- Departments of Neurological Surgery, University of Texas Southwestern Medical Center, Dallas, Texas, United States.
- Departments of Pathology, University of Texas Southwestern Medical Center, Dallas, Texas, United States.
- Departments of Radiation Oncology, University of Texas Southwestern Medical Center, Dallas, Texas, United States.
Correspondence Address:
Toral R. Patel
Departments of Neurological Surgery, University of Texas Southwestern Medical Center, Dallas, Texas, United States.
DOI:10.25259/SNI_111_2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Sumanth Reddy, Aaron Plitt, Jack Raisanen, Ankur R. Patel, Purva Gopal, Robert Timmerman, Toral R. Patel. Intracranial anaplastic hemangiopericytoma presenting with simultaneous extra-cranial metastases: A case report and review of the literature. 26-Jul-2019;10:148
How to cite this URL: Sumanth Reddy, Aaron Plitt, Jack Raisanen, Ankur R. Patel, Purva Gopal, Robert Timmerman, Toral R. Patel. Intracranial anaplastic hemangiopericytoma presenting with simultaneous extra-cranial metastases: A case report and review of the literature. 26-Jul-2019;10:148. Available from: http://surgicalneurologyint.com/surgicalint-articles/9529/
Abstract
Background: Intracranial solitary fibrous tumor/hemangiopericytoma (SFT/HPC) is a rare mesenchymal tumor with a propensity to recur and metastasize extracranially years after treatment. Accordingly, there are no reported cases of a patient presenting with a simultaneous intracranial primary and extracranial metastases. We present the case of a patient presenting with an intracranial SFT/HPC and simultaneous liver metastases and propose a treatment paradigm.
Case Description: A 74-year-old male smoker presented with confusion. An MRI of the brain revealed a heterogeneously enhancing left frontal extra-axial mass. Systemic workup revealed multiple small liver lesions concerning for metastases. The patient underwent gross total resection (GTR) of the intracranial lesion with adjuvant CyberKnife stereotactic radiotherapy to the resection cavity. Pathology was consistent with a WHO Grade III SFT/HPC (previously known as anaplastic HPC). The liver lesions were biopsied and confirmed to be metastases. They were subsequently treated with stereotactic body radiation therapy, temozolomide, and bevacizumab. Eighteen months postoperatively, the patient is alive with no evidence of intracranial malignancy and regression of the hepatic lesions.
Conclusion: Several studies support GTR and adjuvant radiotherapy to treat intracranial SFT/HPC. The role of adjuvant chemotherapy is less clear. Metastatic disease is typically detected several years after the initial diagnosis, and there is no consensus regarding the optimal treatment strategy. We propose that the rare presentation of intracranial SFT/HPC with simultaneous extracranial metastases should be treated in a multidisciplinary fashion with surgical resection, adjuvant radiotherapy, and chemotherapy.
Keywords: Extracranial metastasis, Hemangiopericytoma, Intracranial, Simultaneous, Solitary fibrous tumor, Synchronous
INTRODUCTION
Intracranial solitary fibrous tumor/hemangiopericytoma (SFT/HPC) is a rare tumor of mesenchymal origin.[
Grade I SFT/HPC is characterized by low cellularity and dense collagen deposition, which corresponds to the prior diagnosis of SFT. Grade II SFT/HPC is characterized by “staghorn” vasculature and increased cellularity. The presence of at least five mitoses per 10 high-power fields denotes a Grade III SFT/HPC, previously recognized as an anaplastic HPC.[
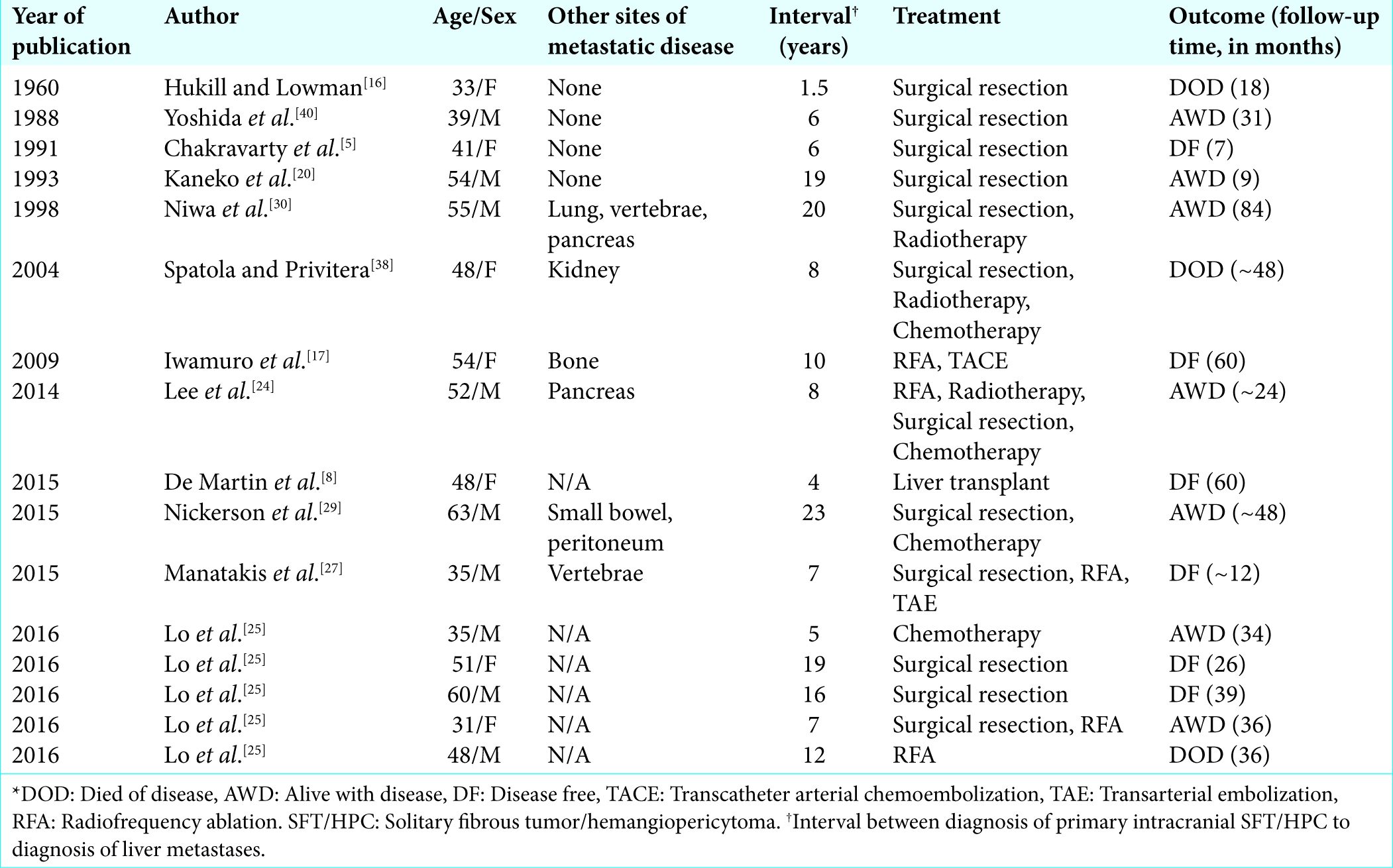
Intracranial SFT/HPCs metastasize to extracranial locations at a rate of 11.6 to 69% within 10 years of initial diagnosis, most commonly to the lung/pleura, liver, and bone.[
In this report, we detail the case of a patient presenting with a symptomatic anterior cranial fossa WHO Grade III SFT/HPC and synchronous liver metastases. We will describe the patient’s clinical course and briefly review the literature in an effort to provide a rational treatment approach to this unusual clinical scenario.
CASE DESCRIPTION
A 74-year-old, right-handed, male smoker was taken to an outside hospital after he became confused at a gas station. This episode was preceded by 1 month of cognitive decline, personality changes, and urinary incontinence, as well as self- reported clear rhinorrhea while eating. His neurologic exam was notable for a flat affect and a relative paucity of speech.
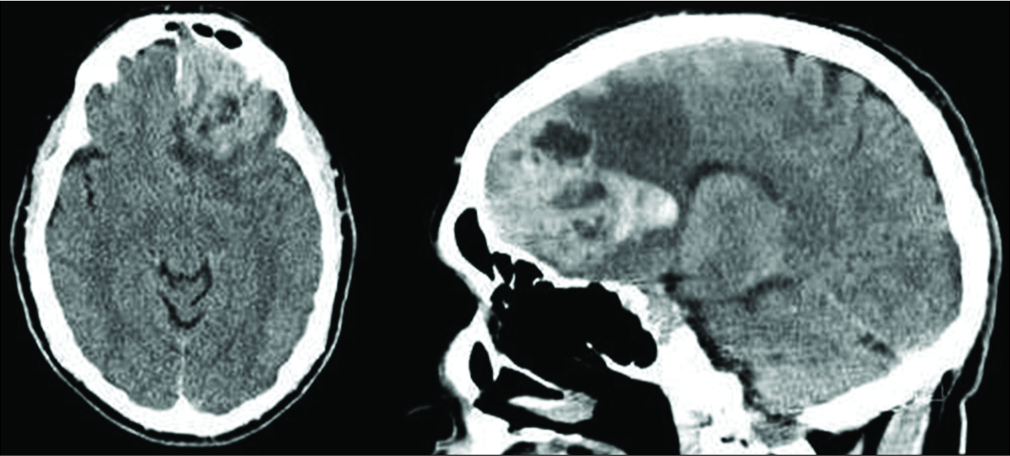
A head computed tomography (CT) was obtained and demonstrated a large, hyperdense left frontal extra- axial mass along the floor of the anterior cranial fossa, with bony erosion into the frontal sinus [
Figure 2:
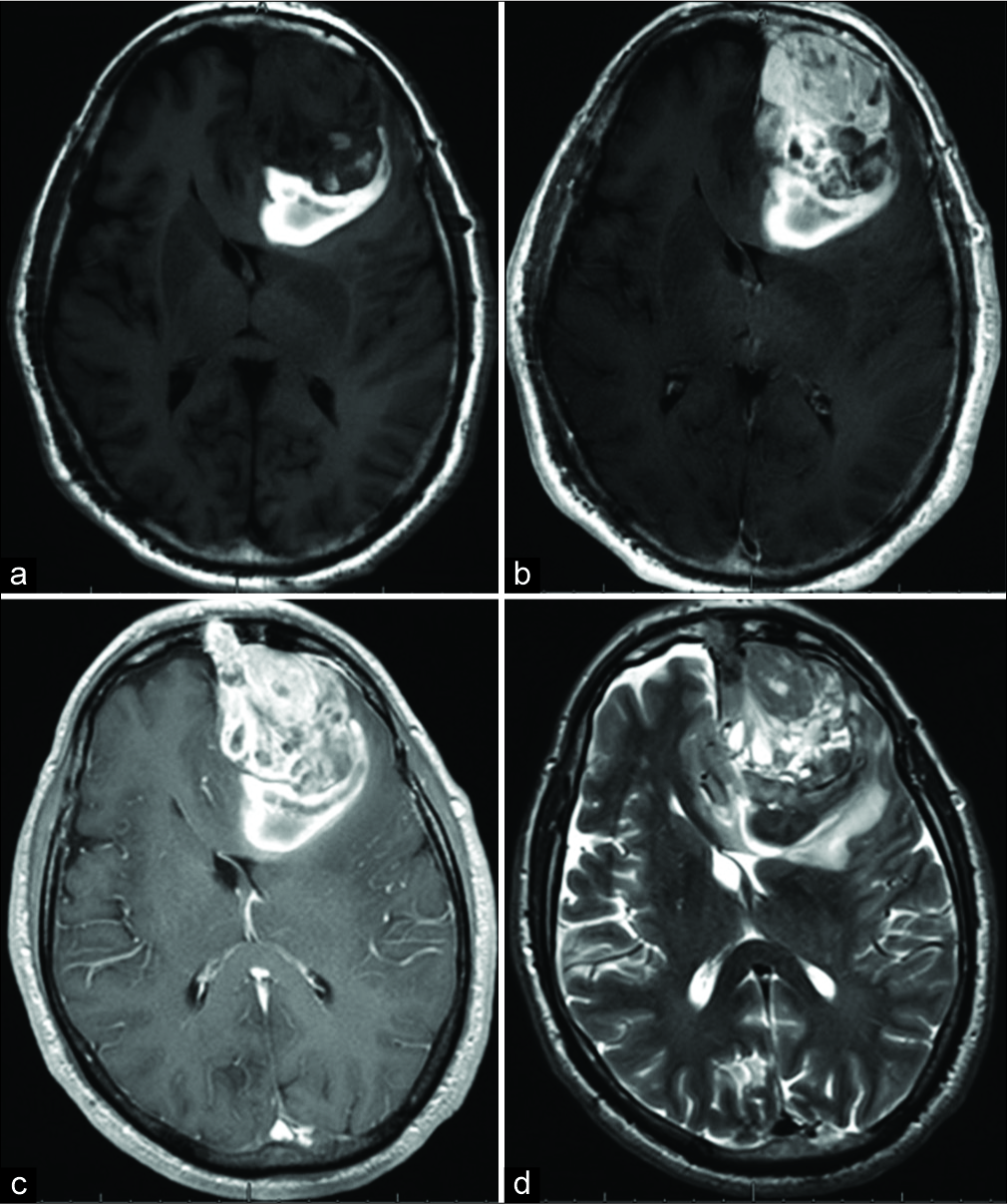
MRI brain. (a) T1 noncontrast and (b) T1 postcontrast sequences demonstrate a 7.0 cm×5.0 cm×4.9 cm heterogeneously enhancing left frontal mass with T1 shortening at the posterior aspect of the lesion, suggestive of prior hemorrhage. (c) T1 postcontrast sequences also reveal a dural tail and (d) T2-weighted sequences demonstrate a partial cerebrospinal fluid cleft around the periphery of the lesion; both findings suggest that the mass is extra- axial in origin.
An inferior vena cava filter was placed, and the patient was subsequently taken to the operating room for resection of the intracranial tumor. A two-piece bifrontal craniotomy was performed since the tumor was noted to invade both the frontal sinus and the superior sagittal sinus on the preoperative imaging. The lesion was of mixed consistency, with both firm and soft components. There was significant hypervascularity, which was controlled with bipolar electrocautery. The involved portions of the superior sagittal sinus and frontal sinus were removed. The frontal sinus was subsequently obliterated with an abdominal fat graft and reconstructed with a combination of methyl methacrylate and titanium plates. Intraoperative frozen section analysis was consistent with a high-grade neoplasm (SFT/HPC vs. anaplastic meningioma vs. metastasis). A gross total resection (GTR) was achieved. The patient tolerated the procedure well; his speech and affect improved postoperatively. A postoperative MRI confirmed GTR.
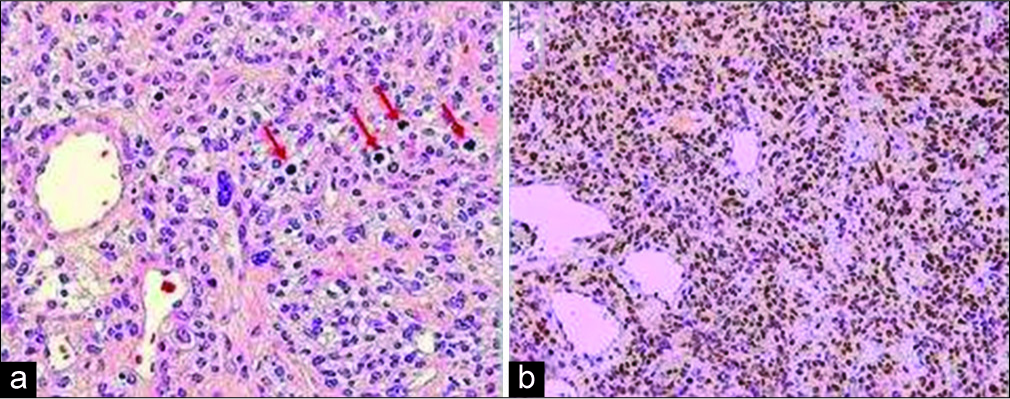
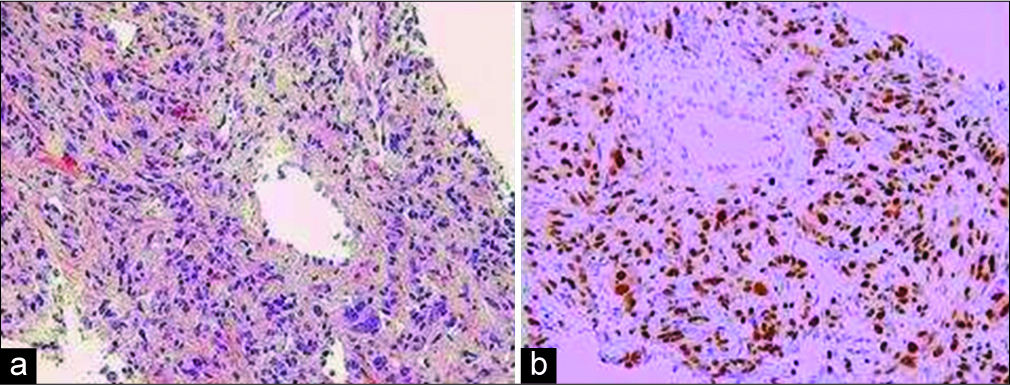
The pathological assessment revealed that the left frontal tumor was a SFT/HPC. The tumor was histologically characterized by a proliferation of relatively small cells with round to oval nuclei, some with small nucleoli, and scant or indistinguishable cytoplasm. The cells were arranged haphazardly among staghorn-like vessels. There were >5 mitotic figures in 10 high-power microscopic fields and foci of tumor necrosis, and thus, the neoplasm was designated as anaplastic (WHO Grade III). Immunoperoxidase stains demonstrated nuclear expression of STAT6 by many of the neoplastic cells indicating fusion of the NAB2 and STAT6 genes [
The patient underwent adjuvant CyberKnife stereotactic radiation therapy to the intracranial resection cavity. He received a total dose of 50 Gy (in 25 fractions). In addition, stereotactic body radiation therapy (SBRT) was used to treat the liver lesions: a segment 6 liver lesion was treated with 40 Gy in one fraction and a segment eight liver lesion was treated with 60 Gy in five fractions.
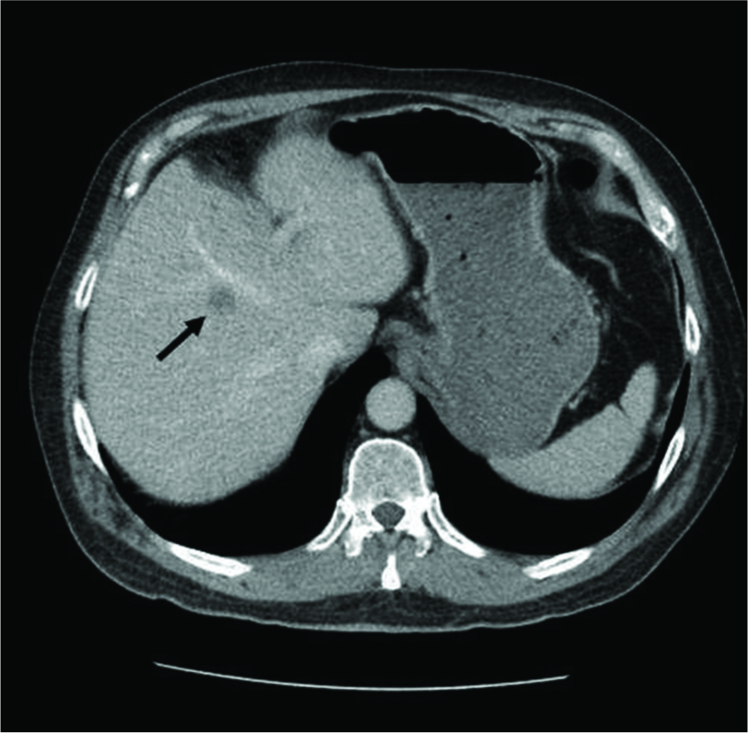
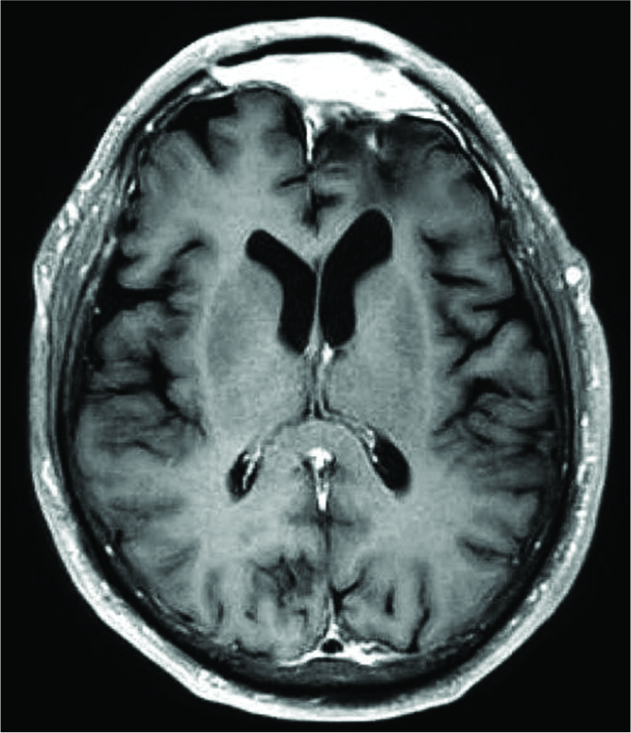
Sequential surveillance MRIs of the brain and abdomen were obtained following the completion of radiation treatment. MRIs of the brain remained stable, with no evidence of disease recurrence at 18 months postoperatively [
DISCUSSION
Surgical resection remains the primary treatment for SFT/HPCs. Numerous studies have demonstrated that GTR improves both overall survival (OS) and progression- free survival (PFS) in patients with SFT/HPC, as compared to subtotal resection (STR).[
The role of adjuvant radiotherapy for treatment of intracranial SFT/HPC remains controversial. Some studies have found that postoperative radiotherapy prolongs PFS, but that it does not provide a survival benefit.[
There is a paucity of published literature addressing the role of adjuvant systemic chemotherapy for SFT/HPC. In general, treatment with traditional cytotoxic chemotherapeutic agents has yielded disappointing results.[
Of note, a Phase II trial of sunitinib (another tyrosine kinase inhibitor, which targets VEGFR, PDGFR, and c-KIT) for atypical and anaplastic meningioma included an exploratory cohort of six patients with SFT/HPC. Three patients discontinued therapy due to toxicity, two withdrew, and one had a progression of disease at 1.6 months.[
Finally, combination therapy with temozolomide and bevacizumab has been shown to be effective in the treatment of patients with refractory disease, although it is unclear if this benefit is from temozolomide, bevacizumab, or a synergistic response. A case series of 14 patients, with a median follow-up of 34 months, demonstrated either partial response (11 patients) or stable disease (2 patients) in all but one patient. For those patients with a partial response, the treatment effect was observed after 2 to 4 cycles of therapy, at a median of 2.5 months. Median PFS was 9.67 months and 78.6% of patients were progression-free at 6 months. Five patients had a PFS of at least 20 months.[
Extracranial metastases are a relatively common finding in patients with SFT/HPC. A recent large case review found that 28% of all patients with intracranial SFT/HPC will develop extracranial metastases, most commonly to the bone, lung, or liver.[
Given that our patient had an excellent performance status and GTR of his intracranial disease, we felt that the aggressive treatment of his liver metastases was indicated. Due to his recent intracranial surgery and the preoperative pulmonary emboli, the decision was made to initially treat the hepatic lesions with SBRT alone. In addition, given the WHO Grade III histology, we also opted to treat the intracranial resection cavity with adjuvant CyberKnife stereotactic radiosurgery. When follow-up imaging demonstrated the mild progression of the liver metastases, systemic chemotherapy was advised. Two different regimens were offered: pazopanib alone versus combination temozolomide and bevacizumab. Given the hepatic location of the metastases and the history of pulmonary emboli, the decision was made to treat with temozolomide and bevacizumab. The patient completed 6 cycles and his subsequent MRIs demonstrated improvement in his disease burden. He is now 18 months from initial diagnosis and is being observed with surveillance MRIs of the brain and abdomen every 3 months. If future imaging studies demonstrate disease progression, evaluation of his original tumor specimens (brain and liver) for expression of pazopanib-targeted tyrosine kinase sequences and/or PD-L1 activity would be of particular utility to guide the selection of salvage therapies.
CONCLUSION
The average time from diagnosis of a primary intracranial SFT/HPC to the detection of metastatic disease is approximately 7.5 years.[
Declaration of patient consent
The authors certify that they have obtained IRB approval for this retrospective review and that all appropriate patient consent forms have been obtained. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understands that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Apra C, Alentorn A, Mokhtari K, Kalamarides M, Sanson M. Pazopanib efficacy in recurrent central nervous system hemangiopericytomas. J Neurooncol. 2018. 139: 369-72
2. Apra C, Mokhtari K, Cornu P, Peyre M, Kalamarides M. Intracranial solitary fibrous tumors/hemangiopericytomas: First report of malignant progression. J Neurosurg. 2018. 128: 1719-24
3. Bassiouni H, Asgari S, Hübschen U, König HJ, Stolke D. Intracranial hemangiopericytoma: Treatment outcomes in a consecutive series. Zentralbl Neurochir. 2007. 68: 111-8
4. Bastin KT, Mehta MP. Meningeal hemangiopericytoma: Defining the role for radiation therapy. J Neurooncol. 1992. 14: 277-87
5. Chakravarty BJ, Munn S, Lane MR. Hepatic metastasis from a meningeal haemangiopericytoma. Aust N Z J Med. 1991. 21: 884-5
6. Chen H, Zeng XW, Wu JS, Dou YF, Wang Y, Zhong P. Solitary fibrous tumor of the central nervous system: A clinicopathologic study of 24 cases. Acta Neurochir (Wien). 2012. 154: 237-48
7. Chen LF, Yang Y, Yu XG, Gui QP, Xu BN, Zhou DB. Multimodal treatment and management strategies for intracranial hemangiopericytoma. J Clin Neurosci. 2015. 22: 718-25
8. De Martin E, Coilly A, Guettier C, Samuel D. Liver metastases from meningeal hemangiopericytoma. Liver Int. 2015. 35: 2337-
9. Ebata T, Shimoi T, Bun S, Miyake M, Yoshida A, Shimomura A. Efficacy and safety of pazopanib for recurrent or metastatic solitary fibrous tumor. Oncology. 2018. 94: 340-4
10. Fargen KM, Opalach KJ, Wakefield D, Jacob RP, Yachnis AT, Lister JR. The central nervous system solitary fibrous tumor: A review of clinical, imaging and pathologic findings among all reported cases from 1996 to 2010. Clin Neurol Neurosurg. 2011. 113: 703-10
11. Fritchie K, Jensch K, Moskalev EA, Caron A, Jenkins S, Link M. The impact of histopathology and NAB2-STAT6 fusion subtype in classification and grading of meningeal solitary fibrous tumor/hemangiopericytoma. Acta Neuropathol. 2019. 137: 307-19
12. Ghia AJ, Allen PK, Mahajan A, Penas-Prado M, McCutcheon IE, Brown PD. Intracranial hemangiopericytoma and the role of radiation therapy: A population based analysis. Neurosurgery. 2013. 72: 203-9
13. Ghia AJ, Chang EL, Allen PK, Mahajan A, Penas-Prado M, McCutcheon IE. Intracranial hemangiopericytoma: Patterns of failure and the role of radiation therapy. Neurosurgery. 2013. 73: 624-30
14. Ghose A, Guha G, Kundu R, Tew J, Chaudhary R. CNS hemangiopericytoma: A systematic review of 523 patients. Am J Clin Oncol. 2017. 40: 223-7
15. Guthrie BL, Ebersold MJ, Scheithauer BW, Shaw EG. Meningeal hemangiopericytoma: Histopathological features, treatment, and long-term follow-up of 44 cases. Neurosurgery. 1989. 25: 514-22
16. Hukill PB, Lowman RM. Visceral metastasis from a meningioma: Report of a case. Ann Surg. 1960. 152: 804-8
17. Iwamuro M, Nakamura S, Shiraha H, Kobayashi Y, Fukatsu H, Yamamoto K. A case of primary intracranial hemangiopericytoma with hepatic metastases: Successful treatment with radiofrequency ablation and transcatheter arterial chemoembolization. Clin J Gastroenterol. 2009. 2: 30-5
18. Kaley TJ, Wen P, Schiff D, Ligon K, Haidar S, Karimi S. Phase II trial of sunitinib for recurrent and progressive atypical and anaplastic meningioma. Neuro Oncol. 2015. 17: 116-21
19. Kamamoto D, Ohara K, Kitamura Y, Yoshida K, Kawakami Y, Sasaki H. Association between programmed cell death ligand-1 expression and extracranial metastasis in intracranial solitary fibrous tumor/hemangiopericytoma. J Neurooncol. 2018. 139: 251-9
20. Kaneko T, Harada A, Isshiki K, Murakami H, Nakao A, Nonami T. Hemangiopericytomatous meningioma metastasized to the liver: Report of a case and review of the literature. Surg Today. 1993. 23: 644-8
21. Kim BS, Kim Y, Kong DS, Nam DH, Lee JI, Suh YL. Clinical outcomes of intracranial solitary fibrous tumor and hemangiopericytoma: Analysis according to the 2016 WHO classification of central nervous system tumors. J Neurosurg. 2018. 129: 1384-96
22. Kim JH, Jung HW, Kim YS, Kim CJ, Hwang SK, Paek SH. Meningeal hemangiopericytomas: Long-term outcome and biological behavior. Surg Neurol. 2003. 59: 47-53
23. Kinslow CJ, Bruce SS, Rae AI, Sheth SA, McKhann GM, Sisti MB. Solitary-fibrous tumor/hemangiopericytoma of the central nervous system: A population-based study. J Neurooncol. 2018. 138: 173-82
24. Lee SJ, Kim ST, Park SH, Choi YL, Park JB, Kim SJ. Successful use of pazopanib for treatment of refractory metastatic hemangiopericytoma. Clin Sarcoma Res. 2014. 4: 13-
25. Lo RC, Suriawinata AA, Rubin BP. Liver metastasis of meningeal hemangiopericytoma: A study of 5 cases. Clin Mol Hepatol. 2016. 22: 188-91
26. Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK. The 2016 world health organization classification of tumors of the central nervous system: A summary. Acta Neuropathol. 2016. 131: 803-20
27. Manatakis DK, Delis SG, Ptohis N, Korkolopoulou P, Dervenis C. Multidisciplinary approach to hepatic metastases of intracranial hemangiopericytoma: A case report and review of the literature. Case Rep Oncol Med. 2015. 2015: 214306-
28. Melone AG, D’Elia A, Santoro F, Salvati M, Delfini R, Cantore G. Intracranial hemangiopericytoma our experience in 30 years: A series of 43 cases and review of the literature. World Neurosurg. 2014. 81: 556-62
29. Nickerson TP, Fahy AS, Bingener J. Laparoscopic resection of intra-abdominal metastasis from intracranial hemangiopericytoma. Int J Surg Case Rep. 2015. 15: 50-3
30. Niwa M, Kobayashi T, Kuchiwaki H, Furuse M. A case of hemangiopericytoma with multiple extracranial metastasis: A case report. No Shinkei Geka. 1998. 26: 241-5
31. Park MS, Patel SR, Ludwig JA, Trent JC, Conrad CA, Lazar AJ. Activity of temozolomide and bevacizumab in the treatment of locally advanced, recurrent, and metastatic hemangiopericytoma and malignant solitary fibrous tumor. Cancer. 2011. 117: 4939-47
32. Patel AR, Flores BC, Ban VS, Hatanpaa KJ, Mickey BE, Barnett SL. Intracranial hemangiopericytomas: Recurrence, metastasis, and radiotherapy. J Neurol Surg B Skull Base. 2017. 78: 324-30
33. Ramakrishna R, Rostomily R, Sekhar L, Rockhill J, Ferreira M. Hemangiopericytoma: Radical resection remains the cornerstone of therapy. J Clin Neurosci. 2014. 21: 612-5
34. Ratneswaren T, Hogg FR, Gallagher MJ, Ashkan K. Surveillance for metastatic hemangiopericytoma-solitary fibrous tumors-systematic literature review on incidence, predictors and diagnosis of extra-cranial disease. J Neurooncol. 2018. 138: 447-67
35. Rutkowski MJ, Sughrue ME, Kane AJ, Aranda D, Mills SA, Barani IJ. Predictors of mortality following treatment of intracranial hemangiopericytoma. J Neurosurg. 2010. 113: 333-9
36. Schiariti M, Goetz P, El-Maghraby H, Tailor J, Kitchen N. Hemangiopericytoma: Long-term outcome revisited. Clinical article. J Neurosurg. 2011. 114: 747-55
37. Schutz FA, Choueiri TK, Sternberg CN. Pazopanib: Clinical development of a potent anti-angiogenic drug. Crit Rev Oncol Hematol. 2011. 77: 163-71
38. Spatola C, Privitera G. Recurrent intracranial hemangiopericytoma with extracranial and unusual multiple metastases: Case report and review of the literature. Tumori. 2004. 90: 265-8
39. Trifiletti DM, Mehta GU, Grover S, Sheehan JP. Clinical management and survival of patients with central nervous system hemangiopericytoma in the national cancer database. J Clin Neurosci. 2017. 44: 169-74
40. Yoshida K, Yamazaki S, Ota K, Makuuchi M, Hasegawa H, Takayasu K. A case report of meningioma with multiple liver metastases. Kanzo. 1988. 29: 1528-34
41. Zhang GJ, Zhang LW, Li D, Wu Z, Zhang JT. Analysis of prognostic factors, survival rates, and treatment in anaplastic hemangiopericytoma. World Neurosurg. 2017. 104: 795-801
42. Zweckberger K, Jung CS, Mueller W, Unterberg AW, Schick U. Hemangiopericytomas grade II are not benign tumors. Acta Neurochir (Wien). 2011. 153: 385-94