cozzens JW. Craniopharyngiomas: Transsphenoidal method of approach -- for the virtuoso only?. Clin Neurosurg. 1980. 27: 169-87
4. Danesh-Meyer HV, Papchenko T, Savino PJ, Law A, Evans J, Gamble GD. In vivo retinal nerve fiber layer thickness measured by optical coherence tomography predicts visual recovery after surgery for parachiasmal tumors. Invest Ophthalmol Vis Sci. 2008. 49: 1879-85
5. Duff TA, Levine R. Intrachiasmatic craniopharyngioma. Case Report. J Neurosurg. 1983. 59: 1766-8
6. Erdheim J. Über Hypophysengangsgeschwülste und Hirncholesteatome. Sitzungsb Kais Akad Wissen Math Naturw Klin. 1904. 113: 537-726
7. Higashi S, Yamasita J, Fujisawa H, Yamamoto Y, Kadoya M. “Moustache” appearance in craniopharyngiomas: Unique magnetic resonance imaging and computed tomographic findings of perifocal edema. Neurosurgery. 1990. 27: 993-6
8. Hoyt WF, Luis O. The primate optic chiasm. Details of visual fiber organization studied by silver impregnation techniques. Arch Ophthalmol. 1963. 70: 69-85
9. Jacon M, Raverot G, Jouanneau E, Matheos K, Stylli S, Nichols A. Predicting visual outcome after treatment of pituitary adenoma with optical coherence tomography. Am J Ophthalmol. 2009. 147: 64-70
10. Kidd D. The optic chiasm. Clinical Anatomy. 2014. 27: 1149-58
11. Lindenberg R, Walsh FB, Sacks JG.editorsNeuropathology of Vision: An Atlas. Philadelphia: Lea & Febiger; 1973. p. 274-5
12. Mediero S, Noval S, Bravo-Ljubetic L, Contreras I, Carceller F. Visual outcomes, visual fields, and optical coherence tomography in paediatric craniopharyngioma. Neuro-ophthalmology. 2015. 39: 132-9
13. Newman SA, Laws ER, Sheehan JP.editors. Ophthalmologic evaluation and management. Sellar and Parasellar Tumors. Diagnosis, Treatments and Outcomes. New York, NY: Thieme Medical Publishers Inc; 2012. p. 65-123
14. Pascual JM, Carrasco R, Prieto R, Gonzalez-Llanos F, Alvarez F, Roda JM. Craniopharyngioma classification. J Neurosurg. 2008. 109: 1180-2
15. Pascual JM, González-Llanos F, Barrios L, Roda JM. Intraventricular craniopharyngiomas: Topographical classification and surgical approach selection based on an extensive overview. Acta Neurochir (Wien). 2004. 146: 785-802
16. Pascual JM, Prieto R, Carrasco R. Infundibulo-tuberal or not strictly intraventricular craniopharyngioma: Evidence for a major topographical category. Acta Neurochir (Wien). 2011. 153: 2403-26
17. Pascual JM, Prieto R, Carrasco R, Barrios L. Displacement of mammillary bodies by craniopharyngiomas involving the third ventricle: Surgical-MRI correlation and use in topographical diagnosis. J Neurosurg. 2013. 119: 381-405
18. Pascual JM, Rosdolsky M, Prieto R, Strauss S, Winter E, Ulrich W. Jakob Erdheim (1874-1937): Father of hypophyseal-duct tumors (craniopharyngiomas). Virchows Arch. 2015. 467: 459-69
19. Pomeranz HD, Aldrich EF. Intrachiasmal craniopharyngioma: Treatment with a cisternal catheter drainage and radiation. J Neuroophthalmol. 2004. 24: 27-30
20. Prasad S, Galetta S. Anatomy and physiology of the afferent visual system. Handb Clin Neurol. 2011. 102: 4-19
21. Prieto R, Pascual JM, Barrios L. Optic chiasm distortions caused by craniopharyngiomas: Clinical and magnetic resonance imaging correlation and influence on visual outcome. World Neurosurg. 2015. 83: 500-29
22. Prieto R, Pascual JM, Rosdolsky M, Castro-Dufourny I, Carrasco R, Strauss S. Craniopharyngioma adherence: A comprehensive topographical categorization and outcome-related risk stratification model based on the methodical examination of 500 tumors. Neurosurg Focus. 2016. 41: E13-
23. Steno J. Microsurgical topography of craniopharyngiomas. Acta Neurochir Suppl (Wien). 1985. 35: 94-100
24. Yang L, Qu Y, Lu W, Liu F. Evaluation of macular ganglion cell complex and peripapillary retinal nerve fiber layer in primary craniopharyngioma by Fourier-domain optical coherence tomography. Med Sci Monit. 2016. 22: 2309-14
25. Youl BD, Plant GT, Stevens JM, Kendall BE, Symon L, Crockard HA. Three cases of craniopharyngioma showing optic tract hypersignal on MRI. Neurology. 1990. 40: 1416-9