- Department of Neurosurgery, University of Texas Health Science Center, San Antonio, Texas, USA
- Department of Neurosurgery, Neurosurgical Associates of San Antonio PA, San Antonio, Texas, USA
- Department of Neurosurgery, University of Texas School of Law, Austin, Texas, USA
Correspondence Address:
Byron C. Branch
Department of Neurosurgery, Neurosurgical Associates of San Antonio PA, San Antonio, Texas, USA
Department of Neurosurgery, University of Texas School of Law, Austin, Texas, USA
DOI:10.4103/2152-7806.157308
Copyright: © 2015 Branch BC. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.How to cite this article: Branch BC, Hilton DL, Watts C. Minimally invasive tubular access for posterior cervical foraminotomy. Surg Neurol Int 19-May-2015;6:81
How to cite this URL: Branch BC, Hilton DL, Watts C. Minimally invasive tubular access for posterior cervical foraminotomy. Surg Neurol Int 19-May-2015;6:81. Available from: http://surgicalneurologyint.com/surgicalint_articles/minimally-invasive-tubular-access-posterior-cervical/
Abstract
Background:Minimally invasive tubular access for posterior cervical foraminotomy can be an effective and safe technique for decompression of the nerve root utilizing minimally invasive muscle splitting with routine outpatient discharge. This technique has come under scrutiny calling into question the associated learning curve, a subjective limited exposure provided, and an argument that the risks and complications are largely unknown. In response to previously published critiques, this study aims to describe the outcomes and complications associated with this technique in a large patient series.
Methods:A retrospective chart review was performed from 1999 to 2013 capturing a single surgeon's experience with the minimally invasive tubular access for posterior cervical foraminotomy technique from a single institution, encompassing 463 patients. Surgical outcome documented at follow-up and complications were obtained from this patient series. Additional variables analyzed include: Hospital length of stay, number of levels operated, targeted root for decompression, side operated, length of surgery, and estimated blood loss.
Results:Outpatient discharge was achieved in 91.6% of cases. There were 10 complications (2.2%) among the 463 patients undergoing this technique from 1999 to 2013. Patients were followed for an average of 1 year and 2 months postoperatively. Improvement from the preoperative condition was observed in 98.2% of patients and excellent outcomes with patients reporting complete relief of symptoms with no or mild residual discomfort was seen in 92.2%.
Conclusions:Compared with open techniques, minimally invasive tubular access for posterior cervical foraminotomy demonstrates comparable, if not superior, complication rates, and patient outcomes.
Keywords: Cervical, complications, foraminotomy, minimally invasive surgery, outcomes, tubular retractor
INTRODUCTION
Cervical radicular root syndromes were first described in 1943 by Semmes and Murphey.[
As with any innovation, some have questioned the technique's utility, arguing that a limited exposure is provided, and raising concerns that the risks and complications are largely unknown.[
MATERIALS AND METHODS
A retrospective chart review was performed from 1999 to 2013 capturing a single surgeon's experience with the MITAC technique (DLH). All patients in this series underwent this technique as previously described [
CLINICAL EXPERIENCE/RESULTS
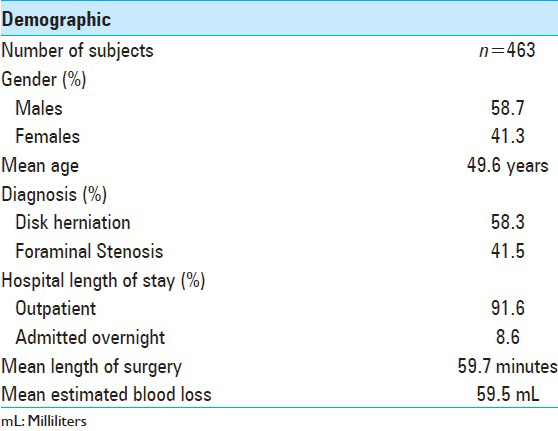
Demographics
The average age of these 463 patients at time of surgery was 49.6 years. There were 272 (58.7%) males and 191 (41.3%) females. Average length of surgery was 59.7 min and mean estimated blood loss was 59.5 cc. Only 37 patients (8.6%) required overnight hospital admission, and the remaining 395 patients (91.6%) were treated as outpatients [
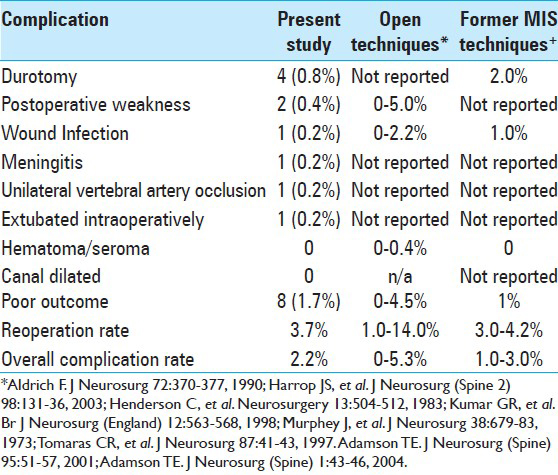
Complications
There were 10 complications (2.2%) among the 463 patients undergoing this technique from 1999 to 2013. Durotomy was the most frequent complication and occurred in four patients (0.8%). The durotomy was repaired by placing a small piece of DuraGen Plus™ Integra LifeSciences Corp. over the defect prior to closure. None of the four durotomies went on to develop a persistent cerebrospinal fluid (CSF) leak, wound break-down, or required additional surgery. There was one surgical wound infection (0.2%), which required incision and drainage followed by a course of antibiotics. There was one case of meningitis (0.2%) and after a thorough review of the patient record, we are unable to identify the cause, there was no durotomy during surgery, and no known preexisting infection. This patient was readmitted to the hospital and placed on a course of IV antibiotics and ultimately did well. While the CSF profile was suggestive of meningitis CSF cultures never grew an organism; however, the specimens were sent after the initiation of antibiotics. There was one case of unilateral vertebral artery occlusion (0.2%) early in the series; no obvious cause was identified upon review of the patient's record. One patient was extubated in the prone position during surgery; this was thought to be secondary to a poorly secured endotracheal tube. The tubular retractor was quickly removed and tegaderm was stapled over the incision allowing for the patient to quickly be positioned supine and reintubated while keeping the surgical field sterile. After the patient was reintubated the surgery was completed and the patient had no further complications. Two patients (0.4%) undergoing a two level ipsilateral decompression awoke with marked biceps weakness, which ultimately resolved completely within 2 months. Immediate postoperative imaging was performed on these two patients and did not identify an etiology for this weakness. Patients were treated with steroids and eventually had complete resolution of these symptoms. Both of these patients had ipsilateral two level decompressions of the C7 and C8 roots. There were no surgical site hematomas, wound dehiscence, death, and no instances of entering the cervical canal with the tubular retractor. None of the durotomies occurred as result of the dilating process, but rather were related to dural dissection during microdiskectomy with very tight neural components.
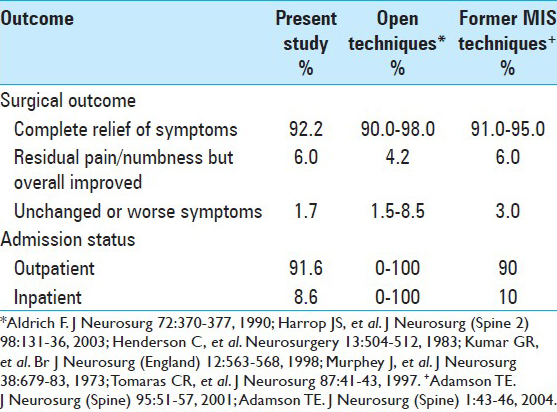
Clinical outcome/follow-up period
Follow-up data was available on 450 patients. Average follow up was 1 year and 2 months. A total of 415 (92.2%) patients were pleased with their result reporting complete relief of symptoms with no or mild residual discomfort. Twenty-seven (6.1%) said they had some degree of residual pain and/or numbness but were improved from the preoperative state. Eight patients (1.7%) reported unchanged or worse symptoms and seven of these patients eventually underwent anterior cervical fusion with satisfactory outcomes.
Secondary outcomes
Secondary outcomes were obtained on all patients undergoing this technique from 2008 to 2013, (n = 241). Of these 241 patients, 21 (8.7%) had two ipsilateral levels decompressed at the time of surgery, and 220 patients (91.3%) had a single level decompressed. There were no patients in which a bilateral decompression was attempted or performed. One hundred and forty patients (58.1%) underwent a left sided decompression while 101 patients (41.9%) underwent decompression on the right. The distribution of the cervical roots targeted for decompression in these 241 patients is presented in [
An ipsilateral two level decompression was performed in 21 of the 241 patients (8.7%). The mean age of patients undergoing a two level decompression was 54.2 years. A diagnosis of disk herniation was present in 2 patients (9.5%) and foraminal stenosis in 19 patients (90.5%). The average length of surgery was 82.1 min and the mean estimated blood loss was 81.4 cc. Decompression was performed on the left side in 8 patients (38.1%) and on the right in 14 patients (66.7%). The C6 and C7 roots were the most frequent roots targeted for decompression in 66.7% of patients.
Recurrent disk herniation at the same level of prior surgery occurred in 9 of the 241 patients (3.7%) with available electronic medical records from 2008 to 2013. Recurrent disk herniations were diagnosed and additional surgery was performed from 7 to 32 months after the initial decompression. Five recurrences (3.5%) occurred at C7, three (3.8%) at C6, and one (4.5%) at C8. In all nine patients with recurrent disk herniation at the same level of prior surgery, an anterior cervical diskectomy and fusion (ACDF) was the technique used upon revision. Two of the 241 patients (0.8%) went on to develop a de novo adjacent level disk herniation within 12–18 months after the initial surgery. These two patients underwent a minimally invasive tubular posterior cervical foraminotomy to address their adjacent level disk herniation.
DISCUSSION
The MITAC procedure was developed as an alternative to the minimally invasive endoscopic approach to the spine, permitting direct 3-D microscopic visualization of the operative field through a tube, several millimeters in diameter, which is used both as a dissector and a retractor. The patient is placed in the prone position, and intraoperative imaging for localization is carried out in the A-P plane of the spine. The A-P view for localization is one of the major advantages of the MITAC. It is carried out in the plane of the surgical procedure, which intuitively improves accuracy. The tubular retractor provides a path of light to enhance visualization. Although the tube is primarily a retractor, it can be used as a dissector, allowing the surgeon to visualize the entire operative field during dissection and retraction.
The approach is through a muscle splitting incision, which results in less blood loss during the opening phases of the surgery, and less pain during the postoperative period. When the procedure is over, the tube retractor is removed and the muscles return to their usual positions, avoiding the need for deep layered closure.
Some have raised a question about the learning curve in the acquisition of the skills necessary to perform this procedure. There are few relevant papers in the literature concerning morbidity and mortality and learning curves. It is for this reason we report our cases, to establish a large data base, perhaps the largest in the literature on this subject, which can be used by others as they seek various ways of presenting their outcomes.
The learning curve for this procedure is unique for each surgeon, considering the knowledge base each surgeon brings to each learning process. Neurosurgery residents and fellows leave training with an understanding of the gross and microscopic anatomy of the cervical spine and its nerves. Additionally, each has experience with the microscope intraoperatively, and with intraoperative imaging for localization. They have had experience with operating at depths, including through a “tube” in the transphenoidal approach to the pituitary gland. Some have used muscle splitting procedures in harvesting iliac bone. These and other skills can be honed in the surgical laboratory. General principles to a MITAC can be observed while assisting and, then, finally conducting the lumbar variant of this procedure before performing this technique on the cervical spine.
Although a purpose of this paper was not to provide a tutorial for the procedure, some of the key elements of the procedure will be stressed to provide the reader with a frame of reference for a discussion of complications.
A tubular exposure viewed under the microscope gives the surgeon a high resolution 3-D and magnified view of the medial facet and foramen targeted for decompression. This exposure provides a focus on the area of pathology negating the need for extraneous dissection for the purposes of maintaining an open exposure on a small area of pathology [
To evaluate the complication rates of MITAC we first review the published complication rates in open posterior cervical laminoforaminotomy. Reports on open cervical laminoforaminotomy describe complication rates ranging from 0% to 5.3%, and reoperation rates ranging from 1% to 14% [
Much of the data on the outcomes of the open technique were published in the 1980–1990s, with only small case series being published more recently. The few larger case series published more recently in the 1990s cite complication rates of 2.2–3.8% among series with 89–183 patients.[
There were 10 complications in our series of 463 patients undergoing this technique from 1999 to 2013 for an overall complication rate of 2.2%. This is well within the published complication rate ranges of the open techniques of 1.5–3.8% among the larger series.[
The four cases of durotomy reported in our series all occurred after the foraminotomy had been performed and during microdissection around the nerve root. Interestingly all the durotomies were located in the root axilla; no cases of central canal durotomy occurred. Frequently seen in these situations was an adherent piece of ligamentous or scar tissue in the root axilla, which we attempted to dissect free. Also of note, each durotomy was characterized by a small pin hole sized defect in the dura of the root axilla when viewed under the high powered surgical microscope. There were no situations in which we observed a contused thecal sac or large dural defect at any point during the procedure, and specifically upon completion of the foraminotomy exposure.
In our series of patients, 91.6% were performed on an outpatient basis and only 8.6% required inpatient admission. This compared with a majority of the open technique patients requiring inpatient admission and only one series of the open technique publishing the technique performed on an outpatient basis [
Extended data were available on 450 patients who were followed for an average of 1 year and 2 months. Eight patients (1.7%) had postoperative dysesthesias or radiculopathy where persistence or exacerbation of symptoms postoperatively is reported at 1.5–8.5%.[
Of interest, one small prospective randomized study comparing open cervical foraminotomy with minimally invasive tubular cervical foraminotomy has been reported in the literature.[
Of the secondary outcomes reported, a disk recurrence requiring additional surgery occurred in 9 of 241 patients (3.7%) in our series. Recurrent disk herniations requiring additional surgery have been reported at rates of 1.0–6.7% in the larger open technique series.[
Based on this comparison we would postulate that the minimally invasive tubular posterior cervical laminoforaminotomy technique has a risk/complication profile similar to the open technique when performed by surgeons experienced in tubular spine surgery and utilizing AP fluoroscopy for localization.
LIMITATION
This study, as with any of retrospective design, suffers primarily in that data are acquired before the appropriate study questions may be asked. This is also the principal limitation created by the change that occurred in the institutions electronic medical recordkeeping. However, this problem did not materially affect our goals of identifying complications and addressing the “learning curve.” Most neurosurgeons who have finished a program of neurosurgical education and training have the basic skills to be trained in this procedure.
Some might say the single surgeon experience is also limiting, if so, the limitation was minor. At the worst it created a constant and homogeneous data base.
CONCLUSIONS
We hope to raise awareness on the outcomes and complications of the minimally invasive tubular access for posterior cervical foraminotomy technique by presenting the largest published series to date on this technique. When compared with the complications and outcomes of the open techniques, we find surprisingly that the complication rates are within the range of those published for the open technique, and that a majority of cases are able to be performed on an outpatient basis with good patient outcomes. Our series and discussion highlight the MITAC technique providing evidence that this minimally invasive technique serves as an excellent tool for foraminal pathology. As previously stated, this technique should be reserved for surgeons experienced in tubular spine surgery who have already developed competency in the lumbar spine. We strongly advocate the utilization of AP fluoroscopy for localization in order to safely and successfully perform this technique.
The advantage of the minimally invasive tubular posterior cervical foraminotomy over traditional techniques is found in several facets. Foraminotomy as explained here does not destabilize the facet joint. Muscle splitting access provided by the tubular dilation system may reduce intraoperative bleeding, and postoperative pain. The tube, of course, is continuously curved. The retracted tissues are constantly subjected to the local forces of the curve. Even when used as a dissector there are no sharp edges along the shaft of the tube over which tissues can be left drapped over sharp edges, common with standard retractors. Upon removal of the tube from the wound, there is immediate closure of the wound and reconstitution of fascial layers, leaving no space for the accumulation of fluid. Using the surgical microscope and a tubular retractor, the surgeon is provided a high resolution exposure and working corridor. AP fluoroscopy is performed in the same plane as that of the surgery facilitating a simple yet safe and reliable means of localization and retractor insertion. As result of these characteristics this technique can adequately address the pathology, has a reasonable complication profile, and can be performed on an outpatient basis in a majority of patients.
Videos available at www.surgicalneurologyint.com
References
1. Adamson TE. Microendoscopic posterior cervical laminoforaminotomy for unilateral radiculopathy: Results of a new technique in 100 cases. J Neurosurg (Spine). 2001. 95: 51-7
2. Adamson TE. The impact of minimally invasive cervical spine surgery. J Neurosurg (Spine). 2004. 1: 43-6
3. Aldrich F. Posterolateral microdiscectomy for cervical monoradiculopathy caused by posterolateral soft cervical disc sequestration. J Neurosurg. 1990. 72: 370-7
4. Epstein NE. Minimally invasive/endoscopic vs “open” posterior cervical laminoforaminotomy: Do the risks outweigh the benefits?. Surg Neurol. 2009. 71: 330-1
5. Epstein NE. A review of laminoforaminotomy for the management of lateral and foraminal cervical disc herniations or spurs. Surg Neurol. 2002. 57: 226-33
6. Foley KT, Smith MM. Microendoscopic discectomy. Tech Neurosurg. 1997. 3: 301-7
7. Frykholm R. Cervical root compression resulting from disc degeneration and root sleeve fibrosis. Acta Chin Scand. 1951. 160: 1-149
8. Harrop JS, Silva MT, Sharan AD, Dante SJ, Simeone FA. Cervicothoracic radiculopathy treated using posterior cervical foraminotomy/discectomy. J Neurosurg (Spine 2). 2003. 98: S131-6
9. Henderson C, Hennessy RG, Shuey HM, Shackelford EG. Posterior-lateral foraminotomy as an exclusive operative technique for cervical radiculopathy: A review of 846 consecutively operated cases. Neurosurgery. 1983. 13: 504-12
10. Hilton DL. Minimally invasive tubular access for posterior cervical foraminotomy with three-dimensional microscopic visualization and localization with anterior/posterior imaging. Spine J. 2007. 7: 154-8
11. Kim K, Kim Y. Comparison between open procedure and tubular retractor assisted procedure for cervical radiculopathy: Results of a randomized controlled study. J Korean Med Sci. 2009. 24: 649-53
12. Kumar GR, Maurice-Williams RS, Bradford R. Cervical foraminotomy: An effective treatment for cervical spondylotic radiculopathy. Br J Neurosurg (England). 1998. 12: 563-8
13. Murphey J, Simmons JC, Brunson B. Surgical treatment of laterally ruptured cervical disc: review of 648 cases, 1939 to 1972. J Neurosurg. 1973. 38: 679-83
14. Scoville WB, Dohrmann GJ, Corkill G. Late results of cervical disc surgery. J Neurosurg. 1976. 45: 203-10
15. Semmes RE, Murphey F. Syndrome of unilateral rupture of the sixth cervical intervertebral disk, with compression of the seventh cervical nerve root.Report of four cases with symptoms simulating coronary disease. JAMA. 1943. 121: 1209-14
16. Spurling RG, Scoville WB. Lateral rupture of the cervical intervertebral disc.A common cause of shoulder and arm pain. Surg Gynecol Obstet. 1944. 798: 350-8
17. Tomaras CR, Blacklock CB, Parker WD, Harper RL. Outpatient surgical treatment of cervical radiculopathy. J Neurosurg. 1997. 87: 41-3
18. Zeidman SM, Ducker TB. Posterior cervical laminoforaminotomy for radiculopathy: Review of 172 cases. Neurosurgery. 1993. 33: 356-62