- Department of Neurosurgery, IRCCS Galeazzi, Milan, Italy,
- Department of Neurology, Center for Neurorehabilitation and Paraplegiology, Basel, Switzerland.
Correspondence Address:
Domenico Servello, Department of Neurosurgery, IRCCS Galeazzi, Milan, Italy.
DOI:10.25259/SNI_349_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Domenico Servello1, Christian Saleh2, Edvin Zekaj1. Intraoperative mobile computed tomography in deep brain stimulation: Comparison between Airo CT and O-arm CT. 17-Jun-2022;13:258
How to cite this URL: Domenico Servello1, Christian Saleh2, Edvin Zekaj1. Intraoperative mobile computed tomography in deep brain stimulation: Comparison between Airo CT and O-arm CT. 17-Jun-2022;13:258. Available from: https://surgicalneurologyint.com/surgicalint-articles/intraoperative-mobile-computed-tomography-in-deep-brain-stimulation-comparison-between-airo-ct-and-o-arm-ct/
Abstract
Background: A new intraoperative mobile device, called Airo computed tomography (CT), is becoming increasingly used in surgery adding to the current most widespread intraoperative imaging in form of the O-arm CT device. Intraoperative CT imaging has the advantage to reduce the discomfort derived from the patient’s transfer from the operative room to the radiological unit and also the time of control or time of reposition in cases of lead misplacement. This is the first rapport on Airo CT device application in DBS surgery.
Methods: In our retrospective study, we have evaluated 52 patients who had DBS from October 2020 to November 2021. All patients underwent a preoperative brain magnetic resonance imaging (MRI) and a stereotactic brain CT scan. We performed an intraoperative CT scan with the Airo device. Images were subsequently transferred to the neuronavigation system (BrainLab cranial software) and merged with preoperative planning images to confirm correct position of leads. Five aspects were compared: patient and surgeon comfort, surgical planning, parenchymal visualization, radioscopic lead visualization, and costs.
Results: Both imaging devices are easy-to-use, precise, and safe and have their pros and cons.
Conclusion: This is the first study reporting on CT-Airo application in DBS. We advise that if only DBS is considered, the O-arm application might be more suitable. If in addition to DBS cranial surgery is performed, the Airo device is more suitable.
Keywords: Airo CT, Cranial surgery, Deep brain stimulation, Imaging, O-arm CT
INTRODUCTION
Deep brain stimulation (DBS) efficacy depends foremost on two main factors, (a) patient selection and (b) correct lead positioning; the former depends on the experience of the multidisciplinary DBS equip, while the latter on the experience of the neurosurgeon.
To verify lead positioning in most centers, radiological evaluation is done, using lateral and anteroposterior X-ray, while intraoperative CT scan or MRI imaging is present in few centers.
Intraoperative CT has the advantage in improving surgical precision and reducing for the patient and the surgical equip discomfort due to the patient’s transfer from the operating room to the radiological unit reducing the time of control or time of reposition in cases of lead misplacement.
At present, two intraoperative CT scan systems are available: O-arm (Medtronic) and Airo CT (BrainLab), while the experience with O-arm is well consolidated (26 papers published so far), Airo CT is becoming increasingly more used in surgery.
In this paper, we present the specific workflow on the use of intraoperative CT-Airo system and differences compared to O-arm. To the best of our knowledge, this is the first report of intraoperative Airo use in DBS.
MATERIALS AND METHODS
We retrospectively analyzed 52 patients who underwent DBS at our unit. Intraoperative electrodes position was confirmed with Airo CT scan.
Surgery was performed for Parkinson disease (47 patients), essential tremor (two patients), obsessive-compulsive disorder (three patients), and major depression (one patient). There were 17 females and 35 males. Targets were in 32 patients the subthalamic nucleus, in 14 patients the posterolateral globus pallidus internus, in two patients the anteromedial globus pallidus internus, in two patients the posterior subthalamic area, in one patient the middle forebrain bundle, and in one patient the bed nucleus of the stria terminalis.
This study did not need an ethics committee approval as retrospective in nature. Patient’s consent was obtained for surgery and publication.
Workflow description
One day before surgery, we perform a brain magnetic resonance imaging (MRI 1.5 Tesla), which consists in a volumetric gadolinium-enhanced T1-weighted sequence and axial T2-weighted or proton-density (PD) images, with a 2 mm slice thickness. On the day of surgery after positioning the Cosman-Roberts-Wells stereotactic frame, a stereotactic CT scan is acquired. MRI and CT scan images are transferred to the BrainLab planning station and fused together. The choice of the target is based on direct viewing on MRI. The BrainLab creates automatically based on each patient specific neuroanatomy 3D objects that include critical structures, the region of interest, and the basal ganglia territory. Tissue-based automatic segmentation allows to define the exact anatomy of each patient for precise individual-based targeting, overcoming the limitations of the more traditional approach of indirect targeting based on atlas coordinates. To avoid sulcal vessels and ventricles for each side of implantation, the trajectories are computer-based determined. Once stereotactic target coordinates and ring and arc frame values are obtained, the surgical procedure begins.
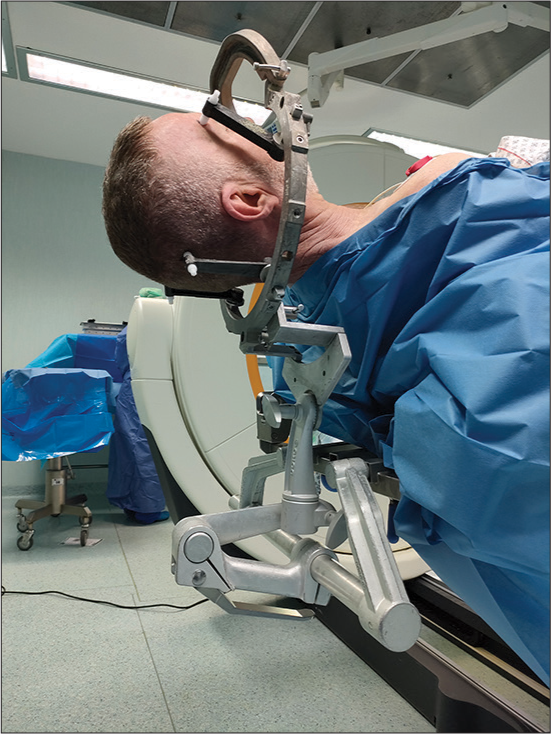
The patient is positioned on the operating table integrated to the Airo system with the frame fixed on the Mayfield head holder. At this point, it is imperative to achieve a correct assembly of frame and head holder which must be in a vertical position to avoid collision with Airo system and to permit intraoperative CT scan image acquisition with gantry 0° [
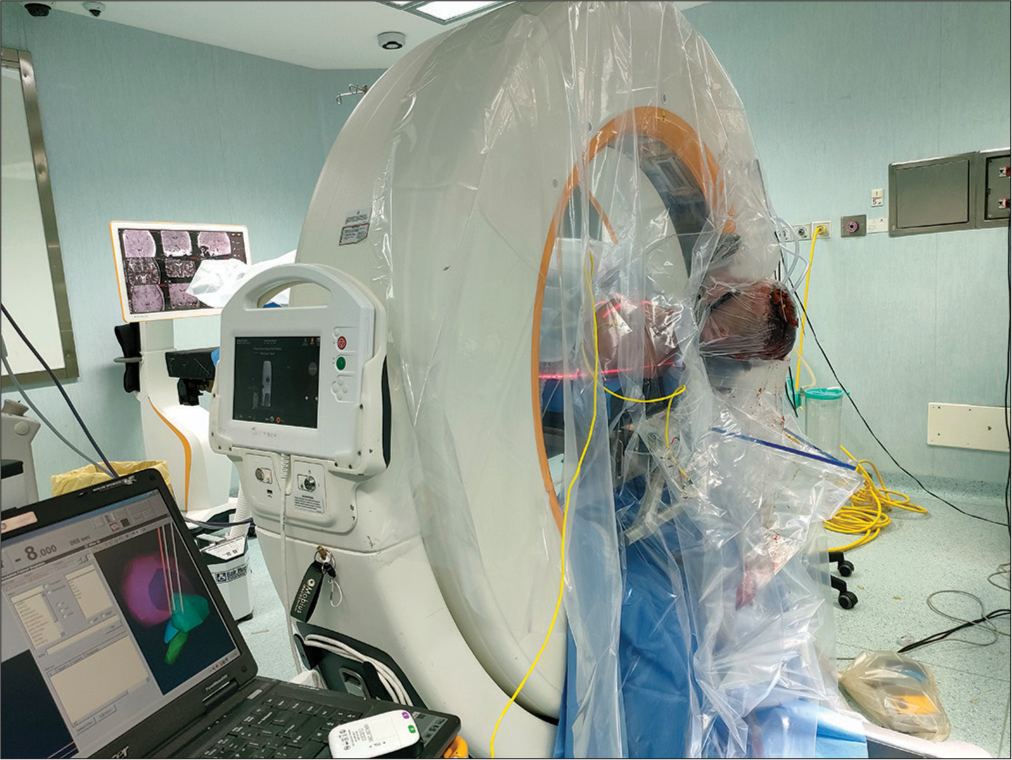
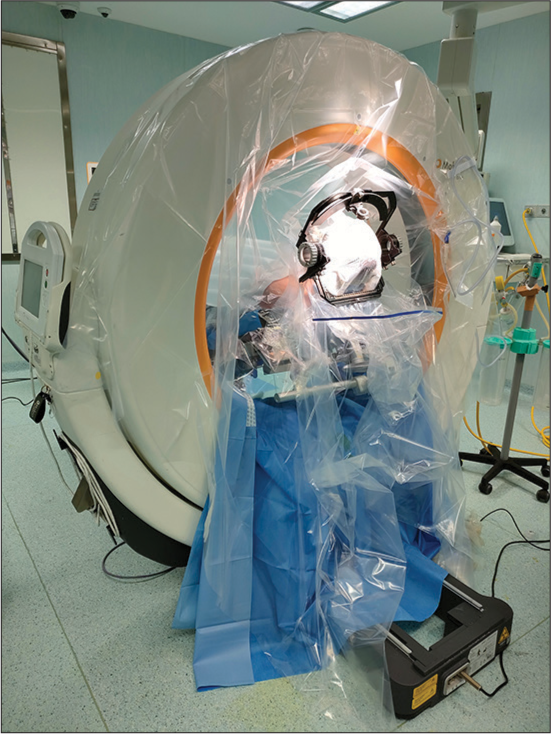
Lasers of Airo are used to evaluate the craniocaudal and mediolateral extension of the CT and the first and final acquiring positions are registered. Then, a scout image that includes the area of interest is obtained to confirm the correct extension of intraoperative CT scan. The images obtained are then transferred to the neuronavigation system and fused with preoperative planning and the uploaded trajectories. If images confirm correct lead positioning, the second lead is implanted, while in case of a distance of more than 2 mm from the chosen target to the implanted site, the lead is repositioned.
In the 1st postoperative day, we perform a CT scan and MRI that include axial T2-weighted or PD images which are then transferred to the planning station.
The DBS implants (electrode contracts, orientation in 3D, and targets) are automatically detected and visualized by the Elements Lead Localization.
RESULTS
In the first intraoperative CT scan, acquiring the arc system was left in situ, how we did we the O-arm. This, however, caused important artifacts requiring a repeat of the CT scan. In all subsequent patients, the arc system was removed avoiding successfully arc system-induced artifacts.
In all patients, fusion of intraoperative CT scan with presurgical planning was optimal which permits to compare final lead position with the one selected in the planning. Intraoperative CT scan allowed immediate reposition in 2 cases (1.9%) for leads with a target error greater than 2 mm.
One patient presented a deterioration of consciousness and the Airo CT imaging detected an intraparenchymal hematoma with mass effect. An emergent surgical evacuation was performed without having to move the patient from the operating room.
In another patient, an epileptic seizure required interruption of the procedure. In this case, we could perform with the Airo CT device prompt imaging excluding a hemorrhage.
DISCUSSION
In our experience, Airo CT proved to be safe and effective: we obtained fusion successfully in all patients: postoperative CT and MRI confirmed correct leads positioning in all cases. In one case, CT was able to detect a hemorrhagic complication immediately. However, we did notice some significant differences between the two systems, which we discuss here under.
Comparison between O-Arm and Airo
We had a large experience with O-arm from 2009 until 2020 (more than 500 patients treated) and 1 year of experience with Airo CT.
Several points between these two intraoperative imaging systems need comparison [ Patient and surgeon comfort, Lead location, Parenchymal visualization, Radioscopic lead visualization and Costs.
Patient and surgeon comfort
On patient comfort during surgical procedure, this is identical for both Airo and O-arm as with correct positioning (backup and anti-Trendelenburg), the patient is quite comfortable. While during images acquisition with Airo, we must change the patient’s position until the head is perfectly vertical, to adapt to a gait entry 0°. This position is not comfortable for the patient. With the O-arm device, the gantry is adapted according to the patient’s position without having to move the operative table. On surgeon’s comfort with Airo system, due to sterility measures adopted, the surgeon has to work in proximity of the Airo tracks, which creates discomfort [
Unfortunately in DBS surgery, due to sterility measures adopted, it is not possible turning the operative table 90°, which would allow the surgeon to work more comfortably.
Another problem with Airo is the necessity to remove the ring of the frame every time a CT scan has to be done, otherwise artifacts preclude fusion. This is not necessary with O-arm CT imaging where images are acquired with frame ring.
Surgical planning
As each system is coupled with own planning station, we must compare BrainLab to Stealth. Both are easy-to-use and reliable. The former, tissue-based automatic segmentation, is superior for reliable extraction of stimulation target structures allowing a more precise targeting above all for GPi as it is often less visible on MRI in its entirety. BrainLab guide XT allows reconstruction of the lead in relation to the target suggesting the best contacts to use.
Parenchymal visualization
An advantage of Airo in respect to O-arm is that, as a true CT scanner, the images produced provide for better soft-tissue definition of the brain parenchyma which permits evaluation of bleeding complication and enables more critical verification of image fusion through comparison of the cerebral ventricles and other soft-tissue landmarks.
Radioscopic lead visualization
Radioscopic images are utilized to evaluate lead orientation when we use directional lead. To date with Airo-CT it is not possible to acquire radioscopic images, in contrast to O-Arm, therefore an evaluation of the marker in the directional lead and of the correct positioning of the same is not possible. Both systems were reliable on lead localization, which was confirmed with postoperative CT scan and MRI.
Costs
Airo has higher costs than O-arm, at our institution, we have paid inclusive the navigator 1.200.000 €. For the O-arm device, the estimate of costs was 800.000 €.[
CONCLUSION
There is already some literature describing Airo application in spinal surgery[
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Faust K, Schneider GH, Vajkoczy P. Utilization of the intraoperative mobile AIRO(R) CT scanner in stereotactic surgery: Workflow and effectiveness. Stereotact Funct Neurosurg. 2019. 97: 303-12
2. Hecht N, Kamphuis M, Czabanka M, Hamm B, König S, Woitzik J. Accuracy and workflow of navigated spinal instrumentation with the mobile AIRO((R)) CT scanner. Eur Spine J. 2016. 25: 716-23
3. Malham GM, Wells-Quinn T. What should my hospital buy next?-Guidelines for the acquisition and application of imaging, navigation, and robotics for spine surgery. J Spine Surg. 2019. 5: 155-65
4. Scarone P, Vincenzo G, Distefano D, Del Grande F, Cianfoni A, Presilla S. Use of the Airo mobile intraoperative CT system versus the O-arm for transpedicular screw fixation in the thoracic and lumbar spine: A retrospective cohort study of 263 patients. J Neurosurg Spine. 2018. 29: 397-406