- Department of Neurosurgery, Hospital Universitario Marqués de Valdecilla, Santander, Cantabria, Spain.
DOI:10.25259/SNI_371_2020
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Jesus Manuel Esteban Garcia, David Mato Mañas, Enrique Marco De Lucas, Guillermo Garcia Catalan, Patricia Lopez Gomez, Carlos Santos Jimenez, Ruben Martin Laez. Invisible compression, anterior fossa tumor causing trigeminal neuralgia. 17-Mar-2021;12:106
How to cite this URL: Jesus Manuel Esteban Garcia, David Mato Mañas, Enrique Marco De Lucas, Guillermo Garcia Catalan, Patricia Lopez Gomez, Carlos Santos Jimenez, Ruben Martin Laez. Invisible compression, anterior fossa tumor causing trigeminal neuralgia. 17-Mar-2021;12:106. Available from: https://surgicalneurologyint.com/surgicalint-articles/10645/
Abstract
Background: Trigeminal neuralgia secondary to posterior and middle fossae tumors, whether ipsilateral or contralateral, has been well described. However, this disabling disease has never been reported in the context of anterior fossa neoplasms.
Case Description: A 75-year-old female with right hemifacial pain was diagnosed with an anterior clinoid meningioma. Despite neuroimaging did not show any apparent anatomical or neurovascular conflict, a detailed MRI analysis revealed a V3 hyperintensity. Not only symptoms completely resolved after surgical resection but also this radiological sign disappeared. Nowadays, the patient remains asymptomatic and V3 hyperintensity has not reappeared during her follow-up.
Conclusion: A surgical definitive treatment can be offered to patients suffering from trigeminal neuralgia secondary to lesions adjacent to Gasserian ganglion or trigeminal branches. In this respect, posterior and middle fossae tumors are well-reported etiologies. Nevertheless, in the absence of evident compression, other neoplasms located in the vicinity of these critical structures and considered as radiological findings may be involved in trigeminal pain. Microvascular and pressure gradient changes could be an underlying cause of these symptoms in anterior skull base lesions. Here, we report the case of a patient with uncontrollable hemifacial pain resolved after anterior clinoid meningioma removal.
Keywords: Anterior fossa, Meningioma, Trigeminal neuralgia
INTRODUCTION
Trigeminal neuralgia attributed to space-occupying lesions is a recognized entity within The International Classification of The Headache Disorders. The diagnosis is made when a structural compromise is evidenced on neuroimaging and when clinical findings led to tumor detection.[
Here, we present the case of a patient whose neuropathic symptoms resolved following an anterior clinoid meningioma resection. We discuss how certain remote lesions considered as radiological findings could be involved in trigeminal neuralgia pathophysiology in the absence of evident anatomical conflict.
CASE REPORT
A 75-year-old female with no significant medical history came to the emergency room complaining of an intense and paroxysmal electric shock-like sensation radiating to the right side and affecting the distribution of all three branches of trigeminal nerve, more in the distribution of V2. Physical examination did not reveal sensory loss and ruled out any other neurological deficit. Initial medical treatment with eslicarbazepine only achieved mild clinical improvement.
An initial cranial CT scan showed a right anterior clinoid lesion consistent with meningioma. The MRI confirmed the suspected diagnosis with no evidence of nerve, Gasserian ganglion, cavernous sinus, or vascular compression. However, an inflammatory thickening of V3 branch was demonstrated [
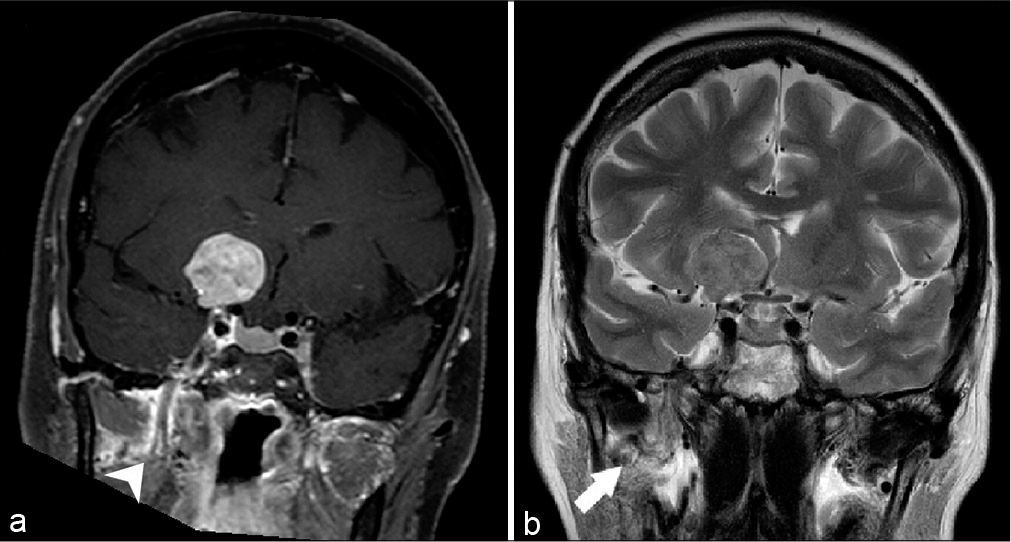
Figure 1:
(a) Coronal T1-weighted with fat saturation after gadolinium enhancement. An extra-axial mass with intense enhancement originating from the right anterior clinoid process is showed. No direct relation was observed the cavum de Meckel or right fifth nerve, mild enhancement around right V3 branch was depicted (arrow head). (b) Coronal T2-weighted image ruling out any relationship or shift between the critical structures. A high-intensity signal and thickening of V3 trigeminal branch was also evidenced (arrow).
A frontal craniotomy with gross total resection was performed (Simpson Grade II). Pathologist reported a WHO Grade I fibroblastic meningioma. Immediately following the procedure, her preoperative pain completely disappeared. After an uneventful postoperative period, our patient was discharged with no neurological deficit. Eslicarbazepine was discontinued during the following months; the patient did not experienced any facial pain relapse during this withdrawal. The MRI performed 1 month after the procedure showed the absence of previous V3 hyperintensity signal and a complete tumor resection [
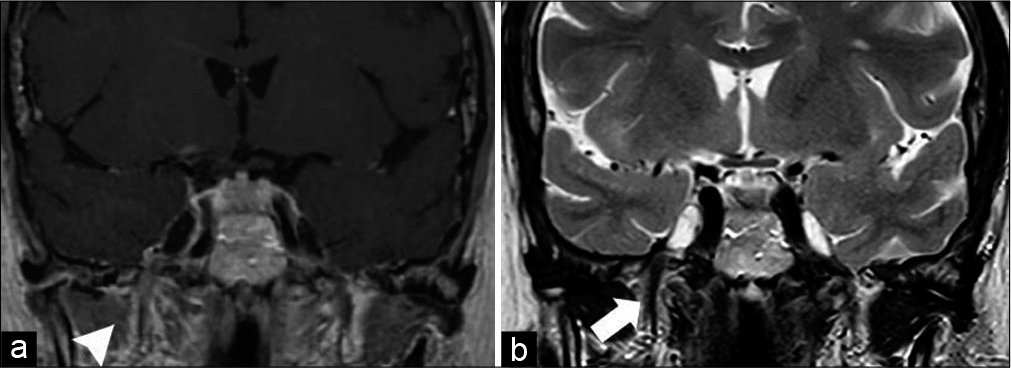
Figure 2:
(a) Coronal T1-weighted after gadolinium enhancement. Complete tumor resection and resolution of the mild enhancement around right V3 branch (arrow head). The same anatomical relationships are maintained in the middle fossa. (b) Coronal T2-weighted image showing resolution of the high-intensity signal of V3 trigeminal branch (arrow).
DISCUSSION
Initially, patients suffering from trigeminal neuralgia without neurological deficit are treated medically with anticonvulsants, muscle relaxants, and analgesics. Refractory cases should be studied and underlying diseases such as multiple sclerosis, ischemia, or other conditions that can require specific management should be discarded.[
Among neoplasms, posterior fossa lesions are the most common finding. The majority of them are benign such as schwannomas, epidermoid cysts, or meningiomas. Tumors may be located ipsilateral or contralateral to the affected nerve. The plausible pathophysiology of these two situations is supposed to be different. Lesions diagnosed on the same side of facial pain tend to cause symptoms by local compression, displacing vessels that could compromise the nerve or producing chemical irritation as in the case of epidermoid cysts.[
Regarding the case submitted, as far as we know, it is the first trigeminal neuralgia secondary to an anterior fossa tumor reported in the literature and constitutes a unique description. Although no neurovascular shift was identified on preoperative MRI, surgical treatment was effective. The most likely cause could be the displacement of microstructures, pressure gradients involvement or microvascular changes not visible by neuroimaging. This pathophysiology proposed here follows the same principles but in a reverse way as the case described by Berti et al. They reported a small meningioma with non-apparent compression and whose rapid volume reduction after radiosurgery triggered trigeminal neuralgia.[
Considering the distance, only one remote tumor without apparent mass effect on trigeminal structures and producing similar symptoms has been described in the literature. Gan and Choksey reported a case of a parafalcine parieto-occipital meningioma with complete sagittal sinus occlusion causing paroxysmal facial pain. The tumor was located around somatosensory cortex, an area that is supposed to be involved in perception and modulation of pain. Authors hypothesized a cortical theory of pain based on parietal location and on the fact that surgical removal succeeded as if a trigeminal decompression had been performed.[
One interesting point was the V3 radiological changes evidenced on MRI. This finding has never reappeared after the surgery. Although similar signal alterations have been described in cases of infiltrative and inflammatory diseases,[
CONCLUSION
Tumors are well-reported etiologies of trigeminal neuralgia. Although the most frequent mechanism is direct neural compression or brainstem shift as a result of a contralateral mass located in posterior fossa, patients with non-apparent compromise demonstrable on neuroimaging may benefit from surgical treatment. We have reported the unique case of a patient with anterior clinoid meningioma and ipsilateral trigeminal neuralgia resolved after tumor resection. Further studies must demonstrate the role of surgery in these clinical scenarios and the potential involvement of pressure gradients or microvascular changes in the vicinity of critical elements that could be implicated in the pathophysiology of facial pain.
Special attention should be paid to trigeminal branches on MRI. Some changes could be a radiological mark of these microstructural disturbances in the surroundings of gasserian ganglion and invisible to conventional neuroimaging.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Berti A, Granville M, Wu X, Huang D, Schwade JG. Delayed development of trigeminal neuralgia after radiosurgical treatment of a tentorial meningioma. Cureus. 2017. 9: e1628
2. Borges A, Casselman J. Imaging the trigeminal nerve. Eur J Radiol. 2010. 74: 323-40
3. Bullitt E, Tew JM, Boyd J. Intracranial tumors in patients with facial pain. J Neurosurg. 1986. 64: 865-71
4. Desai K, Nadkarni T, Bhayani R, Goel A. Cerebellopontine angle epidermoid tumor presenting with “Tic convulsif ” and tinnitus. sNeurol Med Chir (Tokyo). 2002. 42: 162-5
5. Florensa R, Llovet J, Pou A, Galitó E, Vilato J, Colet S. Contralateral trigeminal neuralgia as a false localizing sign in intracranial tumors. Neurosurgery. 1987. 20: 1-3
6. Fontaine D, Almairac F, Mondot L, Lanteri-Minet M. Cluster-like headache secondary to parasagittal. Headache. 2013. 53: 1496-8
7. Gan YC, Choksey MS. Parafalcine meningioma presenting with facial pain: Evidence for cortical theory of pain?. Br J Neurosurg. 2001. 15: 350-2
8. Harsha KJ, Kesavadas C, Chinchure S, Thomas B, Jagtap S. Imaging of vascular causes of trigeminal neuralgia. J Neuroradiol. 2012. 39: 281-9
9. .editors. Headache classification committee of the international headache society (IHS) the international classification of headache disorders. Cephalalgia. 2018. 38: 1-211
10. Ishi Y, Asaoka K, Sugiyama T, Yokoyama Y, Yamazaki K, Echizenya S. Case report: Trigeminal neuralgia caused by a minute meningioma with hyperostosed suprameatal tubercle. Case Rep Neurol. 2015. 7: 167-72
11. Jain N, Tadghare J, Patel A. An epidermoid cyst of the cerebellopontine angle presenting with the contralateral trigeminal neuralgia : An extremely rare case and the review of literature. World Neurosurg. 2018. 122: 220-3
12. Jannetta PJ. Vascular compression is the cause of trigeminal neuralgia. APS J. 1993. 2: 217-27
13. Leclercq D, Thiebaut J, Héran F. Trigeminal neuralgia. Diagn Interv Imaging. 2013. 94: 993-1001
14. Liu P, Liao C, Zhong W, Yang M, Li S, Zhang W. Symptomatic trigeminal neuralgia caused by cerebellopontine angle tumors. J Craniofac Surg. 2017. 28: e256-8
15. Mizobuchi Y, Shouno K, Nakajima K, Nagahiro S. Trigeminal neuralgia caused by fibrous bands: Case illustration. J Neurosurg. 2016. 2016: 186-7
16. Sepehrnia A, Schulte T. Trigeminal neuralgia caused by contralateral cerebellopontine angle meningioma-case report. Zentralbl Neurochir. 2001. 62: 62-4
17. Shulev Y, Trashin A, Gordienko K. Secondary trigeminal neuralgia in cerebellopontine angle tumors. Skull Base. 2011. 21: 287-94
18. Zhao H, Tang DZ, Zhu J, Zhang X, Tang YD, Li ST. Diffuse large B cell lymphoma presented as trigeminal neuralgia: 2 Cases reported and literature review. World Neurosurg. 2019. 123: 383-9