- Department of Neurosurgery, All India Institute of Medical Sciences, Patna, Bihar, India.
Correspondence Address:
Vikas Chandra Jha, Department of Neurosurgery, All India Institute of Medical Sciences, Patna, Bihar, India.
DOI:10.25259/SNI_942_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Vikas Chandra Jha, Mohammad Shahnawaz Alam, Vivek Sharan Sinha, Rahul Jain. Management dilemma in a rare case of pituitary apoplexy with akinetic mutism in the setting of ruptured junctional brain aneurysm: A case report and literature review. 06-Jan-2023;14:4
How to cite this URL: Vikas Chandra Jha, Mohammad Shahnawaz Alam, Vivek Sharan Sinha, Rahul Jain. Management dilemma in a rare case of pituitary apoplexy with akinetic mutism in the setting of ruptured junctional brain aneurysm: A case report and literature review. 06-Jan-2023;14:4. Available from: https://surgicalneurologyint.com/surgicalint-articles/12095/
Abstract
Backgound: Pituitary apoplexy is associated with stroke, head injury, and brain tumors. Still, its presentation due to the ruptured aneurysm is rare and its presentation with akinetic mutism has not been reported.
Case Description: The patient in the present study is 21-year-old female who presented in our emergency department in an altered sensorium with Glasgow comma score (GCS) E2V1M1. She was intubated and resuscitated. Routine blood investigations, lipid profile, and hormonal studies were normal. Initial noncontrast computed tomography (NCCT) head revealed subarachnoid hemorrhage in the interhemispheric fissure and evidence of bleeding in the pituitary gland. Magnetic resonance imaging (MRI) brain was soon done, which showed an infarct and hemorrhage in the pituitary gland; there was an evidence of an infarct in the bilateral medial frontal gyrus, basal ganglia, and supplementary motor area. MR arteriography revealed an aneurysm at the left A1-anterior communicating artery (Acom) junction directed superomedially with diffuse spasm in a bilateral anterior cerebral artery. Pterional craniotomy was done with clipping of the aneurysm and evacuation of blood clots from the interhemispheric fissure and pituitary gland. Histopathology features suggestive of the non-functioning pituitary tumor with interspersed hemorrhagic necrosis. Intraarterial vasodilation with microcatheter injection was given, but vasospasm did not improve. Postoperatively, Levodopa was started. She used to track objects in front of her eye and started nodding her head in “yes and no fashion,” with power in limbs improved to 3/5 at 6 months of follow-up.
Conclusion: Pituitary apoplexy with ruptured A1-Acom junction aneurysm with nonfunctioning pituitary macroadenoma is rare, and its presentation with akinetic mutism has not been reported. As there is scarce literature suggesting an association between pituitary apoplexy and ruptured aneurysm, it is challenging to comment regarding its pathogenesis. Although akinetic mutism generally has a poor prognosis, it may respond to Levodopa with a better outcome.
Keywords: Akinetic mutism, Pituitary apoplexy, Ruptured brain aneurysm, Ruptured intracranial Aneurysm
INTRODUCTION
Pituitary apoplexy is defined as a hemorrhage or infarct in the pituitary gland. Pituitary apoplexy due to a ruptured anterior circulation aneurysm is uncommon and rare due to an A1 segment of the anterior cerebral artery (ACA) aneurysm.[
PATIENT DETAILS
A 20-year-old female patient presented with altered sensorium, and quadriparesis, unable to speak for 7 days. Cranial nerve function, including 2nd, 3rd, 4th, and 6th, could not be assessed. Power in bilateral upper and lower limbs was 1/5. Blood investigations were normal, including hemoglobin (HB), total leucocyte count, differential leucocyte count (TLC, DLC), electrolytes, liver function test, and coagulation profile. Preoperative hormonal assays were normal and viral markers were negative. On radiological evaluation, the initial NCCT head revealed a subarachnoid hemorrhage in the interhemispheric fissure and evidence of bleeding in the pituitary gland [
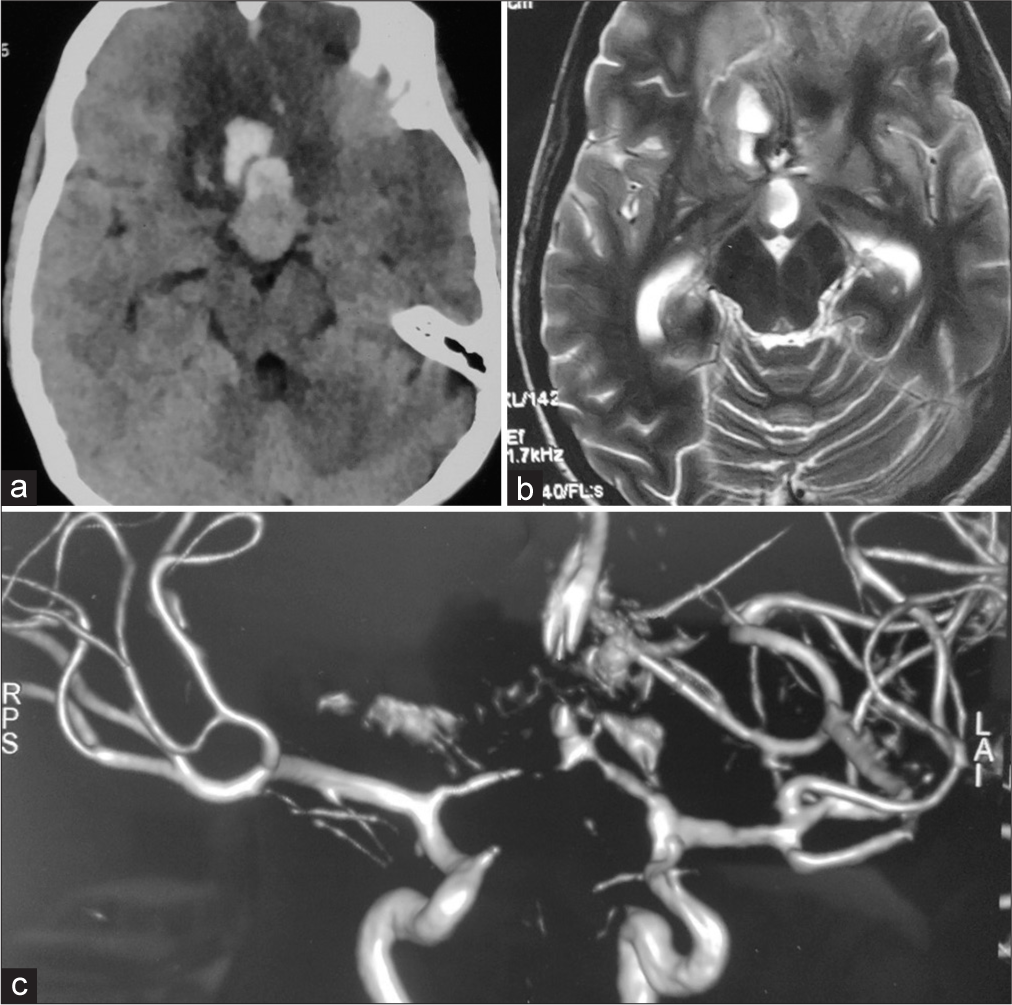
Figure 1:
(a) Non-contrast computed topography head suggests hemorrhage in the pituitary gland and interhemispheric fissure with hypodensities in the bilateral medial frontal lobe suggesting infarct; (b) T2 axial MRI suggests a hyperintense lesion in the isointense pituitary gland and hyperintensity in the medial frontal lobe with bilateral loss of sulci and gyri pattern suggestive of the infarct; (c) MR arteriography suggests an aneurysm at the left A1-Acom junction with no visualization of proximal and distal anterior cerebral artery suggestive of diffuse spasm. RPS: Right posterosuperior; LAI: Left Anteroinferior.
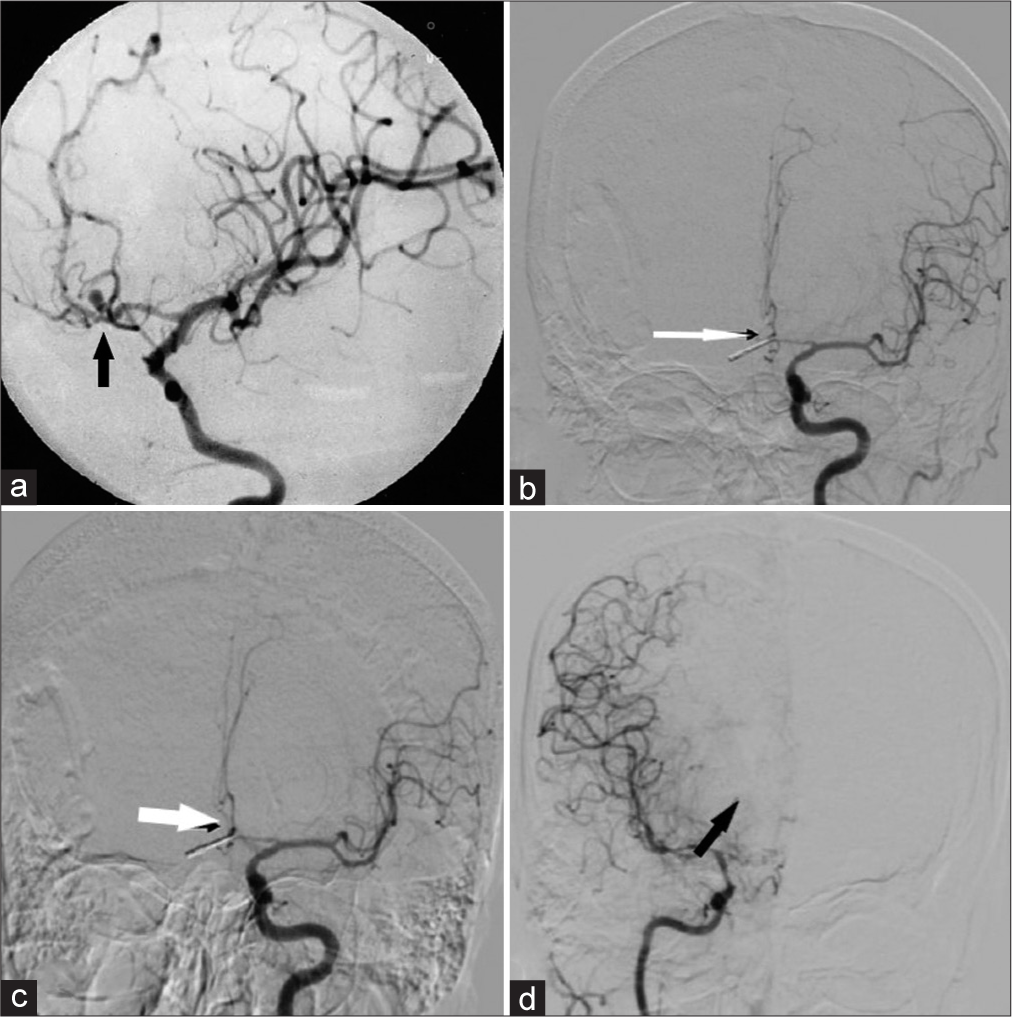
Figure 2:
(a) Preoperative digital subtraction angiography (DSA) showing anterior communicating artery (Acom) aneurysm at the junction of the Acom and left A2 with distal vessels narrow in caliber, suggesting vasospasm (black arrow); (b) postoperative DSA showing clipped aneurysm with diffuse vasospasm of bilateral A2, Left A1, and middle cerebral artery vessels (white arrow); (c) postoperative DSA following vasodilation by intraarterial microcatheter injection of nimodipine revealing persistent vasospasm (white arrow); (d) postoperative DSA of the right internal carotid artery suggestive of persistent vasospasm more in anterior cerebral artery branches than right middle cerebral artery (black arrow).
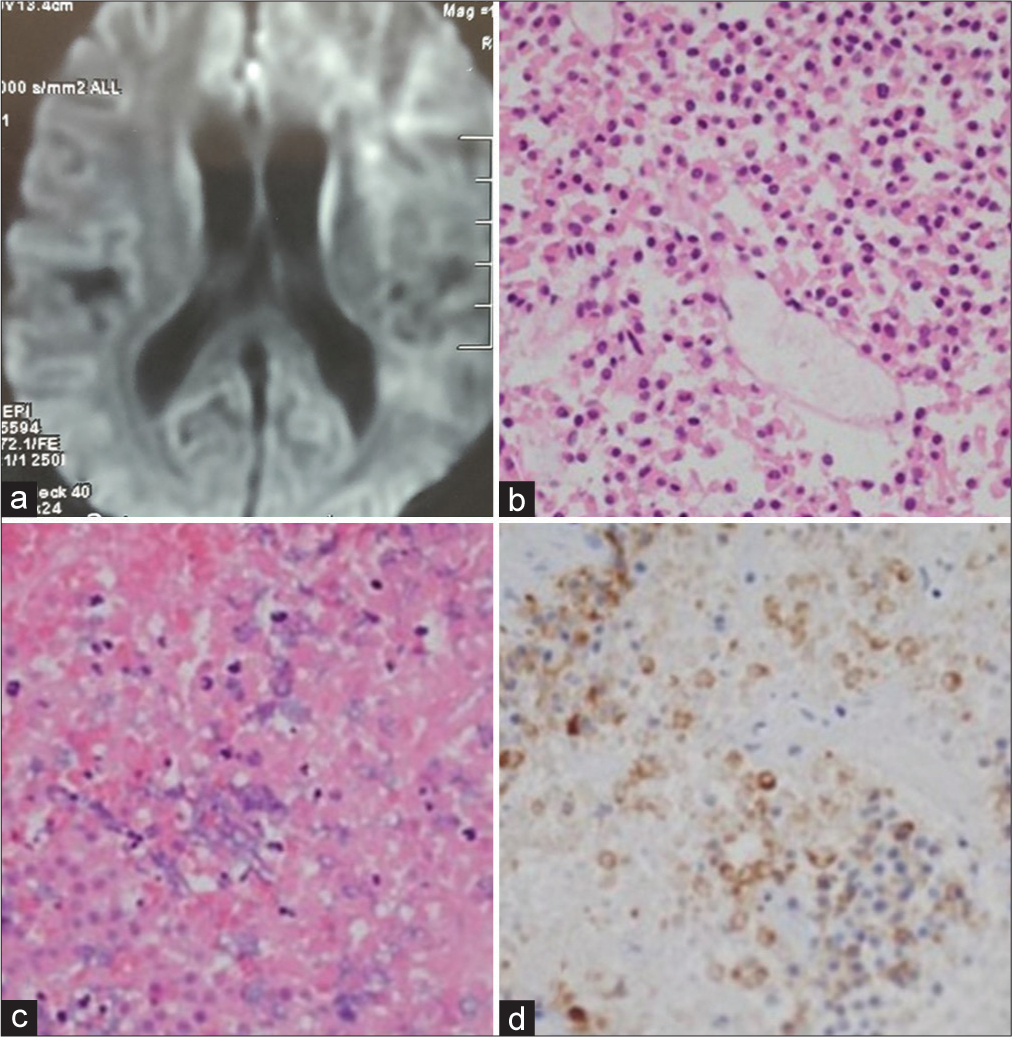
The postoperative MRI brain revealed a bilateral diffuse infarct [
Figure 3:
(a) Postoperative MRI brain with diffusion-weighted sequence suggests restriction diffusion in the bilateral frontal lobe, and basal ganglia region suggestive of the infarct; (b) microscopic sections show a hypercellular tumor composed of solid sheets, nests, and trabeculae. Tumor cells contain scant granular eosinophilic cytoplasm, round nuclei inconspicuous nucleoli; (c) Hematoxylin-Eosin staining of the pituitary macroadenoma revealing hemorrhagic necrosis pattern and extensive tumor coagulative necrosis, consistent with recent hemorrhagic infarction; (d) immunohistochemical results for pituitary hormones show negative staining for growth hormone, leutinising hormone, thyroid stimulating hormone, adreno corticotrophic hormone (GH, LH, TSH, and ACTH); Prolactin immunostains suggest nonfunctional pituitary macroadenoma.
DISCUSSION
The intracranial aneurysm has been reported earlier, with a higher prevalence in pituitary adenoma than in the general population.[
As reported earlier by a few authors, the secretary nature of the tumor may induce weakness in the wall of the vessels close to it.[
In the present case, the tumor was neither functional nor had parasellar extension to support the hypothesis proposed in the earlier studies that pituitary macroadenoma could have influenced the formation of the aneurysm by manipulating the course of the vessels or have influenced the rupture by secretary nature of the tumor. In this case, the aneurysm was directed superomedially at the junction of the left A1 and Acom. The lower lobe of the aneurysm had ruptured, leading to hemorrhage in the pituitary tumor and bleeding in the interhemispheric fissure. Such findings in the patient had been reported by Yoshida et al. in non-functioning pituitary macroadenoma and Laidlaw et al. in growth hormone-secreting tumors. Still, none of them have features suggestive of akinetic mutism.[
The patient in the present study was 21-years-old female; the youngest patient reported pituitary apoplexy due to a ruptured aneurysm. Earlier, Song et al. reported a patient 31 years of age with a left Pcom aneurysm with unknown pituitary secretary function, and clinically, the patient had headaches, vomiting, and visual blurring.[
Preoperative severe vasospasm in the distribution of bilateral distal ACA and MCA territory may have led to infarct in the region of bilateral supplementary motor area, thalamus, hypothalamus, and basal ganglia leading to akinetic mutism in this patient. Akinetic mutism is generally observed following head injury, stroke, and brain tumors. Still, its presence with a ruptured A1-Acom junctional aneurysm is rare, and its association with pituitary apoplexy probably had not been reported to the best of my knowledge.
In a literature review by Choudhari et al., 21 cases have been reported with akinetic mutism following a ruptured aneurysm with subarachnoid hemorrhage. In the majority of the patients, they have infarct in distal ACA territory with poor prognosis.[
CONCLUSION
It is difficult to comment on the exact etiology of the prevalence of aneurysm with pituitary macroadenoma, especially the growth hormone-secreting prolactinoma, as it can also present in nonfunctioning pituitary macroadenoma. Presentation with akinetic mutism with infarct in the distal territory is rare, with pituitary apoplexy with ruptured A1-Acom junctional aneurysm. Improvement following levodopa supplementation has not been encouraging in distal ACA territory infarct with akinetic mutism. Still, its role in improvement cannot be discarded as found in the present case so that it can be given in such a scenario.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Arnts H, van Erp WS, Lavrijsen JC, van Gaal S, Groenewegen HJ, van den Munckhof P. On the pathophysiology and treatment of akinetic mutism. Neurosci Biobehav Rev. 2020. 112: 270-8
2. Choudhari KA. Subarachnoid hemorrhage and akinetic mutism. Br J Neurosurg. 2004. 18: 253-8
3. Combarros O, Infante J, Berciano J. Akinetic mutism from frontal lobe damage responding to Levodopa. J Neurol. 2000. 247: 568-9
4. Laidlaw JD, Tress B, Gonzales MF, Wray AC, Ng WH, O’Brien JM. Coexistence of aneurysmal subarachnoid hemorrhage and pituitary apoplexy: Case report and review of the literature. J Clin Neurosci. 2003. 10: 478-82
5. Ogunyemi AO. Akinetic mutism caused by subarachnoid hemorrhage. A case report. Central Afr J Med. 1984. 30: 273-7
6. Oshino S, Nishino A, Suzuki T, Arita H, Tateishi A, Matsumoto K. Prevalence of cerebral aneurysm in patients with acromegaly. Pituitary. 2013. 16: 195-201
7. Shahlaie K, Olaya JE, Hartman J, Watson JC. Pituitary apoplexy is associated with anterior communicating artery aneurysm and aberrant blood supply. J Clin Neurosci. 2006. 13: 1057-62
8. Sibille FX, Hantson P, Duprez T, van Pesch V, Giglioli S. Reversible akinetic mutism after aneurysmal subarachnoid haemorrhage in the territory of the anterior cerebral artery without permanent ischaemic damage to anterior cingulate gyri. Case Rep Neurol Med. 2016. 2016: 5193825
9. Song RX, Wang DK, Wang Z, Wang ZW, Wang SX, Wei GX. Coexistence of aneurysmal subarachnoid hemorrhage and surgically identified pituitary apoplexy: A case report and review of the literature. J Med Case Rep. 2014. 8: 166
10. Weir B. Pituitary tumors and aneurysms: Case report and review of the literature. Neurosurgery. 1992. 30: 585-91
11. Xu K, Yuan Y, Zhou J, Yu J. Pituitary adenoma apoplexy caused by rupture of an anterior communicating artery aneurysm: Case report and literature review. World J Surg Oncol. 2015. 13: 228
12. Yoshida M, Hiu T, Baba S, Morikawa M, Horie N, Ujifuku K. Ruptured aneurysm-induced pituitary apoplexy: Illustrative case. J Neurosurg Case Lessons. 2021. 1: CASE21169






Marvin Salgado Pérez
Posted January 20, 2023, 10:12 pm
He visto pocos casos de apoplejía pituitaria con resultados fatales a veces