- Department of Neurosurgery, School of Medicine, State University of New York at Stony Brook, NY and c/o Dr. Marc Agulnick, 1122 Frankllin Avenue Suite 106, Garden City, New York, United States.
- The Lancione Law Firm, Rocky River, Ohio, United States.
Correspondence Address:
Nancy E Epstein, MD, Department of Neurosurgery, School of Medicine, State University of New York at Stony Brook, NY, and c/o Dr. Marc Agulnick, 1122 Franklin Avenue Suite 106, Garden City, NY 11530 New York, United States.
DOI:10.25259/SNI_875_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nancy E Epstein1, John Lancione Esq2. Medicolegal corner (spine): Contraindicated use of DuraSeal in anterior cervical spine led to quadriplegia. 19-Oct-2021;12:532
How to cite this URL: Nancy E Epstein1, John Lancione Esq2. Medicolegal corner (spine): Contraindicated use of DuraSeal in anterior cervical spine led to quadriplegia. 19-Oct-2021;12:532. Available from: https://surgicalneurologyint.com/surgicalint-articles/11178/
Abstract
Background: The package insert for DuraSeal (Integra LifeSciences, Princeton NJ) states it is Contraindicated for use in the anterior cervical spine (confined space): “Do not apply DuraSeal® hydrogel to confined bony structures where nerves are present since neural compression may result due to hydrogel swelling (…up to 12% of its size in any direction).” Further, it should not be used to treat massive unrepaired cerebrospinal fluid (CSF) leaks in any location; “…(it) is indicated as an adjunct to sutured dural repair during spine surgery to provide watertight closure,” but it is not to be used “...for a gap greater than 2 mm….”
Methods: A spinal surgeon interpreted a geriatric patient’s MR as showing severe C3-C4 to C5-C6 anterior cord compression due to disc disease/spondylosis. However, he never reviewed the CT report/images that documented marked ossification of the posterior longitudinal ligament (OPLL) with multiple signs of dural penetrance.
Results: The anterior C4, C5 corpectomy, and C3-C6 strut fusion/plating resulted in a massive, irreparable cerebrospinal fluid (CSF) leak. Despite the contraindications, the surgeon mistakenly applied DuraSeal which caused the patient’s postoperative quadriplegia (i.e., as documented on the delayed postoperative MR scan). Following a secondary surgery consisting of a laminectomy/posterior fusion, the patient was still quadriplegic. Further, as he requested no postoperative MR scan and performed no subsequent corrective surgery (i.e., anterior removal of DuraSeal), the patient remained permanently quadriplegic.
Conclusion: DuraSeal is directly contraindicated for use in the anterior cervical spine, with/without a CSF leak. Here, utilizing DuraSeal for anterior cervical OPLL surgery resulted in permanent quadriplegia, and was below the standard of care.
Keywords: Anterior cervical surgery, Cerebrospinal fluid leak, Corpectomy, DuraSeal, Fibrin sealant, Fusion, Ossification of the posterior longitudinal ligament, Quadriplegia
INTRODUCTION
The use of DuraSeal in the anterior cervical spine is directly contraindicated as this is considered a “confined space.” The package insert states: “Do not apply DuraSeal® (Integra LifeSciences, Princeton NJ) hydrogel to confined bony structures where nerves are present since neural compression may result due to hydrogel swelling (…up to 12% of its size in any direction).” Further, DuraSeal should not be applied over an unrepaired dural tear. Rather, DuraSeal “… is indicated as an adjunct to sutured dural repair during spine surgery to provide watertight closure,” but “…(not for) a gap greater than 2 mm….” Here, we present a geriatric patient whose surgeon performed an anterior cervical C4, C5 corpectomy with C3-C6 strut fusion causing quadriplegia due to the contraindicated anterior cervical use/ application of DuraSeal.
PATIENT’S PREOPERATIVE EVALUATION
An elderly patient was originally seen by an otolaryngologist who ordered a neck CT scan. It showed multilevel continuous cervical OPLL extending from C3-C6, resulting in marked anterior cord compression with accompanying signs of dural penetration [
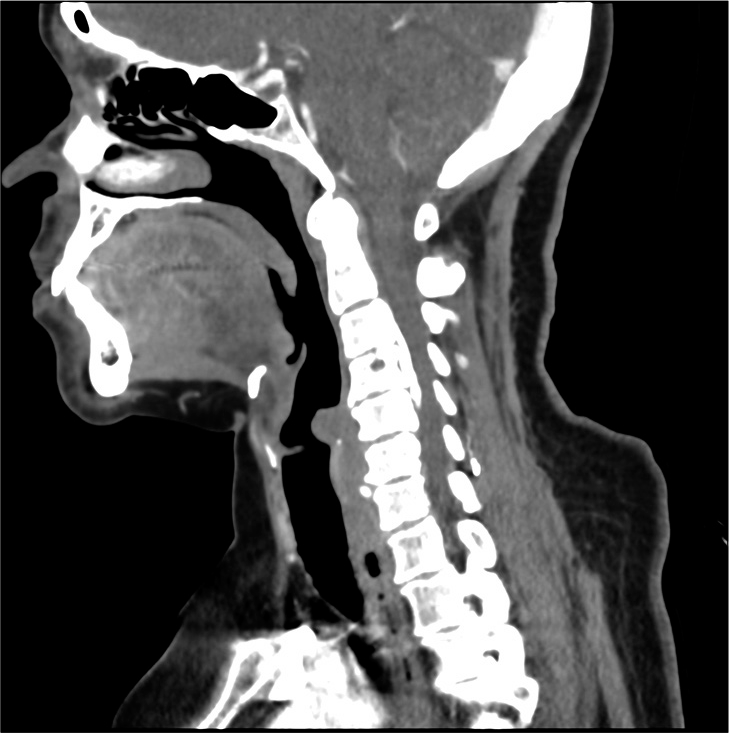
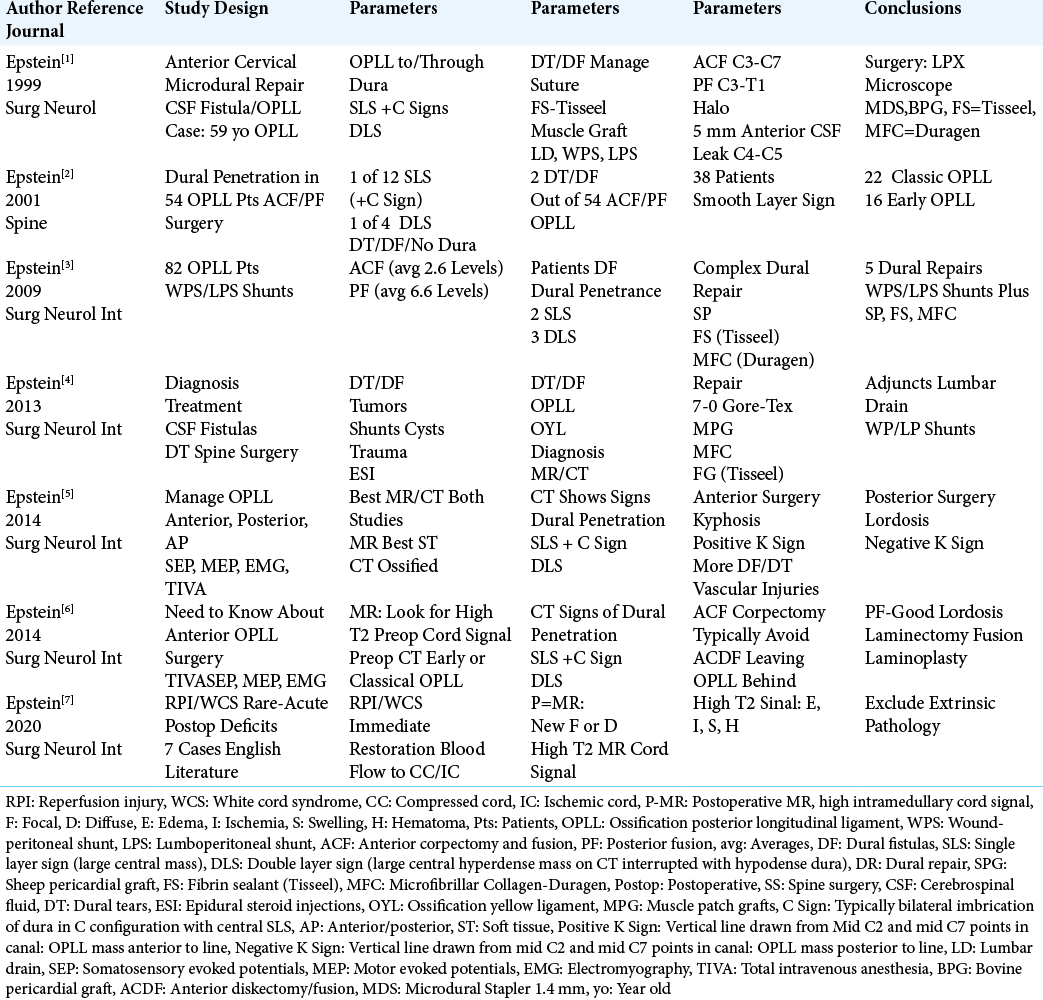
Figure 1:
Preoperative paramedian sagittal soft-tissue CT study showing classical cervical OPLL extending superiorly behind the C3 vertebral body, and inferiorly all the way to the mid portion of C5. The hypodense areas represented dural incorporation between the posterior vertebral bodies and the ossified OPLL. The different configurations represented the single, C, and double layer signs of dural penetrance. Anterior cervical surgery was, therefore, more likely than not, to result in an anterior intraoperative cerebrospinal fluid (CSF) leak. Anticipating this leak would have probably resulted in correctly planning it’s appropriate intraoperative/postoperative management. Further, the negative K sign (measured from the middle of the canal at C2 to the middle of the canal at C7 with a vertical line between the two points) confirmed that a direct anterior approach (i.e., consisting of a corpectomy) would be warranted to adequately resect the ventral OPLL mass, and decompress the cord.
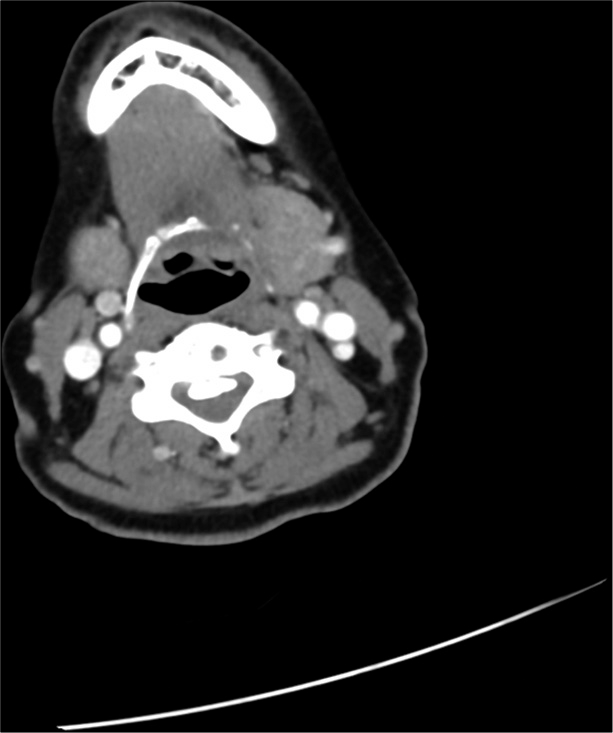
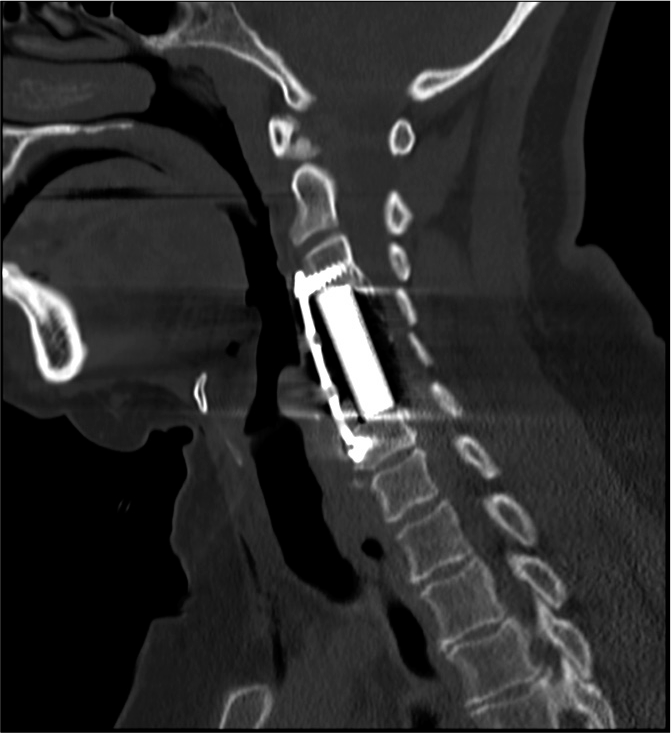
Figure 2:
Preoperative axial CT scan obtained at the mid C4 level showed the classical single layer and bilateral C signs contributing to ventral cord compression. Furhter, these signs had likely already penetrated the ventral dura and surgical resection would more likely than not result in a CSF leak.
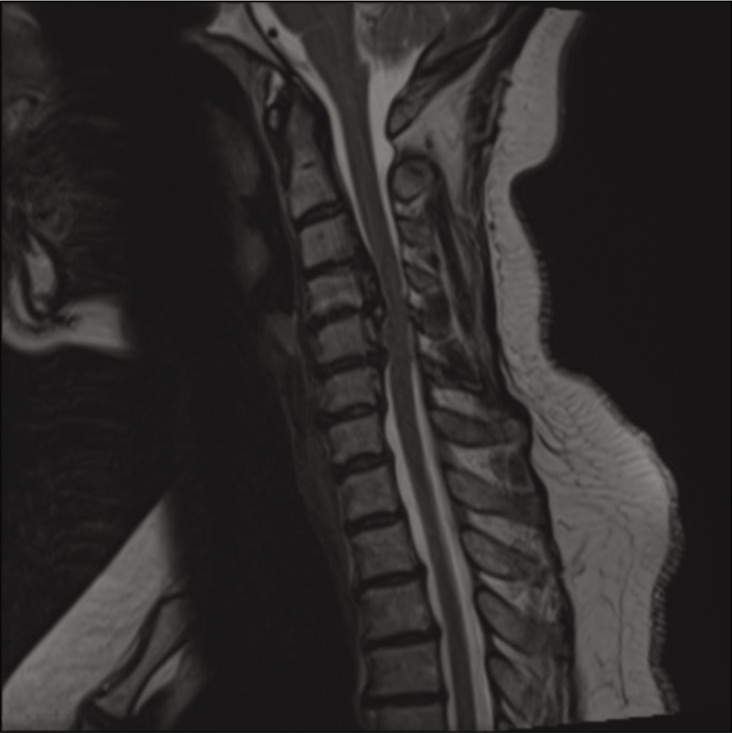
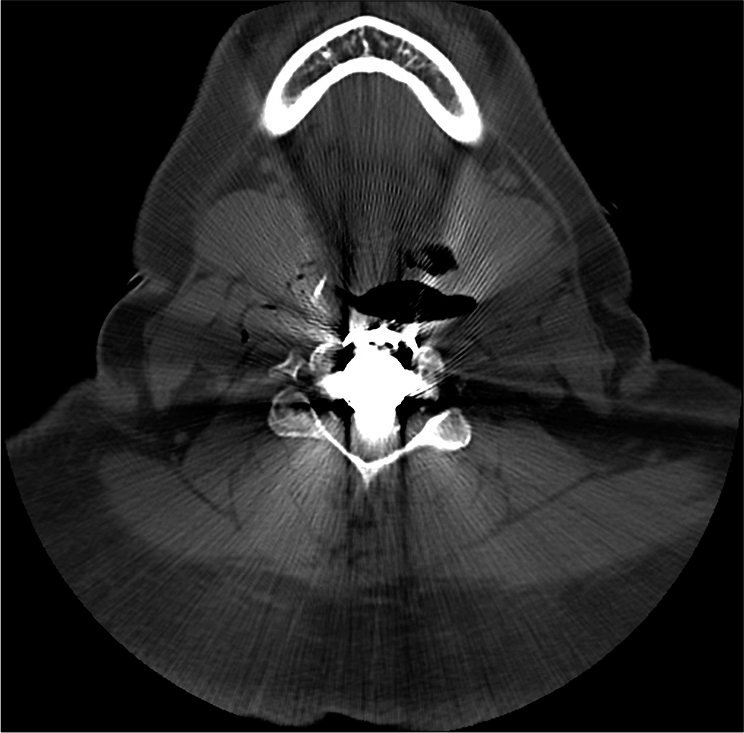
Figure 3:
Preoperative midline sagittal T2-weightd MR of C3-C4 to C5-C6 OPLL. This study confirmed the massive ventral OPLL C3-C6 and the resultant increased cord signal extending from above the C3-C4 level down to the mid C5 level. Further, the inhomogeneous, signal within the OPLL mass reflected active bone marrow production occurring in Haversian canals. This study also showed the negative K sign (vertical line between midpoint C2 to mid-point C7) indicating that a direct anterior approach to OPLL was warranted.
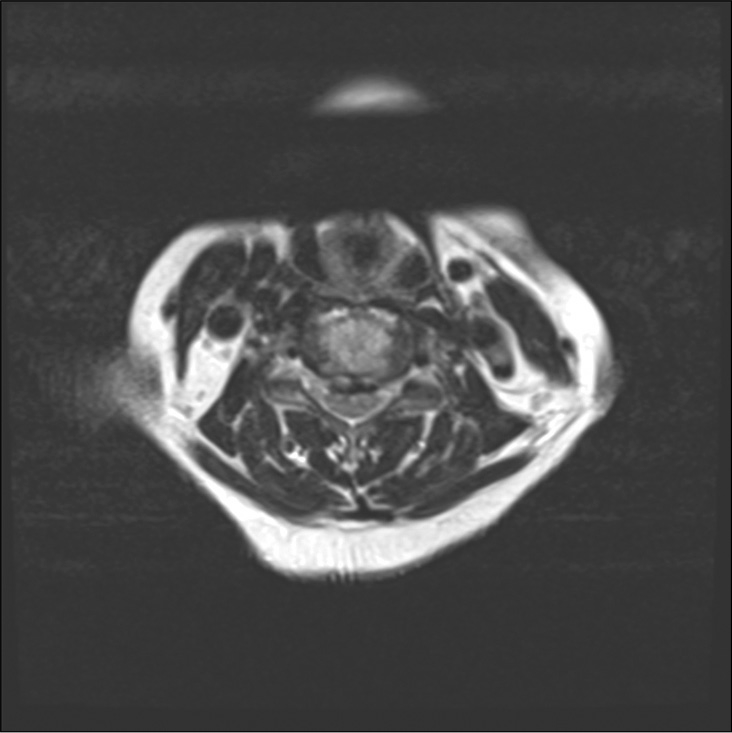
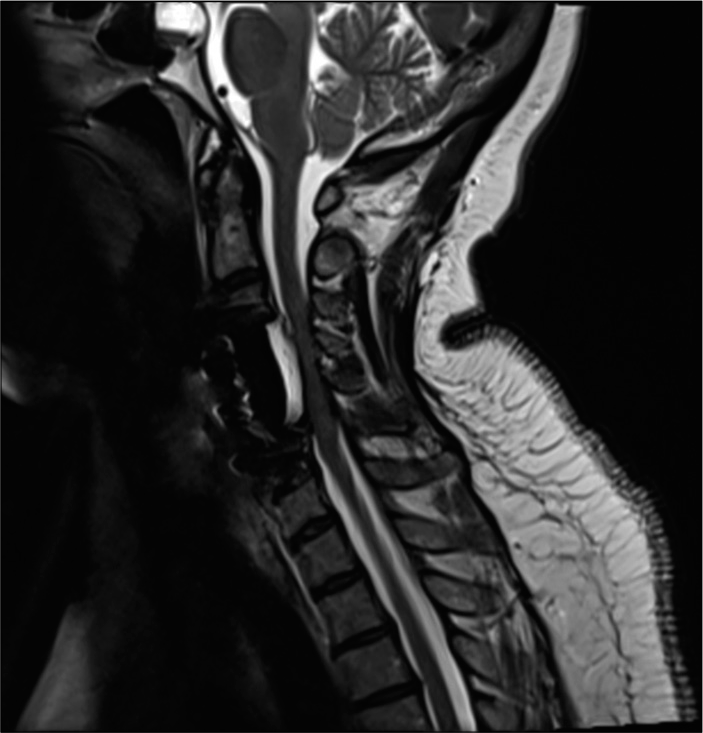
Figure 4:
Axial T2 weighted MR at the C4-C5 level showing massive ventral OPLL resulting in cord compression. Note the inhomogeneity of the ventral mass that indicated active boen marrow production occurring in Haversian canals. There is also some high signal intensity within the cord itself reflecting the severity of cord compression at this level.
FAILURE TO ASSESS PREOPERATIVE CT THAT SHOWED OPLL AND DURAL PENETRANCE
The best way to document the presence of OPLL is with a CT study, but MR’s can also show changes indicative of OPLL’s presence [
Figure 5:
This STAT postoperative parasagittal cervical non-contrast CT study showed the C3-C6 anterior strut graft in place along with the anterior plate. The hypodense mass dorsally extending from the C3-C4 through the C5-C6 levels (i.e. posterior to the graft and anterior to the cord itself) contributed to marked ventral cord compression (i.e., this was due to Duraseal as confirmed on the postoperative MR). Further, the spinal surgeon simply read this STAT postoperative CT study as ruling out an anterior epidural hematoma despite the radiologist’s statement that he (the radiologist) could not discern what was going on inside the spinal canal due to marked metallic artifact, thus indicating an additional MR was warranted.
Figure 6:
STAT postoperative axial non-contrast CT scan at mid C4 level showing marked metallic artifact from cervical disc arthroplasty device. The presence of the device made it difficult for the radiologist to interpret/identify any pathology within the spinal canal. This further confirmed that a STAT MR was warranted.
FAILURE TO USE INTRAOPERATIVE NEURAL MONITORING (IONM); SEP (SOMATOSENSORY EVOKED POTENTIALS), MEP (MOTOR EVOKED POTENTIALS), EMG (ELECTROMYOGRAPHY)
This very complex multilevel anterior cervical corpectomy C4, C5 with C3-C6 fusion for OPLL was, unfortunately, performed without the benefit of intraoperative SEP, MEP or EMG monitoring. Had IONM been utilized, the MEP/ SEP potentials would probably have been lost shortly after the DuraSeal was applied (i.e. the hydrophillic DuraSeal takes several minutes to expand), and would, to a reasonable degree of medical probability, have prompted the surgeon to remove the anteriorly compressive DuraSeal mass. Certainly, the MEP/SEP signals would have dropped out by the time the patient was closed. IONM loss along with the patient’s postoperative severe quadriparesis should have led to an emergent postoperative cervical MR. This would have documented massive anterior cord compression from DuraSeal, and should have led to DuraSeal’s immediate anterior removal. Unfortunately, the postoperative MR was delayed until 9 hours following surgery, by which time the patient was fully quadriplegic. Further, the patient underwent the wrong secondary surgery (i.e. laminectomy/posterior fusion). In short, the lack of IONM contributed to this patient’s postoperative permanent neurological injury.
SPINE SURGEON’S NEGLIGENT SURGERY WITHOUT AN OPERATING MICROSCOPE
In this case, the surgeon performed a multilevel anterior corpectomy (C4, C5) and fusion (C3-C6) for OPLL without an operating microscope. This certainly increased the probability of a CSF fistula, as makes it even more difficult to define a plane between the underlying dura and the overlying OPLL mass (anterior exposure).
SPINE SURGEON’S NEGLIGENT SURGERY WITHOUT AN ARTERIAL LINE
The surgeon proceeded to negligently perform the C4 and C5 anterior corpectomy with C3-C6 strut/plated fusion without insisting that anesthesia place an arterial line. Notably, during the first surgery, the patient lost over 1000 cc of blood over 15 minutes, resulting in at least 30 minutes of severe hypotension requiring massive repeated doses of pressors. Such hypotensive events can readily occur during anterior OPLL resection, not only due to direct bleeding from the OPLL mass itself (i.e. open Haversian canals), but also to increased bleeding from the valveless epidural veins (Batson’s plexus) with/without a CSF leak, or to a potential vertebral artery injury. Here, an arterial line was finally placed 3 hours following the end of the first operation.
ANTERIOR SURGERY RESULTED IN MASSIVE CSF LEAK THAT THE SURGEON NEGLIGENTLY TREATED WITH CONTRAINDICATED GELFOAM/FLOSEAL, DURAGEN, AND DURASEAL
The surgeon performed a C4 and C5 anterior corpectomy, and C3-C6 fusion with plating for what he wrongly diagnosed as disc disease with osteophytes. However, he encountered severe OPLL that he should have known about had he read or reviewed the preoperatively available CT scan. Further, he misleadingly recorded in his operative note that the CSF fistula was just a few mm in size. Nevertheless, during his deposition he finally acknowledged that the dural defect was much larger than that, comprising 50-100% of the C4 and C5 corpectomy expanse.
Use of Contraindicated Gelfoam and FloSeal
When the CSF leak occurred, resulting in copious epidural bleeding (i.e., loss of the dural tamponade of the valveless epidural venous plexus), the spine surgeon placed Gelfoam (Pharmacia & Upjohn Co., Division of Pfizer Inc., NY, NY) and Floseal (Baxter Healthcare Corporation, Hayward, CA). The operative note stated: “Venous bleeding from epidural venous plexus was stopped with Gelfoam/Thrombin and Floseal.” Notably, the warning on the package insert for FloSeal states it is not a substitute for meticulous hemostasis, and should be removed to avoid swelling (i.e., 10-20% after application to surrounding anatomic areas) in confined spaces. The package insert further warns that it’s safety has not been established in neurosurgery.
Contraindicated Application of Duragen
Although the surgeon claimed the intraoperative cerebrospinal fluid leak/fistula (CSF) was readily controlled with a “small cottonoid” (i.e., direct quote from operative report), in fact, there was a major dural tear without any approximated dural edges. Further, during his deposition, the surgeon finally acknowledged he could not close the dura as it was “incompetent,” had “irregular edges,” and could not be “sewn primarily.” Additionally, he “...could not tell the full extent of the dural breach...,” but ultimately admitted it could have been between 50-100% of the length of the 2-level corpectomy. Next, the surgeon placed Duragen (Integra LifeSciences, Plainsboro, NJ) over the massive dural defect despite the warnings of the package insert that state: “DuraGen Secure Dural Regeneration Matrix is not designed, sold or intended for use except as described in the Indications for Use and is contraindicated in the following situations.” One of these included; “For repair of spinal neural tube defects or anterior spinal surgery with dural resection.”
Contraindicated Application of DuraSeal
Next, the surgeon ignored the clear contraindication to applying DuraSeal over Duragen in the anterior cervical spine. He specifically stated: “A piece of Duragen was placed over thecal sac and reinforced with Dural Sealant.” Here, the application of DuraSeal (Integra LifeSciences, Princeton NJ) in the anterior cervical spine was neglitent. It was a direct contraindication to use DuraSeal in a “...confined space...”, that here included the anterior cervical spine. The package insert states: “Do not apply DuraSeal® (Integra LifeSciences, Princeton NJ) hydrogel to confined bony structures where nerves are present since neural compression may result due to hydrogel swelling (…up to 12% of its size in any direction) that it should not be applied over an unrepaired dural tear.”
Failure to Consider Use of Postoperative Lumbar Drain to Treat CSF Fistula
Had the spinal surgeon read the report or reviewed the preopereative CT images, he would have known that the patient had OPLL. This would have enabled him to anticipate and plan to treat an intraoperative CSF fistula. More likely than not, he would have opted to use a postoperative lumbar drain for CSF diversion, and avoided using the directly contraindicated Gelfoam/Floseal, Duragen, and most critically, the DuraSeal.
SPINE SURGEON’S FAILURE TO DIAGNOSE IN A TIMELY FASHION AND APPROPRIATELY TREAT POSTOPERATIVE QUADRIPARESIS/ QUADRIPLEGIA
Following surgery, which included a major intraoperative CSF leak, the patient was taken back to the postoperative care unit “deep” following extubation. Instead of waiting to examine the patient himself once the anesthetic wore off, the surgeon went directly into his next case. This left the anesthesiologist to examine the patient who was severely quadriparetic when she was finally awake. When informed of her paralysis, the surgeon asked for a STAT non contrast scan. This was performed within approximately 2 postoperative hours; he interpreted it as showing no postoperative epidural hematoma. However, even the radiologist commented that the CT’s significant metallic artifact interfered with adequately assessing what was going on within the spinal canal [
POSTOPERATIVE MR DELAYED FOR OVER 9 HOURS ALLOWING QUADRIPARESIS TO EVOLVE TO QUADRIPLEGIA
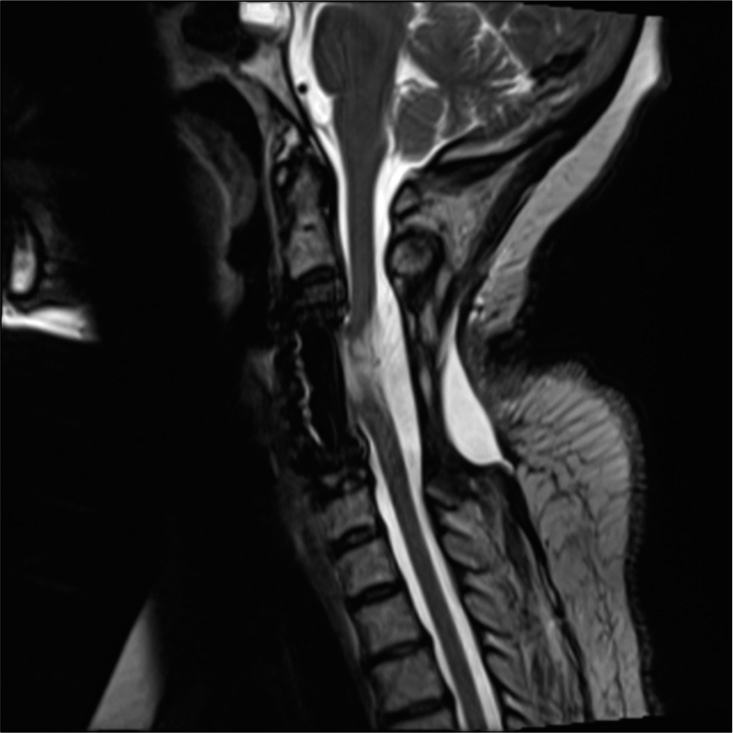
When the patient became fully quadriplegic 9 hours postoperatively, a STAT MR was finally obtained. Notably, there was no adequate explanation for this delay other than the surgeon having just ordered it be done “sometime overnight”. The MR documented massive anterior cord compression due to DuraSeal (i.e., 33 mm cephalad/caudad, and axially 8–13 mm) [
Figure 7:
Delayed postoperative sagittal MR confirming massive anterior cord compression from the C3-C4 through the C5-C6 levels due to hyperintense DuraSeal. This midline sagittal postoperative MR scan, unfortunately obtained another 6 + hours after the CT scan but 9 + hours after the first surgery ended, showed massive anterior cord this postoperative midline sagittal MR scan showed massive anterior cord compression/edema attributed to the hyperintense DuraSeal Mass. Also note the new diffuse increased signal within the cord extending from C3-C6 consistent with acute cord edema.
FAILURE TO REOPERATE IN A TIMELY FASHION AND TO PERFORM THE CORRECT SECOND SURGERY: DIRECT ANTERIOR REMOVAL OF COMPRESSIVE DURASEAL
The surgeon then mistakenly performed the wrong second operation despite the clear MR documentation of a huge anterior DuraSeal Mass compressing the spinal cord. His second surgery consisted of a laminectomy/ posterior fusion rather than directly and correctly removing the anteriorly compressive DuraSeal.
FAILURE TO OBTAIN AN IMMEDIATE POSTOPERATIVE MR SCAN FOLLOWING THE SECOND SURGERY, AND FAILURE TO PERFORM AN ACUTE THIRD OPERATION TO REMOVE ANTERIOR DURASEAL RESULTED IN THE PATIENT’S PERMANENT AND IRREVERSIBLE QUADRIPLEGIA
The spine surgeon failed to order a STAT MR scan following the 2nd surgery, despite the fact that the patient remained quadriplegic. In fact, the only postoperative MR study the surgeon requested/performed was obtained 6 months later. Therefore, the patient received no emergent third operation (i.e. to remove the anteriorly compressive DuraSeal), thus leaving her fully and completely quadriplegic [
Figure 9:
Six month postoperative T2 midline sagittal MR study showing cord atrophy/myelomalacia and ventral adherence of the cord to the previously placed DuraSeal. Note the capacious canal at this point due to the marked cord atrophy. There is also a residual hyperintense posterior fluid-signal collection likely attributed to the prior cervical laminectomy.
DISCUSSION
Limitations of MR in the Documentation of OPLL
MR examinations show OPLL, particularly when there is retrovertebral extension of isointense/hypointense masses consisting of either hypertrophied or ossified posterior longitudinal ligament [
CT: Best Documentation of OPLL (Hypertrophy (HPLL)/ Classic Ossification)
CT scans more typically provide clearer, more direct, hypoisodense (hypertrophied PLL) or hyperdense (mature classical OPLL) images of HPLL/OPLL.[
Contraindicated use of Gelfoam/FloSeal, Duragen and DuraSeal to Treat Massive Anterior Cervical CSF Leak: Placement of a Postoperative Lumbar Drain Would Have Temporarily Sufficed
The spine surgeon was negligent in his “contraindicated” application of Gelfoam/FloSeal, Duragen, and DuraSeal to the anterior cervical spine, in a “confined space” where all of these products could substantially swell. Further, the latter two were directly contraindicated for use in the presence of a massive, unrepaired anterior cervical dural leak. Hence, the choice to apply Duragen followed by DuraSeal resulted in MR-documented severe anterior cord compression and resultant edema, causing the patient’s postoperative quadriparesis/plegia [
The Patient’s Postoperative Quariplegia was Not Due to a Reperfusion Injuyr (RPI) or White Cord Syndrome (WCS)
RPI/WCS is an extremely rare diagnosis attributed to an acute reperfusion of previously markedly compressed/ischemic spinal cord tissues [
CONCLUSION
After the first surgery, the “contraindicated anterior cervical application of DuraSeal into a confined space to treat a massive, unrepaired CSF leak caused the patient’s quadriplegia [
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Commentary
Contraindicated Use of DuraSeal In Anterior Cervical Spine Led to Quadriplegia
This Medicolegal Corner (Spine) highlights the issues of complex spine surgery in this complicated OPLL case. The failure to recognize OPLL and underestimate the degree of difficulty in a case that was destined to be problematic from the start was a big issue in this case. The lack of intraoperative monitoring brings into question if this SCI occurred during the case or with the hydrophilic expansion of the DuraSeal at the end of the case. Our group has experienced five complications using a similar tissue sealant. BioGlue is FDA and CE Mark approved as an adjunct to standard methods of surgical repair to band seal and/or reinforce soft tissues. Dural repair is listed in the soft tissue indications. Five patients were re-operated for neural compression secondary to an inflammatory mass within 6-50 weeks following spinal surgery (& 1 following cranial surgery for frontal sinus repair). In our cases the weakness appeared in a more delayed fashion after the product expanded. With quick MRI identification of the mass compressing the cord, we were able to remove the mass with urgent surgery and restore neurologic function. Pathology revealed an aseptic mass with reactive granulation tissue.
Jamie Baisden, M.D.
Professor of Neurosurgery
MCW -Neurosurgery
HUB 4th Floor
8701 Watertown Plank Road
Milwaukee, WI 53226
Cell 262-902-0059
414-248-4997
References
1. Epstein NE, Hollingsworth RD. Anterior cervical micro-dural repair of cerebrospinal fluid fistula after surgery for ossification of the posterior longitudinal ligament. Technical note. Surg Neurol. 1999. 52: 511-4
2. Epstein NE. Identification of ossification of the posterior longitudinal ligament extending through the dura on preoperative computed tomographic examinations of the cervical spine. Spine (Phila Pa 1976). 2001. 26: 182-6
3. Epstein NE. Wound-peritoneal shunts: Part of the complex management of anterior dural lacerations in patients with ossification of the posterior longitudinal ligament. Surg Neurol. 2009. 72: 630-4
4. Epstein NE. A review article on the diagnosis and treatment of cerebrospinal fluid fistulas and dural tears occurring during spinal surgery. Surg Neurol Int. 2013. 4: S301-17
5. Epstein NE. Cervical surgery for ossification of the posterior longitudinal ligament: One spine surgeon’s perspective. Surg Neurol Int. 2014. 5: S88-92
6. Epstein NE. What you need to know about ossification of the posterior longitudinal ligament to optimize cervical spine surgery: A review. Surg Neurol Int. 2014. 5: S93-118
7. Epstein NE. Reperfusion injury (RPI)/white cord syndrome (WCS) due to cervical spine surgery: A diagnosis of exclusion. Surg Neurol Int. 2020. 11: 320