- Departments of Neurosurgery,
- Departments of Pathology, University Hospital Puerta de Hierro Majadahonda, Madrid, Spain.
Correspondence Address:
Xavier A. Santander
Departments of Neurosurgery,
DOI:10.25259/SNI-104-2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Xavier A. Santander, Raquel Gutiérrez-González, Carlos Cotúa, Eva Tejerina, Gregorio-Boto Rodríguez. Intraventricular cryptococcoma mimicking a neoplastic lesion in an immunocompetent patient with hydrocephalus: A case report. 25-Jun-2019;10:115
How to cite this URL: Xavier A. Santander, Raquel Gutiérrez-González, Carlos Cotúa, Eva Tejerina, Gregorio-Boto Rodríguez. Intraventricular cryptococcoma mimicking a neoplastic lesion in an immunocompetent patient with hydrocephalus: A case report. 25-Jun-2019;10:115. Available from: http://surgicalneurologyint.com/surgicalint-articles/9401/
Abstract
Background: The aim of this study is to highlight the importance of cryptococcosis as one of the most common fungal infections of the central nervous system, stressing the consideration of a cryptococcoma within the list of differential diagnosis of intraventricular masses in immunocompetent hosts.
Case Description: We present the case of an immunocompetent 41-year-old female from Philippines with an intraventricular cryptococcoma due to Cryptococcus neoformans, mimicking an intraventricular primary brain tumor, who had hydrocephalus. She was approached as having a neoplastic lesion and underwent surgical resection plus third ventriculostomy through endoscopy. Later in her evolution, we realized the infectious nature of the lesion, and antifungal systemic therapy was initiated. In the end, she needed the placement of a ventriculoperitoneal shunt to maintain a good neurologic status. Despite all our efforts, she had a fatal outcome due to various complications.
Conclusion: Our case is the first intraventricular cryptococcoma due to C. neoformans, as far as we know, to be reported in English, German, or Spanish literature. Our report stresses the importance to consider this kind of infection in immunocompetent patients, to raise the level of suspicion of this diagnosis, and to know the complications and management options.
Keywords: Cryptococcoma, Cryptococcus neoformans, Hydrocephalus, Immunocompetent, Intraventricular, Neoplastic, Neuroendoscopy
INTRODUCTION
The encapsulated yeasts Cryptococcus neoformans and Cryptococcus gattii cause invasive fungal infections known as cryptococcosis. It is among the most common fungal infection in patients with human immunodeficiency virus (HIV).[
CASE REPORT
A 41-year-old Filipino female, with no medical record of interest, presented with a history of 4 months of gait imbalance, occasional urinary incontinence, and memory impairment. She was afebrile; laboratory investigations were normal; and the chest X-ray showed no evidence of pathology. Chronic hydrocephalus of the adult was initially suspected.
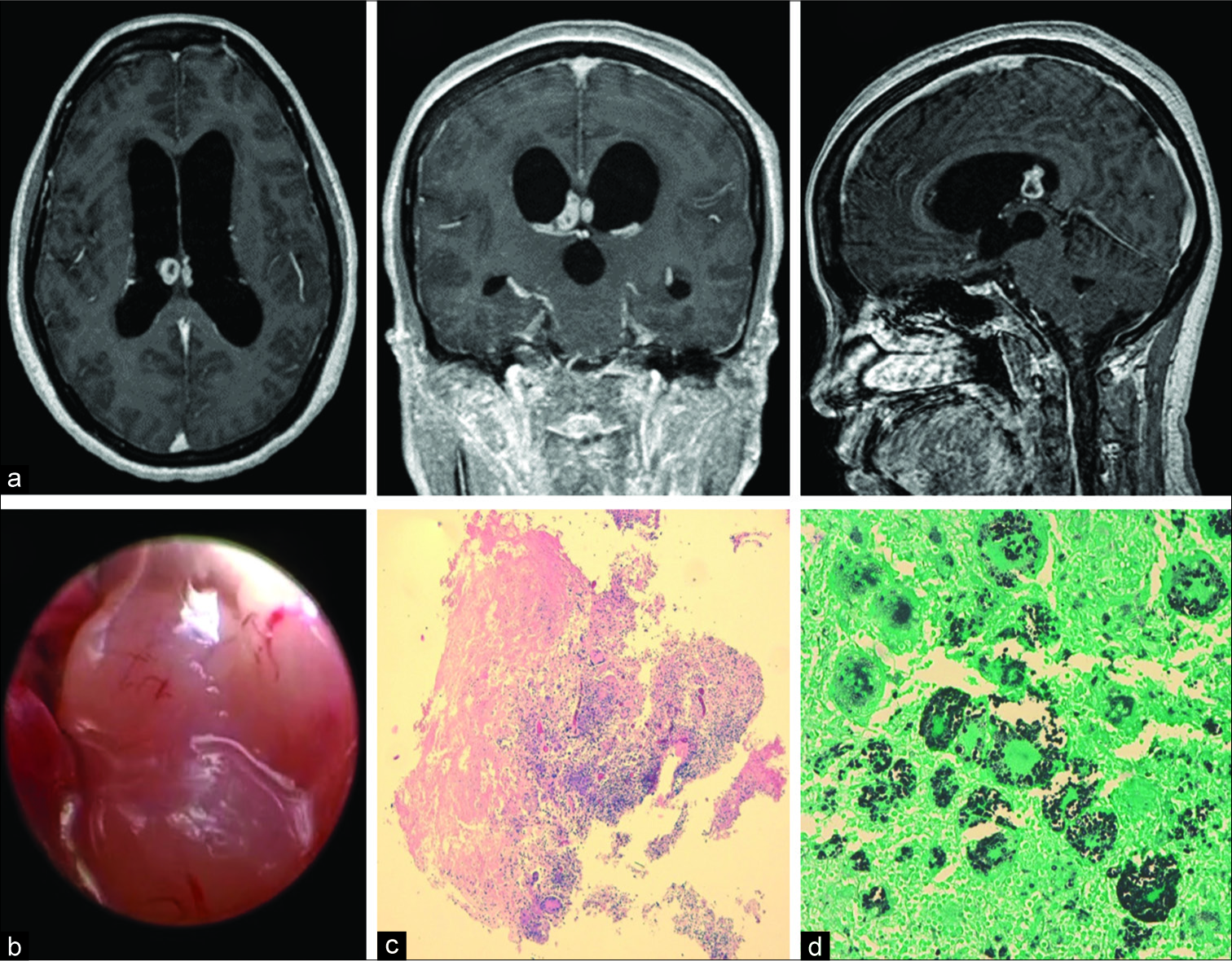
A computed tomography (CT) of the head was performed. It showed an intraventricular mass attached to the posterior third of the septum pellucidum and the fornix commissure and enlargement of the lateral and third ventricles. A magnetic resonance image confirmed an intraventricular polylobulated tumor of 1.6 cm in diameter, enhancing in a ring-like fashion with gadolinium. Furthermore, active hydrocephalus was present [
Figure 1
(a) Initial magnetic resonance imaging sequences confirmed a right predominant mass with biventricular expression clearly attached to the septum pellucidum and invasion of the fornix commissure, as shown in axial, coronal, and sagittal T1WI C+ sequences, with gadolinium enhancement of the lesion in a ring-like fashion. (b) The lesion, as seen through the endoscope, was polylobulated, avascular, shiny, and of fibrous consistency. (c) Intraoperative examination showed a nonencapsulated, partially necrotic lesion surrounded by a dense cellular component that included “atypical” multinucleated cells with hyperchromatic nuclei (Hematoxylin-Eosin, ×2). (d) Numerous round yeasts with a characteristic thick capsule were found both in the cytoplasm of the histiocytes and giant cells (Gomori Methenamine silver stain, ×20).
She underwent removal of the lesion plus third ventriculostomy through endoscopy [
The EVD was removed after 72 h. Routine culture of the EVD tip identified Cryptococcus spp. (>15 colony-forming-units). Cultures confirmed C. neoformans. Systemic antifungal treatment was given immediately. We started amphotericin B 400 mg q24h IV plus 5-flucytosine 1500 mg q6h IV. Blood tests for HIV Types 1 and 2 were negative. Immunoglobulin levels and CD4 cell counts were normal.
The definitive histology identified numerous round yeasts with a characteristic thick capsule found both inside the cytoplasm of the histiocytes and giant cells, confirming its fungal nature [
Four days later, she deteriorated and she developed a clinical situation of perplexity, reiterative speech, and bradypsychia and progressive loss of consciousness. Worsening ventriculomegaly was revealed in a head CT scan. She was moved to the intensive care unit (ICU) and a new EVD was placed. In the subsequent days, she had complications consisting in malfunction due to obstruction in the setting of infection and intraventricular hemorrhage. Several EVD replacements and raised intracranial pressure (ICP) despite maximal antifungal therapy led us to place a ventriculoperitoneal (VP) shunt. Neurological status was recovered at the beginning. Eventually, she showed neurological worsening, intraventricular hemorrhage, and VP malfunction. CSF cultures remained positive and no bacterial coinfection was reported. New CSF analysis from the EVD demonstrated worsening compatible with ventriculitis. In the end, after 10 days of antifungal treatment, the patient died due to complications related to infection, electrolyte imbalance, and multiorganic failure.
DISCUSSION
In this article, we describe an infection caused by C. neoformans in an immunocompetent patient, which is a rare sighting. Nonetheless, there are many reports describing this agent in these patients.[
C. neoformans is found globally and is related with avian feces, whereas C. gattii is prevalent in zones with tropical climates, grows in soil, is not found on avian guanos, and has a relationship with eucalypt trees.[
There are two forms of cryptococcosis in the central nervous system (CNS): meningeal and parenchymal. The first one is the most frequent. The intraventricular formation of cryptococcomas is a rare finding. In a short review of 17 cases, only one was intraventricular.[
Cryptococcomas can be mistaken for malignant lesions in half of cases. Accurate diagnosis is only achieved following histological analysis.[
CSF analysis can reveal some important features such as high opening pressure, positive India ink stain, and detection of its capsular antigen. Important note is to point out that we achieved the diagnosis by culturing the EVD tip after the first surgery, which is a routine behavior after removal in our department.
The medical treatment of choice is the systemic antifungal regimen as a two-step approach: induction and consolidation.[
Antifungal therapy alone is unlikely to penetrate large lesions enough to lead a remission. In these cases, if the lesion is accessible, a cryptococcoma should be treated by a combination of surgical and medical approaches as suggested by Ulett et al.[
In the presence of meningitis, the most important prognostic factor is increased ICP.[
We think our patient developed meningitis after Cryptococcus seeding during her first surgery (mass resection), and most probably related to the fact that she did not receive any antifungal treatment before the intervention. Our case was approached as a glioma, and the working space was exclusively inside the ventricles. We assume that we broke the cryptococcoma capsule during surgery which contributed to seeding and the subsequent ventriculitis and meningoencephalitis. Such risks are reported in the literature and are the main cause of death in these patients.[
Cryptococcomas, as fungal granulomas in general, have bad prognosis even in light of medical treatment and surgery, especially if the intraventricular space is violated during the procedure, causing the subsequent infection. Yeh et al. reported a case of a parenchymal cryptococcoma in a non- HIV patient who, despite the medical treatment and surgical resection, died in the end.[
Despite our efforts and multidisciplinary treatment, our patient died. She had a vegetative status before her death. This development is not extraordinary in such setting, even in immunocompetent patients.[
CONCLUSION
Cryptococcosis is misdiagnosed in HIV-negative patients because of the misconception that it only affects immunocompromised patients. Cryptococcomas have no specific radiologic findings and sometimes, even the histology can be confused with CNS tumors. When the ventricles are compromised, a course of antifungal treatment should be initiated before any surgical procedure. The mainstay of treatment of any fungal granuloma should be radical surgical resection (especially if it is >3 cm in diameter), antifungal therapy, and aggressive treatment of ICH. Placement of a VP shunt should be considered as part of the arsenal if CSF cultures remain positive in spite of optimal antifungal therapy. Finally, our case highlights the importance to obtain cultures from the tip of ventricular catheters as a routine behavior in any neurosurgical procedure.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understands that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
References
1. Calvo A, Hernández P, Spagnuolo E, Johnston E. Surgical treatment of intracranial hypertension in encephalic cryptococcosis. Br J Neurosurg. 2003. 17: 450-5
2. Colom-Valiente MF, Frasés S, Ferrer C, Martín-Mazuelos E, de Mendoza MH, Torres-Rodríguez M. Epidemiologic study of cryptococcosis in Spain: First results. Rev Iberoam Micol. 2001. 18: 99-104
3. Corti M, Priarone M, Negroni R, Gilardi L, Castrelo J, Arechayala A. Ventriculoperitoneal shunts for treating increased intracranial pressure in cryptococcal meningitis with or without ventriculomegaly. Rev Soc Bras Med Trop. 2004. 47: 524-7
4. Dubey A, Patwardhan RV, Sampth S, Santosh V, Kolluri S, Nanda A. Intracranial fungal granuloma: Analysis of 40 patients and review of the literature. Surg Neurol. 2005. 63: 254-60
5. Ellis DH, Pfeifer TJ. Natural habitat of Cryptococcus neoformans var. Gattii. J Clin Microbiol. 1990. 28: 1642-4
6. Harris JR, Lockhart SR, Debess E, Marsden-Haug N, Goldoft M, Wohrle R. Cryptococcus gattii in the United States: Clinical aspects of infection with an emerging pathogen. Clin Infect Dis. 2011. 53: 1188-95
7. Li Q, You C, Liu Q, Liu Y. Central nervous system cryptococcoma in immunocompetent patients: A short review illustrated by a new case. Acta Neurochir. 2010. 152: 129-36
8. Liu L, Zhang R, Tang Y, Lu H. The use of ventriculoperitoneal shunts for uncontrollable intracranial hypertension in patients with HIV-associated cryptococcal meningitis with or without hydrocephalus. Biosci Trends. 2014. 8: 327-32
9. Naik V, Ahmed FU, Gupta A, Garg A, Sarkar C, Sharma B. Intracranial fungal granulomas: A single institutional clinicopathologic study of 66 patients and review of the literature. World Neurosurg. 2015. 83: 1166-72
10. Oliveira M, Severo CB, Guazzelli LS, Severo LC. Cryptococcus gattii fungemia: Report of a case with lung and brain lesions mimicking radiological features of malignancy. Rev Inst Med Trop Sao Paulo. 2007. 49: 263-5
11. Perfect JR, Dismukes WE, Dromer F, Goldman DL, Graybill JR, Hamill RJ. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2010. 50: 291-322
12. Popovich MJ, Arthur RH, Helmer E. CT of intracranial cryptococcosis. Am J Roentgenol. 1990. 154: 603-18
13. Ulett K, Cockburn J, Jefree R, Woods M. Cerebral cryptococcoma mimicking glioblastoma. BMJ Case Rep. 2017. 2017: 218824-
14. Yeh CH, Wang CS, Yeh TC, Lai CC. Central nervous system infection caused by Cryptococcus. Intern Med. 2013. 52: 2387-8