- Department of Neurological Surgery, University of Pittsburgh School of Medicine, Pittsburgh, PA, USA
- Department of Neurological Surgery, Oregon Health and Science University, Portland, OR, USA
- Department of Orthopedic Surgery, Oregon Health and Science University, Portland, OR, USA
Correspondence Address:
D. Kojo Hamilton
Department of Neurological Surgery, University of Pittsburgh School of Medicine, Pittsburgh, PA, USA
DOI:10.4103/2152-7806.180771
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ozpinar A, Liu JJ, Tempel ZJ, Choi PA, Hart RA, Hamilton DK. Intracranial pressure monitoring during adult spinal deformity correction in a patient with critical venous occlusive disease and superior vena cava syndrome: A technical note. Surg Neurol Int 20-Apr-2016;7:47
How to cite this URL: Ozpinar A, Liu JJ, Tempel ZJ, Choi PA, Hart RA, Hamilton DK. Intracranial pressure monitoring during adult spinal deformity correction in a patient with critical venous occlusive disease and superior vena cava syndrome: A technical note. Surg Neurol Int 20-Apr-2016;7:47. Available from: http://surgicalneurologyint.com/surgicalint_articles/intracranial-pressure-monitoring-during-adult-spinal-deformity-correction-in-a-patient-with-critical-venous-occlusive-disease-and-superior-vena-cava-syndrome-a-technical-note/
Abstract
Background:Intracranial pressure (ICP) monitoring is not routinely used during complex spinal deformity correction surgery. The authors report a 66-year-old male who during thoracolumbar deformity surgery required the placement of an ICP monitor due to the underlying history of a superior vena cava syndrome (e.g., s/p right jugular stent).
Case Description:A 66-year-old male with multiple prior lumbar spinal procedures presented with lower back and bilateral lower extremity pain, paresthesias, and weakness. He had a history of chronic left internal jugular and brachiocephalic venous occlusion (e.g., he had a right internal jugular stent). During deformity surgery, a frontal intraparenchymal ICP monitor was placed. During the early portion of the operation, bed adjustments (increasing reverse trendelenburg position) were required to compensate for ICP elevations as high as 30 mm Hg. A subsequent inadvertent durotomy during decompression lowered the ICP to
Conclusion:ICP monitoring may be a useful adjunct for patient safety in selected patients who are at risk for developing intracranial hypertension during extensive spinal deformity surgery.
Keywords: Deformity correction, intracranial pressure monitoring, superior vena cava syndrome
INTRODUCTION
Intracranial pressure (ICP) monitoring is frequently used by neurosurgeons for the management of multiple pathologies including traumatic brain injury, hydrocephalus, and idiopathic intracranial hypertension.[
CASE REPORT
Presentation
A 66-year-old male with multiple prior lumbar spine surgeries presented with intractable, disabling low back and radicular pain, and neurogenic claudication with severe sagittal imbalance refractory to conservative measures. He was unable to walk more than 20 yards at a time and experienced severe pain and cramping with any movement. His quality of life had been severely affected by his symptoms. He had a history of SVC syndrome (right internal jugular stent), managed with long-term warfarin anticoagulation; he also had chronic left internal jugular and brachiocephalic veins occlusions.
Physical examination demonstrated normal motor strength in the upper extremities. No pathologic reflexes were noted in any extremity. He had a slight weakness in the right gastrocnemius and extensor hallicus longus (EHL) graded at 4+/5 strength.
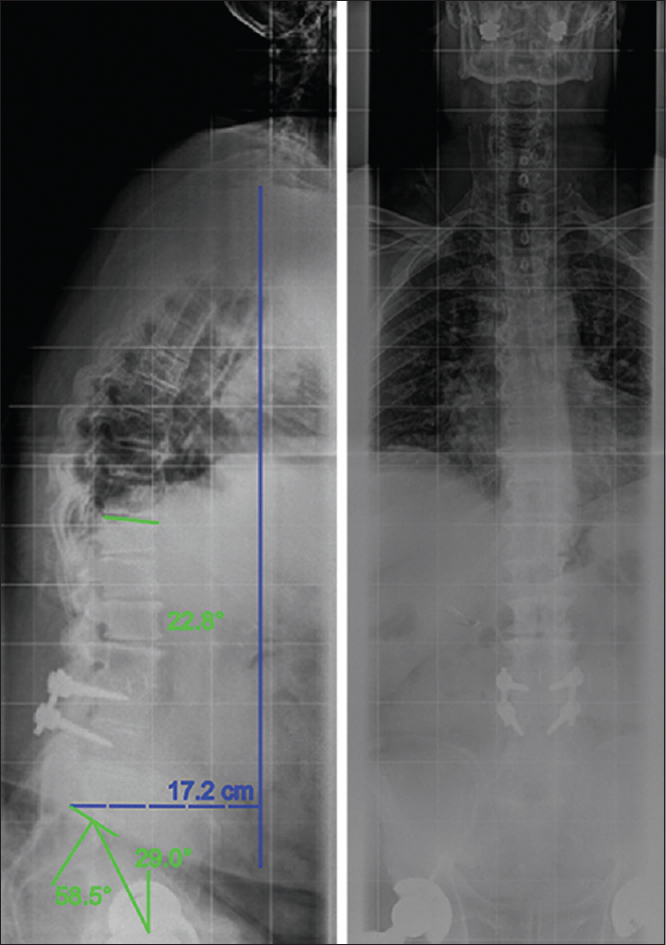
Computed tomography (CT) and plain radiographics indicated pseudarthrosis of his prior construct [
Description of intraoperative procedures
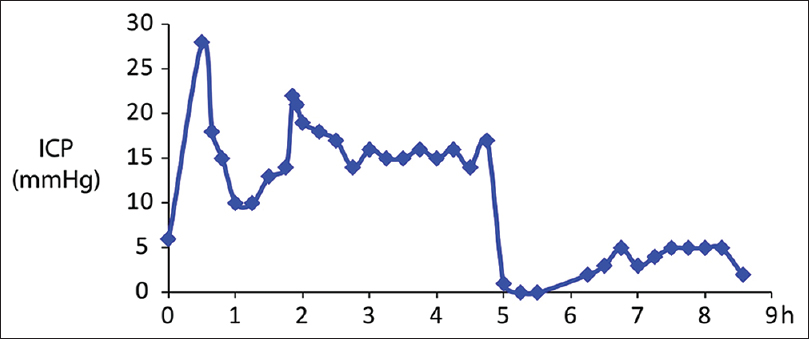
After induction of general endotracheal anesthesia, electrodes were placed for routine neuromonitoring including somatosensory and motor evoked potentials as well as electromyography. Subsequently, a right frontal ICP monitor was placed. The initial pressure reading from the ICP monitor was 6 mm Hg (normal ICP <20 mm Hg). Once all lines and monitors were in place, the patient was then carefully turned prone onto the Jackson frame. The prone-view facial padding device notably needed to be modified to accommodate the anchor of the pressure monitor. A semicircular cut was made, so the anchor would rest without strain on the device. The pressure reading after turning the patient prone was 30 mm Hg. The decision was made to only proceed with the case if the ICP could be maintained <15 mm Hg at rest since surgical manipulation would likely lead to elevation of the ICP. The Jackson frame was then elevated to 30° of reverse Trendelenburg position. The patient's ICP gradually decreased over the next 10 min and stabilized at 14–15 mm Hg [
After successful instrumentation, laminectomies and a discectomy were performed. Next, an interbody spacer was then placed at the L2–L3 level via a transforaminal approach. A PSO was then performed at L2. During the exposure and removal of scar in preparation for the PSO, a durotomy occurred on the right side (approximately 5 h into the procedure) [
Once the PSO was closed, intraoperative long-cassette radiographs were obtained demonstrating significant improvement in lordosis. Two-column Smith-Peterson type osteotomies were then carried out, long segment rods were contoured, cap screws were placed and tightened, and the wound was closed expeditiously.
The patient was then carefully turned back supine onto the bed and taken directly to the Intensive Care Unit still intubated and in stable condition. The head of the bed was maintained at 30° as a compromise between improving cerebral venous drainage and protecting the dural closure.
Postoperative course
ICPs were monitored overnight to evaluate for intracranial hypertension; however, the pressures remained in the normal range. His postoperative examination was grossly stable compared with his preoperative examination. The ICP monitor was removed, and he was extubated shortly thereafter. On discharge, he had significant improvement in his preoperative pain, and he continued to have stable slight weakness of the right EHL and gastrocnemius.
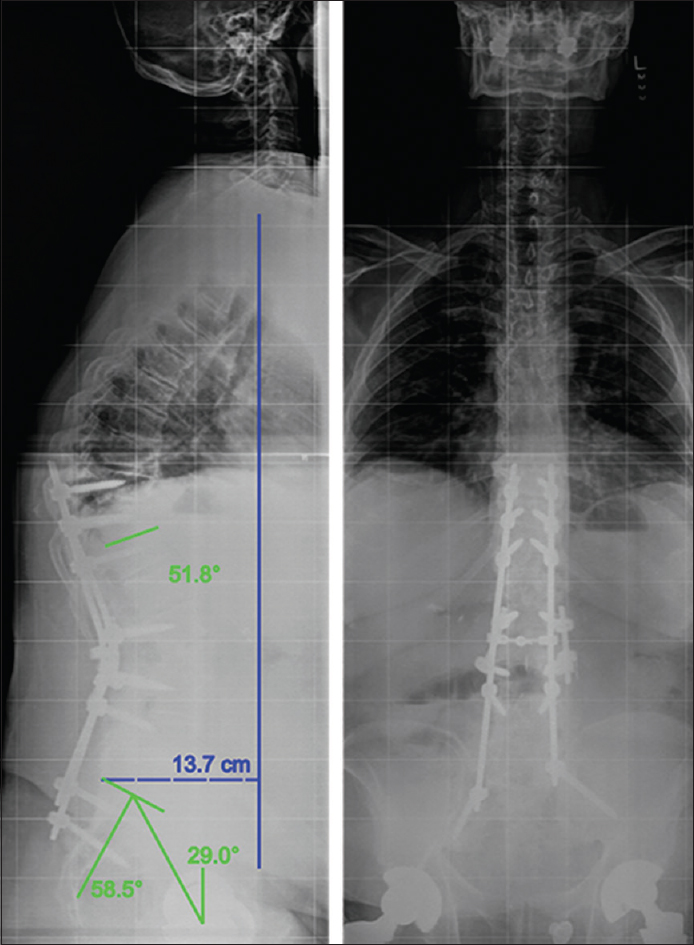
Standing long-cassette radiographs were obtained at his 3-month postoperative visit and revealed slight proximal junctional kyphosis. His postoperative pelvic incidence was 58.5°, his pelvic tilt was 29°, and his sagittal vertical axis (SVA) was 13.7 cm [
DISCUSSION
Several traditional etiologies such as infections, tumors, and fibrosing mediastinitis can cause varying degrees of central venous system obstruction leading to the diagnosis of SVC syndrome. More recently, accurate diagnosis of hypercoagulable states, placement of indwelling central venous devices or pacemaker leads as a cause of SVC syndrome has drawn more attention to the diagnosis.[
During a prone procedure with limited ability for standard maneuvers to increase venous return (such as elevating the head), the ICP monitor, in this case, was a very useful monitoring adjunct to ensure safety.[
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Anderson RC, Kan P, Klimo P, Brockmeyer DL, Walker ML, Kestle JR. Complications of intracranial pressure monitoring in children with head trauma. J Neurosurg. 2004. 101: 53-8
2. Biousse V. Idiopathic intracranial hypertension: Diagnosis, monitoring and treatment. Rev Neurol (Paris). 2012. 168: 673-83
3. Cavanilles-Walker JM, Tomasi SO, Sgier F, Kröber M. Remote cerebellar haemorrhage after lumbar spine surgery: Case report. Arch Orthop Trauma Surg. 2013. 133: 1645-8
4. del Río Solá ML, Fuente Garrido R, Gutiérrez Alonso V, Vaquero Puerta C. Endovascular treatment of superior vena cava syndrome caused by malignant disease. J Vasc Surg. 2014. 59: 1705-6
5. Feldman Z, Kanter MJ, Robertson CS, Contant CF, Hayes C, Sheinberg MA. Effect of head elevation on intracranial pressure, cerebral perfusion pressure, and cerebral blood flow in head-injured patients. J Neurosurg. 1992. 76: 207-11
6. Gelabert-González M, Ginesta-Galan V, Sernamito-García R, Allut AG, Bandin-Diéguez J, Rumbo RM. The Camino intracranial pressure device in clinical practice. Assessment in a 1000 cases. Acta Neurochir (Wien). 2006. 148: 435-41
7. Gill B, Heavner JE. Postoperative visual loss associated with spine surgery. Eur Spine J. 2006. 15: 479-84
8. Herscovici R, Szyper-Kravitz M, Altman A, Eshet Y, Nevo M, Agmon-Levin N. Superior vena cava syndrome – Changing etiology in the third millennium. Lupus. 2012. 21: 93-6
9. Hohloch K, Bertram N, Trümper L, Beissbarth T, Griesinger F. Superior vena cava syndrome caused by a malignant tumor: A retrospective single-center analysis of 124 cases. J Cancer Res Clin Oncol. 2014. 140: 2129-34
10. Horton JC. Continuous intracranial pressure monitoring: A last resort in pseudotumor cerebri. J Neuroophthalmol. 2011. 31: 199-201
11. Kuo JR, Yeh TC, Sung KC, Wang CC, Chen CW, Chio CC. Intraoperative applications of intracranial pressure monitoring in patients with severe head injury. J Clin Neurosci. 2006. 13: 218-23
12. Martinez-Manas R, Rodriguez E, Garcia C. Clinical experience of intracranial pressure monitoring. Retrospective study in 239 Camino (R) monitored patients. Neurocirugia. 1999. 10: 291-6
13. Mauritz W, Steltzer H, Bauer P, Dolanski-Aghamanoukjan L, Metnitz P. Monitoring of intracranial pressure in patients with severe traumatic brain injury: An Austrian prospective multicenter study. Intensive Care Med. 2008. 34: 1208-15
14. Münch E, Weigel R, Schmiedek P, Schürer L. The Camino intracranial pressure device in clinical practice: Reliability, handling characteristics and complications. Acta Neurochir (Wien). 1998. 140: 1113-9
15. Patil CG, Lad EM, Lad SP, Ho C, Boakye M. Visual loss after spine surgery: A population-based study. Spine (Phila Pa 1976). 2008. 33: 1491-6
16. Poca MA, Sahuquillo J, Arribas M, Báguena M, Amorós S, Rubio E. Fiberoptic intraparenchymal brain pressure monitoring with the Camino V420 monitor: Reflections on our experience in 163 severely head-injured patients. J Neurotrauma. 2002. 19: 439-48
17. Roth C, Ferbert A, Deinsberger W, Kleffmann J, Kästner S, Godau J. Does prone positioning increase intracranial pressure?. A retrospective analysis of patients with acute brain injury and acute respiratory failure. Neurocrit Care. 2014. 21: 186-91
18. Wu JC, Chen YC, Liu L, Chen TJ, Huang WC, Thien PF. The risk of stroke after spinal fusion surgery: A national cohort study. Spine J. 2012. 12: 492-9