- Department of Neurosurgery Service, San Fernando Hospital, Buenos Aires, Argentina
- Plastic Surgery Service, Churruca-Visca Medical Complex, Buenos Aires, Argentina
- Neurosurgery Service, Padilla Hospital, Faculty of Medicine, National University of Tucumán, Tucuman, Argentina,
- Department of Educational and Scientific Institute of Neurosurgery, Peoples’ Friendship University of Russia, Moscow, Russian Federation
- Department of Neurosurgery, Sechenov First Moscow State Medical University (Sechenov University), Moscow, Russian Federation
- Department of Neurosurgery, Russian People’s Friendship University, Ulitsa Miklukho-Maklaya, Moscow, Russian Federation,
- Department of Neurosurgery, Azienda Ospedaliera Universitaria Pisana (AOUP), University of Pisa, Pisa, Italy,
- Neurosurgery Service, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado (ISSSTE), Regional Hospital October 1, Mexico City, Mexico,
- Medical Student, Autonomous University of Santo Domingo, Santo Domingo, Dominican Republic,
- 0Laboratory of Microsurgical Neuroanatomy, Second Chair of Gross Anatomy, School of Medicine, University of Buenos Aires, San Fernando, Argentina.
Correspondence Address:
Andreina Rosario Rosario, Medical Student, Autonomous University of Santo Domingo, Santo Domingo, Dominican Republic.
DOI:10.25259/SNI_783_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Mickaela Del Rosario Echavarría Demichelis1, Augusto Barrera2, Alvaro Campero3, Albert Sufianov4,5, Manuel De Jesus Encarnación Ramírez6, Renat Nurmukhametov6, Nicola Montemurro7, Carlos Castillo-Rangel8, Andreina Rosario Rosario9, Matias Baldoncini10. Lipotranferences in post neurosurgical esthetic defects. 22-Dec-2023;14:443
How to cite this URL: Mickaela Del Rosario Echavarría Demichelis1, Augusto Barrera2, Alvaro Campero3, Albert Sufianov4,5, Manuel De Jesus Encarnación Ramírez6, Renat Nurmukhametov6, Nicola Montemurro7, Carlos Castillo-Rangel8, Andreina Rosario Rosario9, Matias Baldoncini10. Lipotranferences in post neurosurgical esthetic defects. 22-Dec-2023;14:443. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=12677
Abstract
Background: There are numerous procedures in which, beyond adequate manipulation of the temporalis muscle and cranial closure, patients may present bone and muscle defects due to atrophy and consequent facial asymmetry, causing psychological discomfort and functional deterioration. The objective of our work is to combine the knowledge of plastic surgery and apply it to cranial reconstructions with fat transfers in post-neurosurgical patients, analyzing its results.
Methods: During the year 2022, 45 fat transfer procedures were performed for the correction of craniofacial defects, of which 29 were female and 16 were male. All had a surgical history of pterional craniotomies and their variants, orbitozygomatic and transzygomatic approaches, with the consequent volume deficit.
Results: The procedure was performed on an outpatient basis, with local anesthesia, and in an average time of 30–40 min. The lower hemiabdominal region was used as the donor area, processing the fat using the decantation technique and injecting it into the receptor area at the craniofacial level. The patients tolerated the procedure adequately without intraoperative complications or superadded events.
Conclusion: Fat transfer is a minimally invasive, effective, and cost-effective technique that plastic surgery offers us to implement in post-neurosurgical patients, as it achieves natural results that stand the test of time.
Keywords: Aesthetic cranial defect, Fat transfer, Post-neurosurgical reconstruction
INTRODUCTION
Neurosurgical interventions, indispensable in many clinical contexts, entail a detailed and meticulous process that often involves incisions in esthetically sensitive areas. These incisions, which frequently pass through the skin, subcutaneous tissue, periosteum, and bone in the frontotemporal region, pose challenges not only in terms of medical precision but also regarding postoperative aesthetic outcomes. Despite surgical expertise and the implementation of the most advanced techniques, some patients may inevitably experience sequelae such as muscular atrophy, facial asymmetry, and other visible defects. These changes, though subtle in some cases, can have a profound impact on self-esteem, emotional well-being, and the patient’s quality of life.[
Returning to harmonious craniofacial morphology is not merely an aesthetic desire. It has deep ramifications in the patient’s recovery, affecting their ability to resume everyday activities, interact in social settings, and ultimately reintegrate fully into society with confidence and comfort. This reintegration is vital for the patient’s mental and emotional health and can be as critical to their overall recovery as any other aspect of postoperative treatment.[
Faced with this challenge, the medical community has sought innovative solutions to address these aesthetic defects. One of the most promising answers is lipotransfer. This surgical technique, which involves the transplant of adipose tissue, has emerged as an effective means to repopulate areas with soft-tissue loss, thus restoring volume and improving the overall texture and appearance of the skin. Even more encouraging is that, in addition to its aesthetic efficacy, lipo transfer has proven to be safe, with a minimal risk profile. Recent studies corroborate these claims, highlighting patient satisfaction and positive outcomes in terms of aesthetic restoration and improved quality of life.[
However, although lipotransfer has shown promising results, it is essential to understand that this technique is only one piece of the puzzle in post-neurosurgical recovery. Every patient is unique, and their individual needs, coupled with the nature and location of the aesthetic defects, will dictate the most suitable approach. Moreover, the implementation of this treatment must be personalized, considering factors such as the patient’s age, medical history, and any potential contraindications that might arise.[
Another crucial consideration is the origin and quality of the adipose tissue used for the transfer. While most lipotransfers use tissue from the patient themselves, minimizing the risk of rejection, this tissue must be carefully selected, processed, and transplanted to ensure the graft’s viability and optimize the integration and survival of the transplanted tissue.[
As we move forward, we must continue to explore, evaluate, and refine the lipotransfer technique, along with other complementary approaches, to ensure we offer patients the best options available. This will not only guarantee superior esthetic outcomes but will also significantly contribute to the emotional and psychological well-being of those who have undergone neurosurgical procedures.
Our work aims to combine the knowledge of plastic surgery and apply it to cranial reconstructions in post-neurosurgical patients, analyzing the results.
MATERIALS AND METHODS
During 2022, lipo transfer procedures for craniofacial defect correction were jointly carried out by the Neurosurgery and Plastic Surgery departments of Hospital Petrona V. de Cordero, San Fernando, Province of Buenos Aires, and Hospital Padilla, San Miguel de Tucumán.
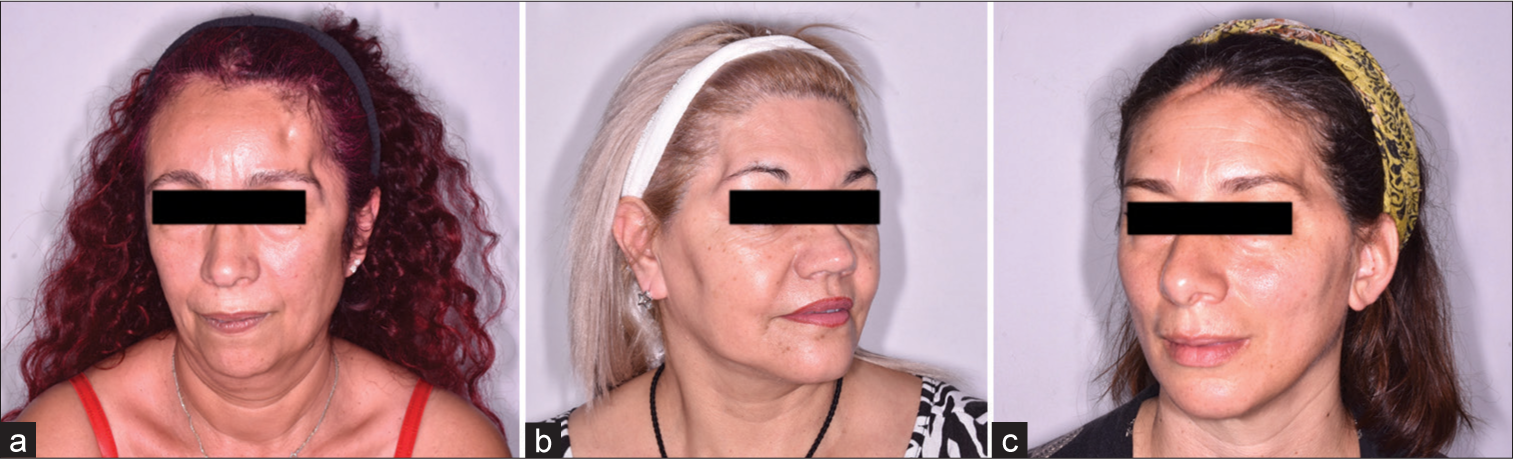
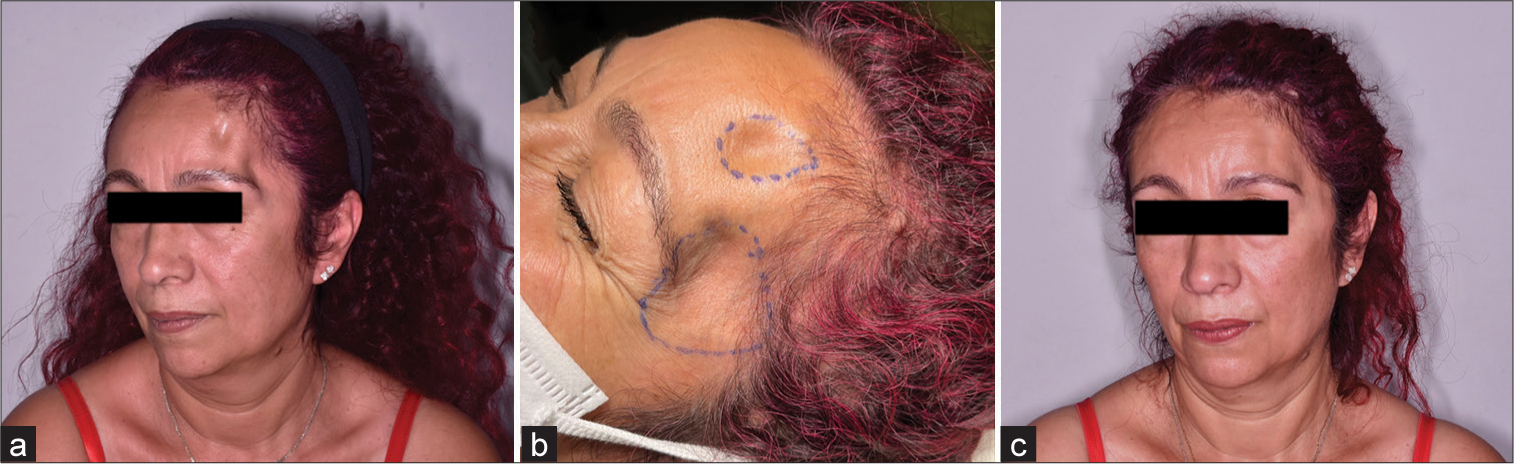
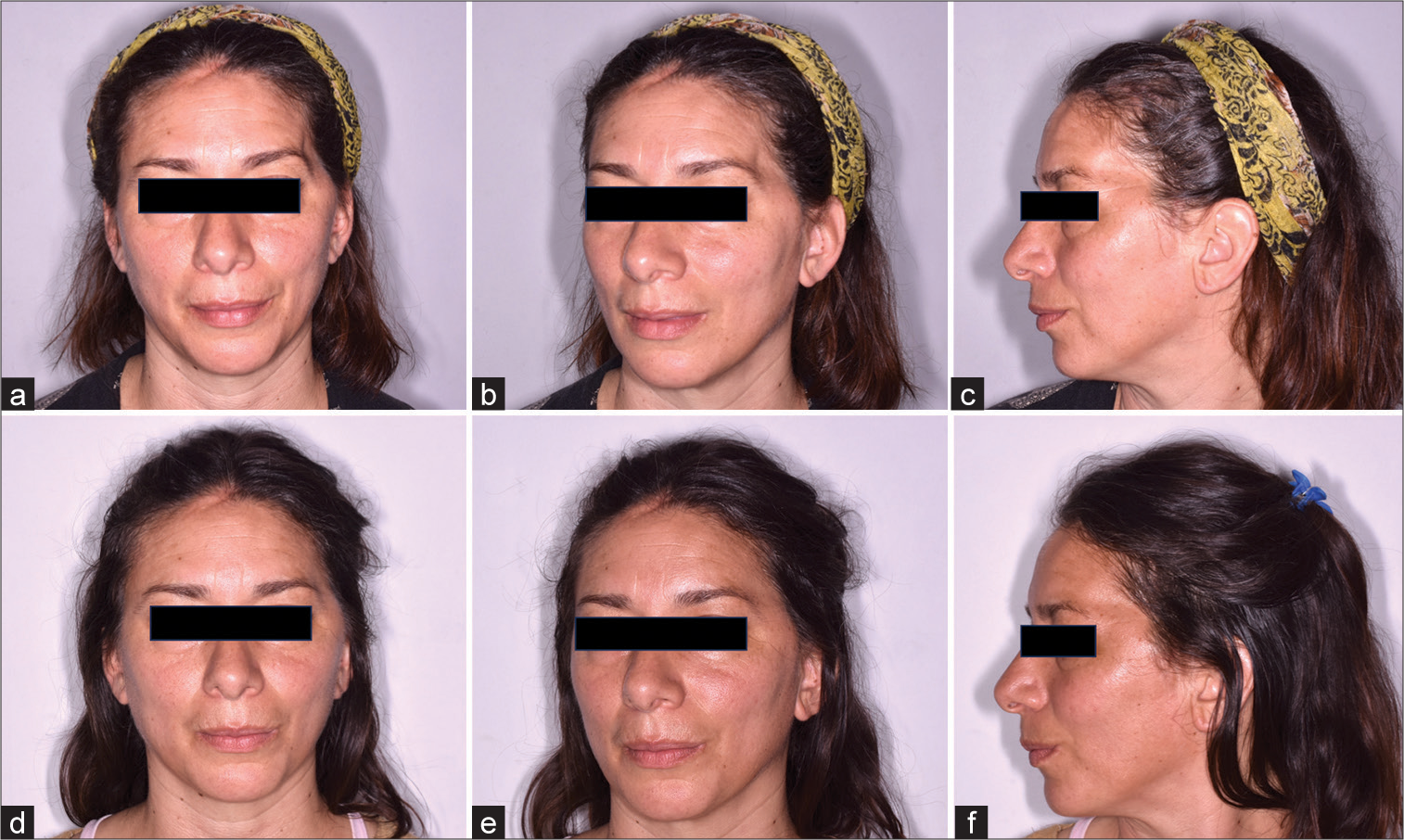
A series of 45 patients were selected: 29 females and 16 males. All had surgical histories of pterional craniotomies and their variants, orbitozygomatic and transcigomatic approaches, leading to a consequent volume deficit [
Included were patients older than 18 years, with at least six months since the craniofacial defect and the presence of bone or replacement material in the defect area. Those with active infections, coagulopathies, undergoing chemotherapy, or any other condition increasing their surgical risk were excluded from the study.
Instruments used were: Harvesting cannulas with 2.5 mm micro-perforations, single-hole 1.2 mm and 1.5 mm injection microcannulas, 1 mm and 2 mm Luer-to-Luer passers, 16 G and 14 G Abbocath, and Luer-Lock syringes of 1 mL, 3 mL, and 10 mL. For infiltration, Klein’s solution was used, composed of 2% lidocaine with epinephrine, ½ ampoule of adrenaline, and 500 mL of physiological solution.
RESULTS
The procedure was performed on an outpatient basis, under local anesthesia, with an average duration of 30–40 min.
Donor area
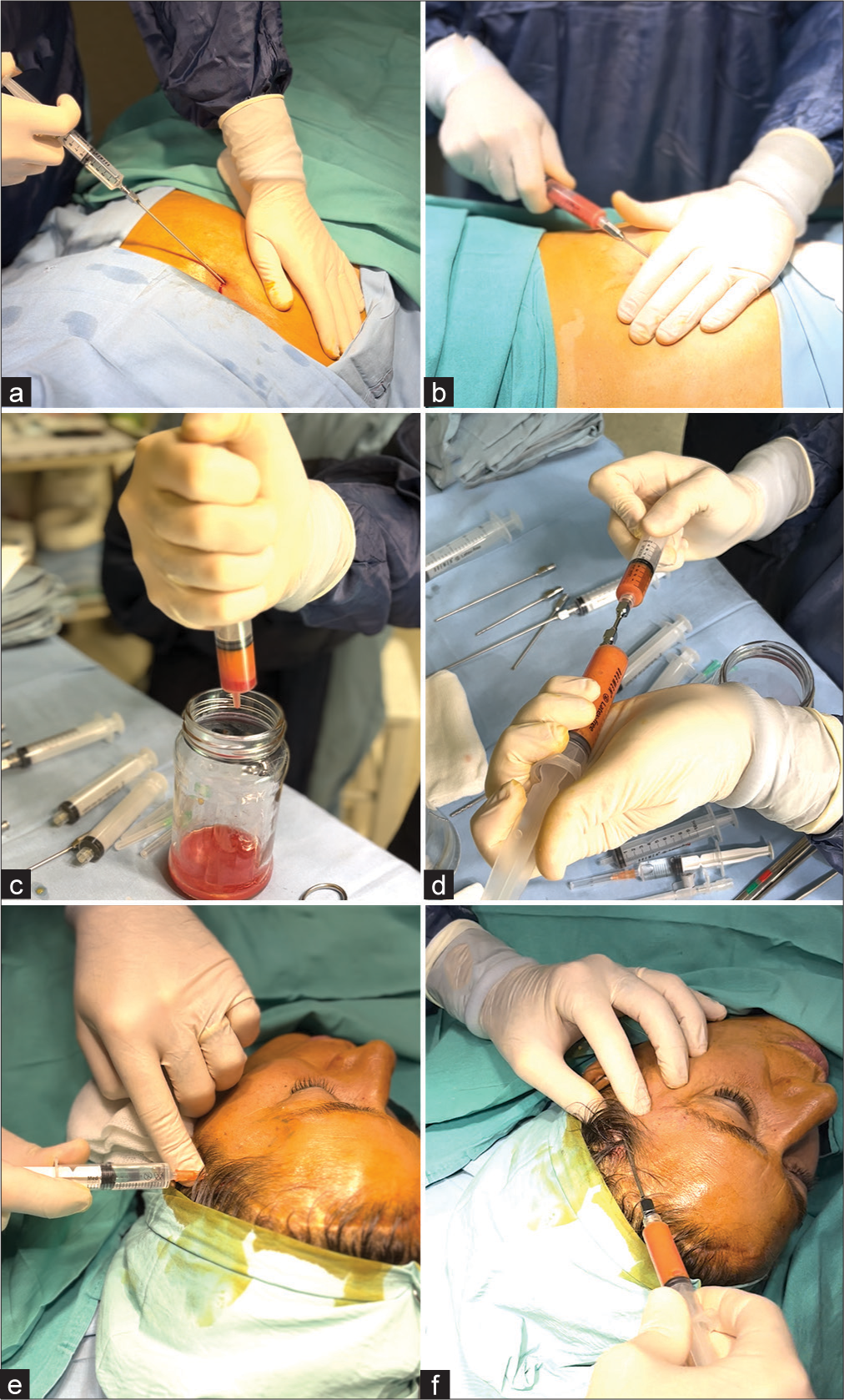
Patients were positioned supine, with prior asepsis and antisepsis of the skin in the lower hemiabdominal region. A 3 mm incision was made in the navel, followed by infiltration of the donor area using between 100 mL and 200 mL of Klein’s solution [
Figure 2:
Sequence of steps of the craniofacial lipotransfer technique. (a and b) Donor area. (a) Injection of Klein solution in the lower hemi abdominal region. (b) Manual liposuction with a syringe and scratching microcannula. (c and d) Fat processing. (c) Decantation of the fatty component and the Klein solution, disposal of the excess, and preservation of pure fat content. (d) Homogenization of the fatty content using a sieve. (e and f) Recipient area. (e) Infiltration with local lidocaine at the cranial defect level. (f) Fat infiltration with a blunt cannula in the form of retroinjection.
Processing
The fat was processed using the decantation method, placing the syringes vertically for 10–15 min until the fatty component and solution separated. The excess solution at the bottom of the syringe was discarded, retaining the pure fat [
Injection
Asepsis and antisepsis were done on the craniofacial recipient area, followed by local anesthesia infiltration (2% lidocaine), avoiding excess volume that might alter the outcome perception [
Given the frequent presence of scars in the craniofacial defect area, rigotomies were performed using the Abocath tip to release adhesions and achieve a more homogeneous result. It is crucial to avoid depositing fat in boluses or creating pockets, as this predisposes to increased reabsorption of the grafted tissue.
Postoperative care
Treated areas were covered with dressing and surgical tape, and slight compression was applied to reduce edema and prevent patient contact. Patients were advised against any manipulation or massaging of the recipient area during the first two weeks due to the risk of fat migration or necrosis. Cold compresses were recommended for the first 24 h postoperatively to reduce inflammation and discomfort, along with anti-inflammatory therapy.
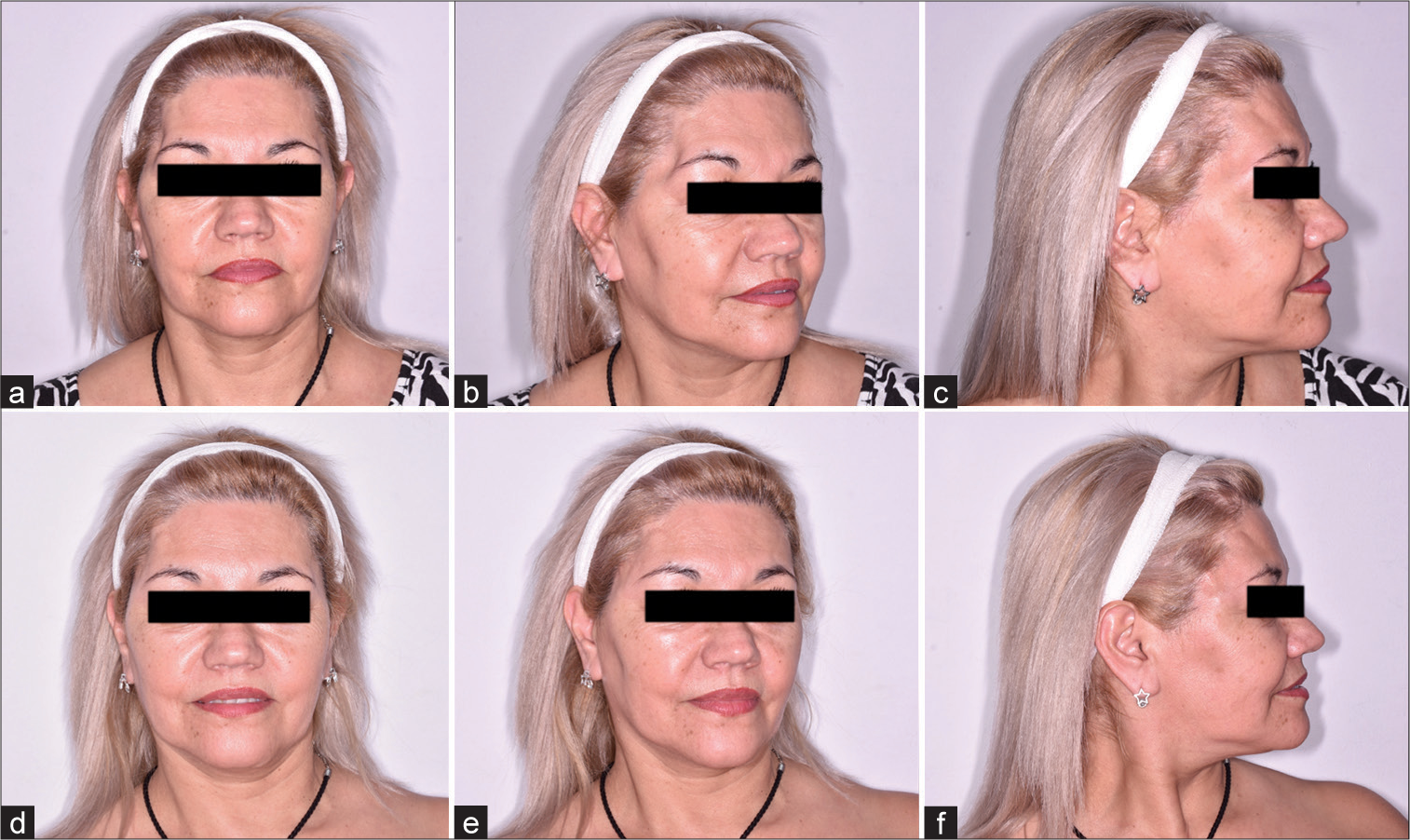
Patients tolerated the procedure well, with no intraoperative complications or additional events [
DISCUSSION
Autologous fat transfer (AFT) is a well-established, accepted, and safe surgical technique that involves injecting autologous adipose tissue into a defective site directly after its collection and processing.
As Clauser et al. mention, it was initially used as an excellent filler for facial enhancement and rejuvenation. Still, it has evolved over the years to become a more complex reconstructive procedures. AFT is now also used to correct and restore cranial and maxillofacial volume defects resulting from traumas, tumors, or congenital disorders.[
The origins of this technique date back to 1893, when Neuber first described the autologous fat grafting technique for soft-tissue augmentation.[
Several authors support the favorable outcomes of this technique. As Gornitsky mentions, fat serves as an ideal soft tissue filler because it is autologous, biocompatible, easily accessible in most patients, relatively permanent, and can integrate into the surrounding tissue at the injection site.[
An interesting contribution from the literature pertains to the role of mesenchymal cells from adipose tissue in the efficiency and functionality of the lipotransfer technique. In addition to being a natural filler, human adipose tissue represents a rich source of mesenchymal stem cells, called adipose-derived stem cells (ADSCs), which exhibit multi-lineage differentiation potential and secrete various angiogenic and anti-apoptotic factors.[
Mesenchymal stem cells represent a great tool in regenerative medicine due to their ability to differentiate into a variety of specialized cells. Among the adult tissues where mesenchymal stem cells reside, adipose tissue seems to be a particularly good source because it contains more multipotent cells per milliliter than bone marrow. Other advantages of adipose tissue (ease of surgical access, availability, and isolation) compared to other sources have made it the ideal large-scale source for clinical and research applications.[
ADSCs possess a unique regenerative potential: they self-renew, secrete a favorable cytokine pattern (angiogenic, immunosuppressive, anti-inflammatory, and antioxidant), and can differentiate along various tissue lineages (adipocytes, osteoblasts, myocytes, chondrocytes, endothelial cells, and cardiomyocytes).[
A group of authors conducted a prospective cohort study to assess the safety and efficacy of fat grafting in the treatment of posttraumatic and postsurgical craniofacial deformities.[
Egro et al. advocate that autologous fat grafting for craniofacial deformities is an excellent alternative, being less invasive and safer than traditional reconstructive options. They noted that the volume stabilization rate is achieved at around three months, leading to positive outcomes as reported by patients.[
The degree of fat resorption and fat uptake depends on the facial area injected. It tends to be high in the labial-perioral area, moderate in the temples, average in the glabella, and medium-low in the malar area and marionette lines. Proper centrifugation enhances the retention of the graft and maximizes regeneration by purifying and condensing the aspirated adipose tissue. Moreover, for better regeneration of adipocytes after lipotransfer and the revitalizing effects on tissues, fat condensation is paramount.[
Although most reported cases process fat through centrifugation, it is worth noting that it is not the only processing method. Filtration or decantation can also be used, as demonstrated in some cases. At present, there is no evidence favoring one technique over another. As Clauser et al. mention, the longevity of the grafted fat remains unknown. Supplementary treatments might be needed to achieve more satisfactory results. The outcome largely depends on the surgeon’s experience and technique. Over time, body weight fluctuations can also impact graft volume. When performing a fat graft, it is crucial to avoid overcorrection or over-grafting and the so-called strangulation effect.[
Gornitsky et al. conducted the first systematic review of fat grafting in the facial region. They evaluated a total of 4577 patients with various facial contour defects treated with autologous fat grafting. Injection sites were categorized by anatomical facial regions: upper face (32.5%), middle (53%), and lower (14.5%). The average volume of injected fat was 16.9 mL. The weighted average volume retention of non-enriched grafts was 41.63%. Graft retention for esthetic and reconstructive facial indications was 45.76% and 38.26%, respectively. They recorded a minimal complication rate of 2.27%. The majority of these complications (84.61%) were due to skin asymmetry or irregularities (excessive or inadequate grafting or graft survival). Fat necrosis (n = 3) and infection (n = 2) accounted for 2.88% and 1.92%, respectively.[
Most of the complications reported in the literature are minor and include reabsorption, under-correction, over-correction, visible irregularities, migration of injected fat, and donor site complications. Reabsorption should be expected to some extent, but studies have shown significant variability. Irregularities or under-correction can be addressed with several rounds of fat grafting. Major complications are much rarer.[
Schiraldi et al. categorize the side effects related to lipotransfer into three categories: Severe, moderate, and minor. Severe side effects (13.4%) are intravascular injection or migration, which often result in permanent disability or death. Moderate side effects (38.3%), such as fat hypertrophy, necrosis, cyst formation, irregularities, and asymmetries, require a touch-up operation. [
Although there is a lack of prospective studies to further determine the actual complication rate of this procedure, with a clear report on complication guidelines, as mentioned by Schiraldi et al., based on the literature, lipotransfer has a high patient satisfaction rate, with few complications and minimal morbidity at the donor site.[
In the study by Ganau et al., a 20-year audit on Cranial Prostheses (CP) materials at their institution revealed that porous hydroxyapatite (PHA) had better outcomes compared to polymethylmethacrylate.[
The FACE-Q craniofacial module is a patient-reported outcome measure (PROM) designed to assess the experiences and outcomes of individuals undergoing craniofacial procedures. It comprises a set of validated questionnaires that gauge aspects such as satisfaction with facial appearance, health-related quality of life, and the psychosocial impact.[
Utilizing the FACE-Q craniofacial module post lipotransference in craniofacial defect cases following neurosurgery offers a comprehensive, patient-centric approach to evaluating and improving the care pathway, fostering a better understanding of the patient experience and ultimately contributing to the advancement of craniofacial reconstructive techniques.
CONCLUSION
Based on the results obtained from our patients and the analysis of the literature, we can confidently state that lipotransfer is a minimally invasive, effective, and cost-efficient technique with low complication rates and high patient satisfaction rates. It is an excellent tool provided by plastic surgery to be implemented in post-neurosurgical patients as it achieves natural results that stand the test of time. The future of lipotransfer in neurosurgery is luminous. As the two fields converge, there will likely be a plethora of innovations that harness the best of both worlds, ensuring optimal outcomes for patients in terms of both neurological function and esthetic restoration. This collaboration heralds a new era in medicine where holistic patient care is not just an ideal but a standard.
Ethical approval
The author(s) declare that they have taken the ethical approval from IEC (Dec/1/2021).
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Agaverdiev M, Shamsov B, Mirzoev S, Vardikyan A, Ramirez ME, Nurmukhametov R. MiRNA regulated therapeutic potential of the stromal vascular fraction: Current clinical applications-A systematic review. Noncoding RNA Res. 2023. 8: 146-54
2. Bourne DA, Bliley J, James I, Donnenberg AD, Donnenberg VS, Branstetter BF. Changing the paradigm of craniofacial reconstruction: A prospective clinical trial of autologous fat transfer for craniofacial deformities. Ann Surg. 2021. 273: 1004-11
3. Clauser L, Lucchi A, Tocco-Tussardi I, Gardin C, Zavan B. Autologous fat transfer for facial augmentation and regeneration: Role of mesenchymal stem cells. Atlas Oral Maxillofac Surg Clin North Am. 2018. 26: 25-32
4. Clauser L, Zavan B, Galiè M, Di Vittorio L, Gardin C, Bianchi AE. Autologous fat transfer for facial augmentation: Surgery and regeneration. J Craniofac Surg. 2019. 30: 682-5
5. Coleman SR. Long-term survival of fat transplants: Controlled demonstrations. Aesthet Plast Surg. 1995. 19: 421-5
6. Coleman SR. The technique of periorbital lipoinfiltration. Oper Tech Plast Reconstr Surg. 1994. 1: 20-6
7. Egro FM, Coleman SR, Rubin JP. Fat grafting for treatment of secondary facial deformity. Clin Plast Surg. 2020. 47: 147-54
8. Ersek RA. Transplantation of purified autologous fat: A 3-year follow-up is disappointing. Plast Reconstr Surg. 1991. 87: 219-27 discussion 228
9. Ganau M, Calisto A, Thomas GP. Considerations regarding FACE-Q® craniofacial as a newly developed QoL-PROMs questionnaire suitable for paediatric and adult cranioplasty patients. Neurosurg Rev. 2023. 46: 141
10. Ganau M, Cebula H, Fricia M, Zaed I, Todeschi J, Scibilia A. Surgical preference regarding different materials for custom-made allograft cranioplasty in patients with calvarial defects: Results from an internal audit covering the last 20 years. J Clin Neurosci. 2020. 74: 98-103
11. Gornitsky J, Viezel-Mathieu A, Alnaif N, Azzi AJ, Gilardino MS. A systematic review of the effectiveness and complications of fat grafting in the facial region. JPRAS Open. 2018. 19: 87-97
12. Khorasani M, Janbaz P. Clinical evaluation of autologous fat graft for facial deformity: A case series study. J Korean Assoc Oral Maxillofac Surg. 2021. 47: 286-90
13. Krastev TK, Beugels J, Hommes J, Piatkowski A, Mathijssen I, van der Hulst R. Efficacy and safety of autologous fat transfer in facial reconstructive surgery: A systematic review and meta-analysis. JAMA Facial Plast Surg. 2018. 20: 351-60
14. Khouri RK, Eisenmann-Klein M, Cardoso E, Cooley BC, Kacher D, Gombos E. Brava and autologous fat transfer is a safe and effective breast augmentation alternative: Results of a 6-year, 81-patient, prospective multicenter study. Plast Reconstr Surg. 2012. 129: 1173-87
15. Klassen AF, Rae C, Wong Riff KW, Bulstrode N, Denadai R, Goldstein J. FACE-Q Craniofacial module: Part 1 validation of CLEFT-Q scales for use in children and young adults with facial conditions. J Plast Reconstr Aesthet Surg. 2021. 74: 2319-29
16. Neuber F. Fat transplantation report on the negotiations of the Deutscht Gesellschaft Chir. Zentralbl Chir. 1893. 22: 66
17. Obagi S, Willis C. Autologous fat augmentation of the face. Atlas Oral Maxillofac Surg Clin North Am. 2018. 26: 41-50
18. Schiraldi L, Sapino G, Meuli J, Maruccia M, Cherubino M, Raffoul W. Facial Fat Grafting (FFG): worth the risk? A systematic review of complications and critical appraisal. J Clin Med. 2022. 11: 4708
19. Toyserkani NM, Quaade ML, Sørensen JA. Cell-assisted lipotransfer: A systematic review of its efficacy. Aesthetic Plast Surg. 2016. 40: 309-18