- Department of Neurosurgery, Hospital Juárez de México, Instituto Politécnico Nacional, Mexico City, Mexico.
Correspondence Address:
Erik Burgos-Sosa, Department of Neurosurgery, Hospital Juárez de México, Instituto Politécnico Nacional, Mexico.
DOI:10.25259/SNI_484_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Erik Burgos-Sosa, Patricia Bergna-Vazquez, Rafael Mendizabal-Guerra, Arturo Ayala-Arcipreste. Microsurgical and endoscopic-assisted supraorbital keyhole approach for intra-suprasellar cysticercosis. 15-Sep-2023;14:328
How to cite this URL: Erik Burgos-Sosa, Patricia Bergna-Vazquez, Rafael Mendizabal-Guerra, Arturo Ayala-Arcipreste. Microsurgical and endoscopic-assisted supraorbital keyhole approach for intra-suprasellar cysticercosis. 15-Sep-2023;14:328. Available from: https://surgicalneurologyint.com/surgicalint-articles/12559/
Abstract
Background: Treatment for intra/suprasellar cysticercosis can be challenging and may result in visual disturbances if not managed properly. Despite its limited knowledge, an effective surgical option exists to treat this condition. This article presents three cases of sellar cysticercosis, comprising one female and two male patients, managed with microsurgical supraorbital keyhole approach (mSKA) and endoscopic-assisted supraorbital keyhole approach (eaSKA).
Case Description: The first patient is a 35-year-old man with no prior medical history who suffered from memory deficits and visual disturbances due to a sellar cyst pushing the orbitofrontal gyrus treated with mSKA. The second case involved a 52-year-old man who experienced visual deficits caused by a rostral sellar cyst with posterior displacement of the pituitary gland treated with eaSKA. The third case was a 46-year-old woman who experienced decreased visual acuity and memory loss due to multifocal neurocysticercosis (NCC) with sellarsuprasellar cyst extension treated with mSKA. All case diagnoses were confirmed by neuropathology department.
Conclusion: The authors confidently suggest that the SKA is an effective surgical option and could be considered for removing sellar cystic lesions with suprasellar extension. With endoscopic assistance, it improves adequate neurovascular structure visualization.
Keywords: Intrasellar/Suprasellar, Neurocysticercosis, Supraorbital keyhole approach
INTRODUCTION
Neurocysticercosis (NCC) is an infection in the central nervous system (CNS) caused by Taenia solium. This disease is common in developing countries, particularly in Latin America, Asia, and sub-Saharan Africa.[
Fortunately, with the emergence of minimally invasive techniques, and perceptive instrumentation, it has become feasible to treat lesions in this area.[
CASE DESCRIPTION
This study was conducted at Hospital Juárez de México from January 2001 to February 2021. We examined the medical records of patients diagnosed with NCC, with selection criteria focusing only on patients with intrasellar NCC treated with the SKA. Patients who received treatment elsewhere or had surgical procedures were excluded from the study. Our database revealed three patients with intrasellar cysticercosis approached with mSKA, and eaSKA. NCC diagnosis was confirmed by the neuropathology department.
Case 1
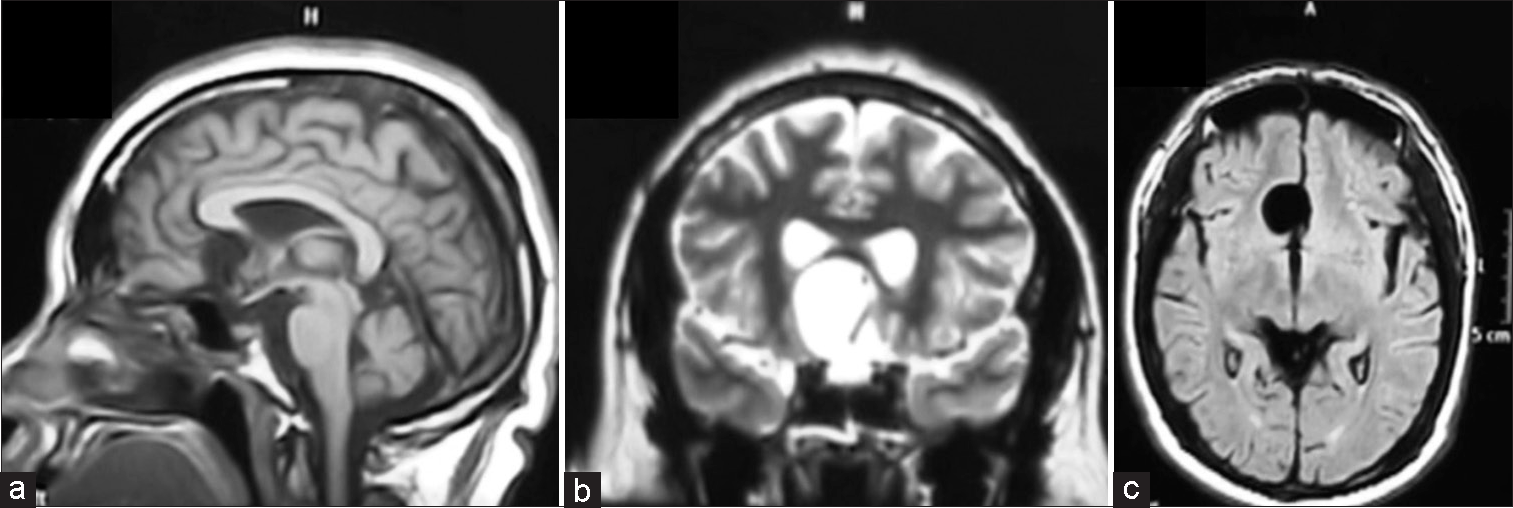
A 35-year-old male patient presented with emotional instability, memory loss, headaches, and changes in vision over the past month. Further, neurological examination revealed limited peripheral vision with decreased visual acuity. Hormonal tests were normal. Magnetic resonance imaging (MRI) scan showed a cystic lesion in the sellar region extending to the suprasellar area [
Figure 1:
Magnetic resonance imaging study of case 1. (a) T1-weighted sagittal plane, hypointense lesion in the sellar region with extension to the suprasellar area adjacent to the mesial frontal area. (b) T2-weighted coronal plane shows the consistency of cystic lesion, (c) with minimal perilesional edema in axial fluid attenuated inversion recovery image.
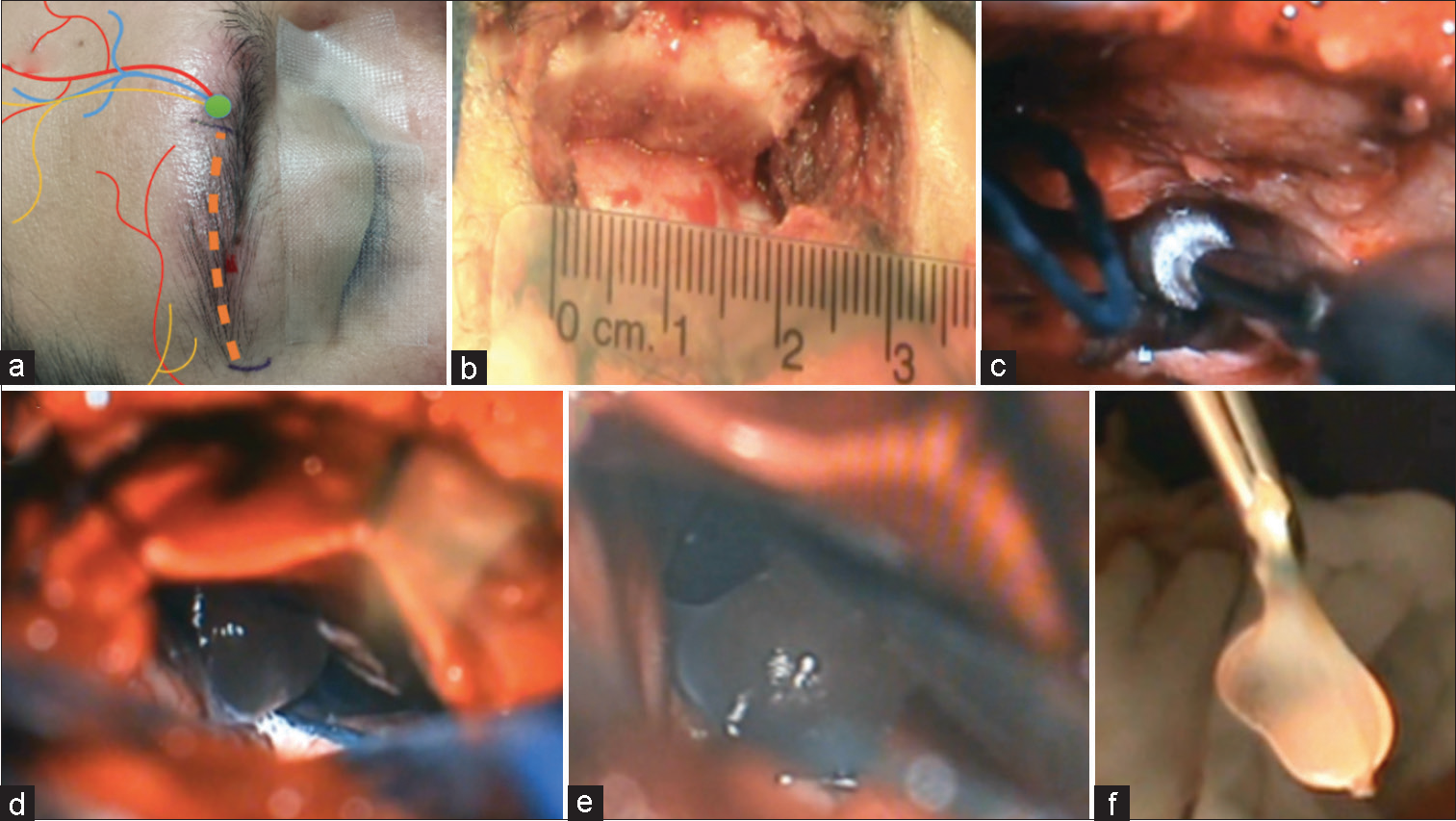
The patient was placed in a supine position, with contralateral 10° rotation, accompanied by slight elevation and extension of the head. The incision was performed over the eyebrow through the supraorbital rim with the temporalis muscle detached. A simple burr-hole was performed slightly below the junction of the superior temporal line-orbital rim. Before accessing the dural aperture during intracranial surgery, drilling the frontal eminences was an essential step to gain proper exposure and visualization. Dura was opened in the usual fashion. The next step involved opening cisterns to drain cerebrospinal fluid (CSF) to minimize frontal lobe retraction and maximize frontal lobe drop. A cyst was encountered during the procedure with slight arachnoid adhesions in the mesial frontal lobe and optic chiasm. During the surgery, one of the cysts was accidentally ruptured, and it was managed using an aspirator to trap the defect, followed by retraction of the compartmental cysts. This effectively minimized the spill-out and reduced the risk of arachnoiditis [
Figure 2:
Microsurgical supraorbital keyhole approach of case 1. (a) Planned eyebrow incision preserving the marked neurovascular structures (orange dotted line). Green dot represent the supraorbital foramen. (b) A small craniotomy was performed in relation to the supraorbital ridge, approximately 3 cm × 3 cm. After dural retraction of the frontal dura, (c) drilling of the frontal eminences with a diamond drill tip to gain more exposure, the dural leaflet was tacked up, (d) visualization of the cyst, and (e) adequate dissection of the cyst, preserving the planes of the capsule cyst to avoid its rupture. (f) The cyst was resected.
It was crucial to administer dexamethasone 8 mg once a day during and after the surgery. This treatment lasted for 72 h, and then, the patient took oral steroids accompanied by oral albendazole for 21 days. The pathology sample confirmed the diagnosis [
Case 2
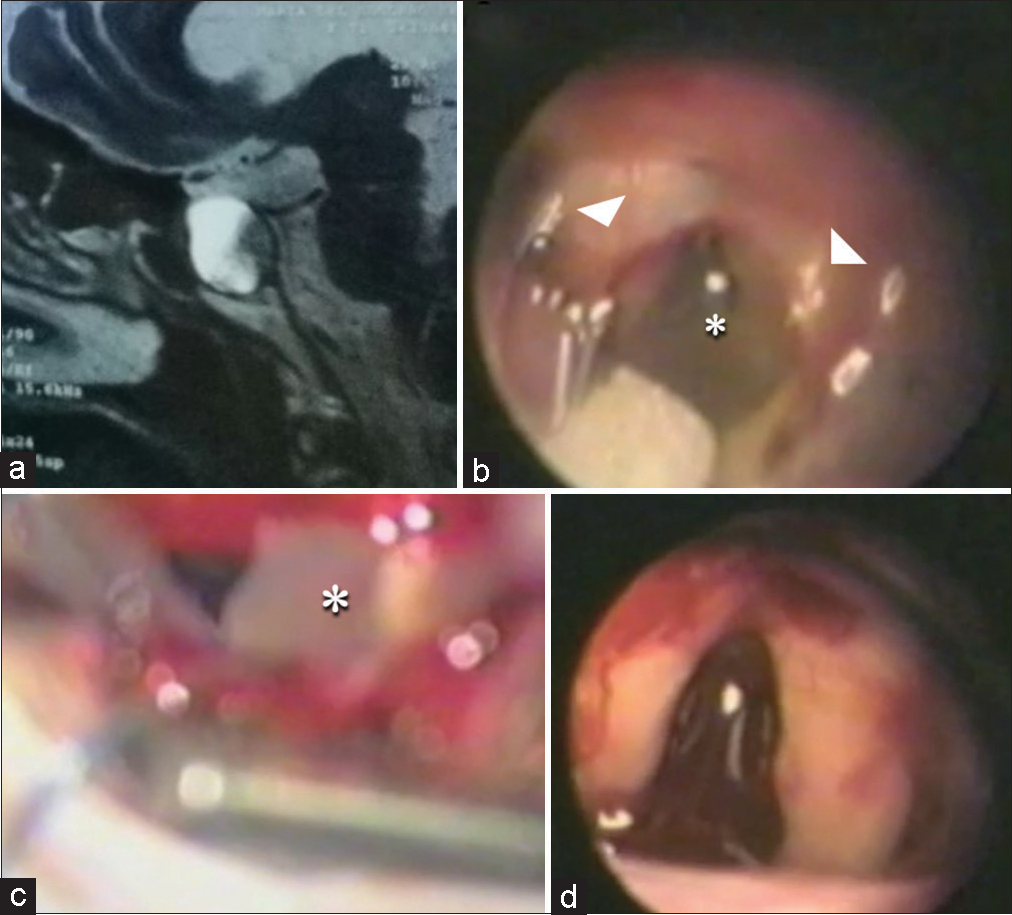
A 52-year-old man, who had a long-term history of diabetes, began experiencing a gradual loss of vision over the past 4 months. On the clinical examination, it was discovered that he had slightly restricted peripheral vision. An MRI revealed a lesion in the sellar location with a small extension in the subchiasmatic and prechiasmatic areas. An endoscope was used to amplify the corridor to reach the affected region, visualize the cyst, and perform the necessary dissection.
For this case, a microscope and a rigid endoscope with lenses 0° and 30° were utilized. We followed the previously described approach, exposing the cyst and dissecting it under the optic nerve. Bayonet forceps were used for slight traction, resulting in a minor cyst spill. To aid in dissection, we re-introduced the endoscope and carefully retracted the cyst. During the process, we observed a yellowish punctate hemorrhage and rubberized consistency [
Figure 4:
Magnetic resonance imaging (MRI) and surgical resection with endoscopic-assisted supraorbital keyhole approach of case 2. (a) MRI, T2-weighted sagittal plane shows sellar cyst with slight suprasellar extension. (b) The endoscope is introduced for endoscopic assessment of the cyst (an asterisk marks the intrasellar parasitic cyst, and head arrows mark the optic nerves). (c) Resection of the cyst with rongeurs under microscopic visualization (asterisk select the cyst), and (d) reassessment of the sellar area.
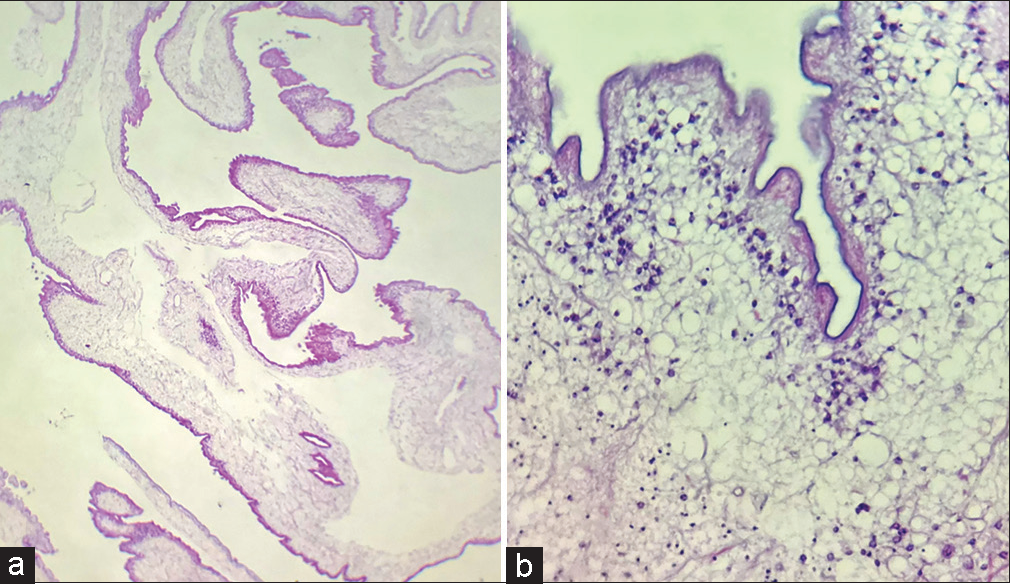
The patient was under dexamethasone and albendazole for 21 days, following the same procedure as the previous case. The pathology department confirmed the diagnosis [
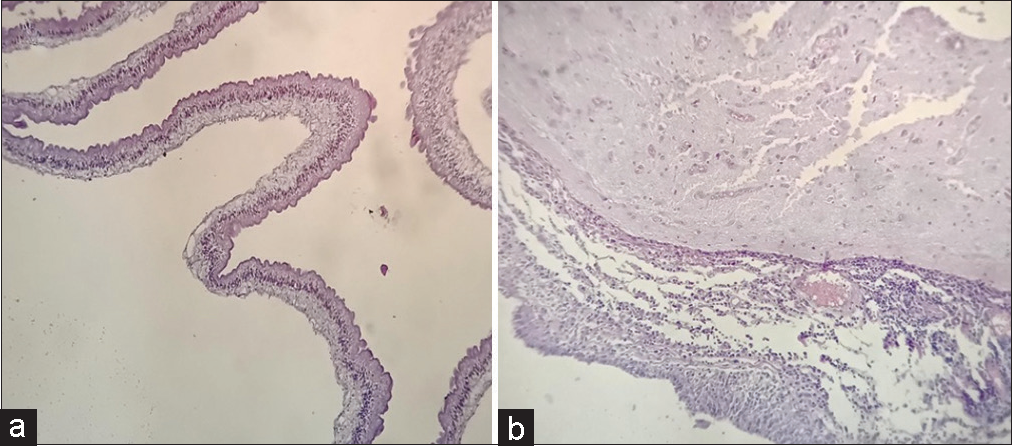
Figure 5:
Histopathological findings of case 2. Under high magnification, ×10, and ×40. (a and b) Histopathology of the cyst showed three layers (cuticular, cellular, and reticular). Gliosis around parenchymal cysticerci, with granulomatous reaction with epithelioid cells and foreign body giant cells.
Case 3
A 46-year-old woman presented with progressive headaches, an unsteady gait, and dorsolateral frontal syndrome over the past 5 months. Her symptoms have worsened over the past 3 weeks, including visual loss and tonic-clonic seizures. Clinical examination revealed decreased visual acuity, altered executive function, and memory loss. MRI scans confirmed multifocal NCC in the temporal and frontal lobes, with compromise in the sellar-suprasellar area. The mSKA procedure was successfully performed as the first case technical description, with pathological findings consistent with NCC. In the postoperative, the patient received steroids and albendazole for 21 days, complemented with an anticonvulsant. After 3 months of follow-up, the patient reported improved visual acuity. At 12 months, the patient recovered her paresis in the leg and her visual acuity.
DISCUSSION
In this study, we described the surgical treatment for three cases of cysticercosis located in the sellar/suprasellar region using microsurgical and endoscopic-assisted SKA. The rarity of cysticercosis in the sellar location has led to a lack of established treatments.[
Helminthic infections in the CNS are commonly caused by NCC and have become a major public health problem. Approximately 50% of NCC in the sellar area may manifest as a solitary cyst. The other cases typically involve either extra sellar or disseminated CNS involvement.[
The treatment of sellar/suprasellar NCC has not been adequately discussed, and the available information regarding surgical treatment options for intrasellar cysticercosis is scarce. Surgical treatment has proven more effective than medical therapy for these lesions. Therefore, the recommended course of action is the surgical removal of the cysticerci.[
The advancements in microsurgery have made it possible to offer various surgical options for pathologies affecting sellar, parasellar, and anterior cranial fossa locations.[
The supraorbital approach is an adequate alternative for anterior skull base lesions in sellar, parasellar, and suprasellar areas. This involves a limited incision with minimal tissue manipulation and minimizes the frontal lobe retraction, with subsequent decreased pain and rapid healing instead of conventional approaches.[
There are some drawbacks to using mSKA, including the risk of hypoesthesia and eyebrow alopecia that can be prevented with gentle manipulation of the tissue and avoiding overuse of the monopolar during the initial stages of the approach. SKA can also be limited by extensive frontal sinus pneumatization and lesions below the sphenoid wing.[
In vascular pathology or large solid lesions, a different approach is mandatory to achieve better exposure and vascular control. The angle view in SKA can be challenging, making it difficult to visualize adjacent structures in the blood field, particularly in cases of massive brain edema.[
The rupture of the helminthic cyst does not typically result in adverse complications or meningitis.[
The mSKA and eaSKA methods could have similar effectiveness with several approaches,[
CONCLUSION
Prompt surgical resection of intra/suprasellar cysticercosis may result in the resolution of clinical symptoms. For a predominance sellar lesion, eaSKA is recommended; this allows a better understanding of the configuration of the cystic capsule and the adjacent neurovascular structures. It is crucial to avoid incidental rupture of the capsule by applying continuous traction during aspiration to reduce the inflammatory reaction, in addition to medical treatment with steroids and anthelmintics (this last is not advisable to administer before the surgery).[
Declaration of patient consent
Patients’ consent not required as patients’ identities were not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The author(s) confirms that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
Acknowledgments
The authors would like to acknowledge T. Cuesta-Mejia, M.D. from the neuropathology department for descriptions and analysis of the pathology lamellas.
References
1. Arnaout MM, Luzzi S, Galzio R, Aziz K. Supraorbital keyhole approach: Pure endoscopic and endoscope-assisted perspective. Clin Neurol Neurosurg. 2020. 189: 105623
2. Boecher-Schwarz HG, Hey O, Higer HP, Perneczky A. Intrasellar cysticercosis mimicking a pituitary adenoma. Br J Neurosurg. 1991. 5: 405-7
3. Colli BO, Carlotti CG, Assirati JA, Machado HR, Valença M, Amato MC. Surgical treatment of cerebral cysticercosis: Long-term results and prognostic factors. Neurosurg Focus. 2002. 12: e3
4. Couldwell WT, Zee CS, Apuzzo ML. Definition of the role of contemporary surgical management in cisternal and parenchymatous cysticercosis cerebri. Neurosurgery. 1991. 28: 231-7
5. Cuellar-Hernandez JJ, Valadez-Rodriguez A, Olivas-Campos R, Tabera-Tarello P, Juan-Orta DS, Segura-López R. Intrasellar cysticercosis cyst treated with a transciliary supraorbital keyhole approach-A case report. Surg Neurol Int. 2020. 11: 436
6. Del Brutto OH. Neurocysticercosis. Handb Clin Neurol. 2014. 121: 1445-59
7. Del Brutto OH, Del Brutto VJ. Intrasellar cysticercosis: A systematic review. Acta Neurol Belg. 2013. 113: 225-7
8. Del Brutto OH, Guevara J, Sotelo J. Intrasellar cysticercosis. J Neurosurg. 1988. 69: 58-60
9. Del Brutto OH, Nash TE, White AC, Rajshekhar V, Wilkins PP, Singh G. Revised diagnostic criteria for neurocysticercosis. J Neurol Sci. 2017. 372: 202-10
10. Fedor K, editors. Chirurgie des gehirns und rückenmarks nach eigenen erfahrungen. Berlin: Urban and Schwarzenberg; 1908. p.
11. Frazier CH. An approach to the hypophysis through the anterior cranial fossa. Ann Surg. 1913. 57: 145-50
12. Garcia HH, Nash TE, Del Brutto OH. Clinical symptoms, diagnosis, and treatment of neurocysticercosis. Lancet Neurol. 2014. 13: 1202-15
13. Kelesidis T, Tsiodras S. Hypopituitarism caused by neurocysticercosis. Am J Med Sci. 2011. 341: 414-6
14. Khan DZ, Muskens IS, Mekary RA, Zamanipoor Najafabadi AH, Helmy AE, Reisch R. The endoscope-assisted supraorbital keyhole approach for anterior skull base meningiomas: An updated meta-analysis. Acta Neurochir (Wien). 2021. 163: 661-76
15. Lupret V, Sajko T, Beros V, Kudelić N, Lupret V. Advantages and disadvantages of the supraorbital keyhole approach to intracranial aneurysms. Acta Clin Croat. 2006. 45: 91-94
16. Ndlovu B, Sattar MO, Mkhaliphi MM, Leola K, Mpanza MN, Ouma JR. Supraorbital eyebrow approach: A single-center experience. Surg Neurol Int. 2022. 13: 566
17. Ormond DR, Hadjipanayis CG. The supraorbital keyhole craniotomy through an eyebrow incision: Its origins and evolution. Minim Invasive Surg. 2013. 2013: 296469
18. Prosser PR, Wilson CB, Forsham PH. Intrasellar cysticercosis presenting as a pituitary tumor: Successful transsphenoidal cystectomy with preservation of pituitary function. Am J Trop Med Hyg. 1978. 27: 976-8
19. Rafael H, Gómez-Llata S. Intrasellar cysticercosis. Case report. J Neurosurg. 1985. 63: 975-6
20. Reisch R, Perneczky A. Ten-year experience with the supraorbital subfrontal approach through an eyebrow skin incision. Neurosurgery. 2005. 57: 242-55
21. Robertson FC, Lepard JR, Mekary RA, Davis MC, Yunusa I, Gormley WB. Epidemiology of central nervous system infectious diseases: A meta-analysis and systematic review with implications for neurosurgeons worldwide. J Neurosurg. 2018. 130: 1107-26
22. Sotelo J, Del Brutto OH. Review of neurocysticercosis. Neurosurg Focus. 2002. 12: e1
23. Stern WE. Neurosurgical considerations of cysticercosis of the central nervous system. J Neurosurg. 1981. 55: 382-9
24. White AC, Coyle CM, Rajshekhar V, Singh G, Hauser WA, Mohanty A. Diagnosis and treatment of neurocysticercosis: 2017 Clinical practice guidelines by the infectious diseases society of America (IDSA) and the American society of tropical medicine and hygiene (ASTMH). Clin Infect Dis. 2018. 66: e49-75
25. Wilson DA, Duong H, Teo C, Kelly DF. The supraorbital endoscopic approach for tumors. World Neurosurg. 2014. 82: e243-56
26. Yasargil MG, Fox JL. The microsurgical approach to intracranial aneurysms. Surg Neurol. 1975. 3: 7-14
27. Zheng X, Liu W, Yang X, Gong J, Shen F, Shen G. Endoscope-assisted supraorbital keyhole approach for the resection of benign tumors of the sellar region. Minim Invasive Ther Allied Technol. 2007. 16: 363-6