- Department of Neurosurgery, University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania, United States.
- Department of Neurosurgery, Medical School, Cooper University, Camden, United States.
- Department of General Surgery, Cooper University Hospital, Camden, United States.
- Department of Radiology, University of Pittsburgh, Pittsburgh, United States.
Correspondence Address:
Matthew Pease, Department of Neurosurgery, University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania, United States.
DOI:10.25259/SNI_38_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Matthew Pease1, Mazen Zaher2, Alejandro J. Lopez2, Siyuan Yu2, Tanya Egodage3, Suzan Semroc1, Dooman Arefan4, Brian Jankowitz2. Multicenter and prospective trial of anti-epileptics for early seizure prevention in mild traumatic brain injury with a positive computed tomography scan. 10-Jun-2022;13:241
How to cite this URL: Matthew Pease1, Mazen Zaher2, Alejandro J. Lopez2, Siyuan Yu2, Tanya Egodage3, Suzan Semroc1, Dooman Arefan4, Brian Jankowitz2. Multicenter and prospective trial of anti-epileptics for early seizure prevention in mild traumatic brain injury with a positive computed tomography scan. 10-Jun-2022;13:241. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=11650
Abstract
Background: Posttraumatic seizures (PTSs) are a major source of disability after traumatic brain injury (TBI). The Brain Trauma Foundation Guidelines recommend prophylactic anti-epileptics (AEDs) for early PTS in severe TBI, but high-quality evidence is lacking in mild TBI.
Methods: To determine the benefit of administering prophylactic AEDs, we performed a prospective and multicenter study evaluating consecutive patients who presented to a Level 1 trauma center from January 2017 to December 2020. We included all patients with mild TBI defined as Glasgow Coma Scale (GCS) 13–15 and a positive head computed tomography (CT). Patients were excluded for previous seizure history, current AED use, or a neurosurgical procedure. Patients were given a prophylactic 7-day course of AEDs on a week-on versus week-off basis and followed with in-person clinic visits, in-hospital evaluation, or a validated phone questionnaire.
Results: Four hundred and ninety patients were enrolled, 349 (71.2%) had follow-up, and 139 (39.8%) were given prophylactic AEDs. There was no difference between seizure rates for the prophylactic AED group (0.7%) and those without (2.9%; P = 0.25). Patients who had a PTS were on average older (81.4 years) than patients without a seizure (64.8 years; P = 0.02). Seizure rate increased linearly by age groups: 80 years old (4.6%).
Conclusion: Prophylactic AEDs did not provide a benefit for PTS reduction in mild TBI patients with a positive CT head scan.
Keywords: Anti-epileptic medications, Anti-seizure medicine, Mild traumatic brain injury, Prophylaxis, Seizures
INTRODUCTION
Every year, 42 million people worldwide suffer a mild traumatic brain injury (TBI).[
The benefits of prophylactic AEDs in mild TBI are unknown[
Although there is a lack of evidence for the use of AEDs, mild TBI patients may still benefit from prophylactic AED use. Seizures account for an large portion of emergency room visits and readmissions after TBI.[
To address this shortcoming, we performed a study to prospectively evaluate the incidence and effectiveness of prophylactic AEDs in mild TBI patients with a positive CT head scan in a prospective and multicenter setting.
MATERIALS AND METHODS
Data collection
Consecutive mild TBI patients older than 18 years old with acute intracranial blood were prospectively collected at two Level 1 trauma centers. Our observational study recruited patients from January 2017 to February 2019 at Mercy Hospital of the University of Pittsburgh Medical Center and from January to December 2020 at Cooper University Hospital. Patients were excluded from this study for a previous seizure history, current use of AEDs (i.e., mood stabilizer or for trigeminal neuralgia), or a neurosurgical procedure before or within 7 days of TBI. Both institutions received approval to complete this study as a quality improvement project.
The primary outcome was the effect of prophylactic AEDs on the rate of early PTS within 7 days of the trauma date. All trauma patients with a GCS 13–15 and intracranial blood on CT head scan received prophylactic AEDs on a week-by-week basis based on the day of admission. Patients who received AEDs at outside facilities before transfer were included in the cohort that received AED prophylaxis. Advanced practice providers, neurosurgery residents, and medical students collected the results but were blinded to the analysis completed by a statistician. Secondary outcomes included risk factors for seizure, overall incidence of early PTS, and a post hoc power analysis.
An inpatient seizure was defined as any witnessed seizure activity confirmed by the treating clinical team to be a seizure or a positive EEG, both of which were adjudicated by a neurologist blinded to the study. A positive EEG was defined as the neurologist reading the EEG stating an electrographic seizure occurred. Patients with seizures before arrival at the hospital were prescribed AEDs for therapeutic intent and excluded from this study.
Patient demographic, radiological, and follow-up information was collected by neurosurgery care providers. All patients were screened for alcoholism and an EtOH level was obtained in the emergency room. A neurosurgery resident physician or advanced care provider recorded a GCS for every patient within 24 h of admission. CT head scans were reviewed for the presence of subarachnoid hemorrhage (SAH), subdural hematoma (SDH), epidural hematoma (EDH), contusion, intraventricular hemorrhage (IVH), or skull fracture by a radiologist. Patients were either called or their hospital charts reviewed at 7-day posttrauma, depending on their admission status at the time. All patients were instructed to follow up 30 days after presentation. For patients who did not follow up in the outpatient office, three attempts were made to call the patient and speak with them. Patients were screened for seizures using a modification of a validated questionnaire.[
Statistical analysis
Statistical analysis was performed using R package (version 3.2.3), MATLAB version 2020b (MathWorks, Natick, MA), and G*Power (version 3.1.9.4) as needed. P-values were calculated using Fisher’s exact test, Chi-squared test, and Wilcoxon rank-sum test as appropriate. We performed a post hoc power analysis assuming two independent study groups and measured Cohen’s w effect size for a Chi-squared goodness-of-fit test.[
RESULTS
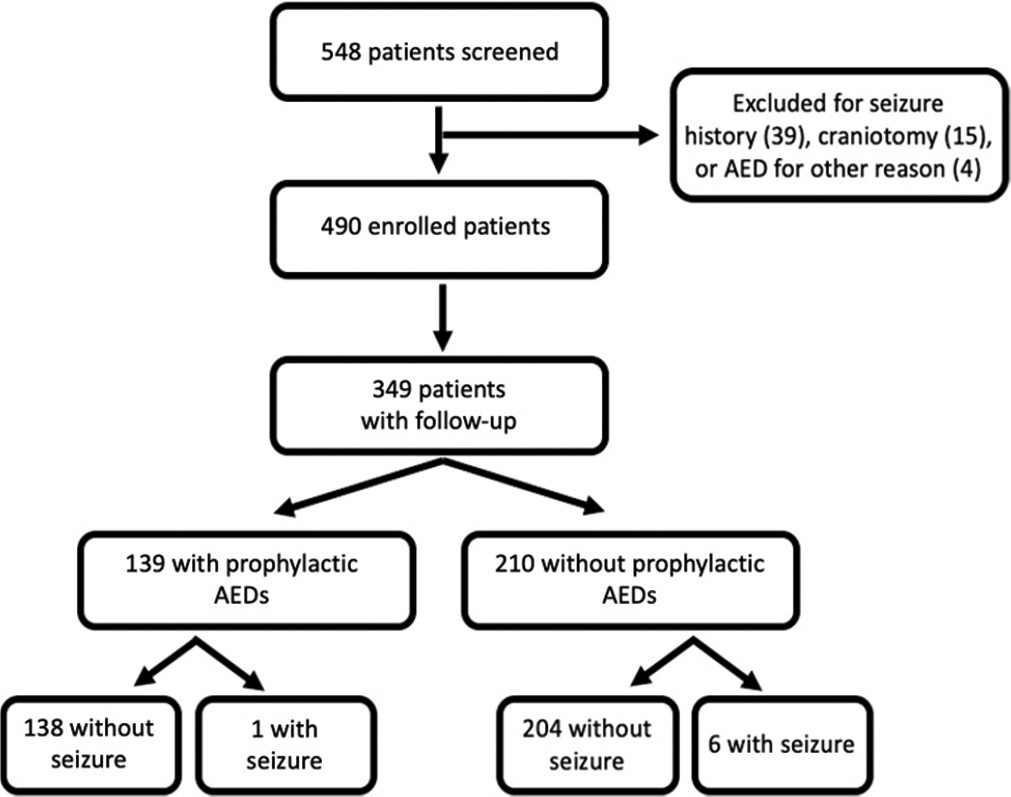
Five hundred and forty-eight patients who presented with a mild TBI and a positive CT head were entered into the study database. Patients were excluded for seizure history (39), craniotomy before or within 7 days of TBI (15), or AED use (4). Four hundred and ninety patients remained after exclusion [
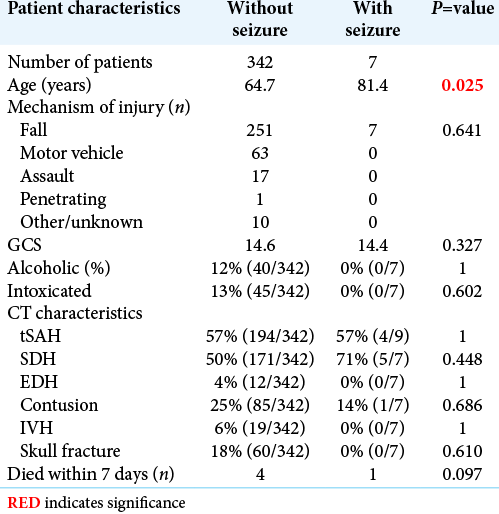
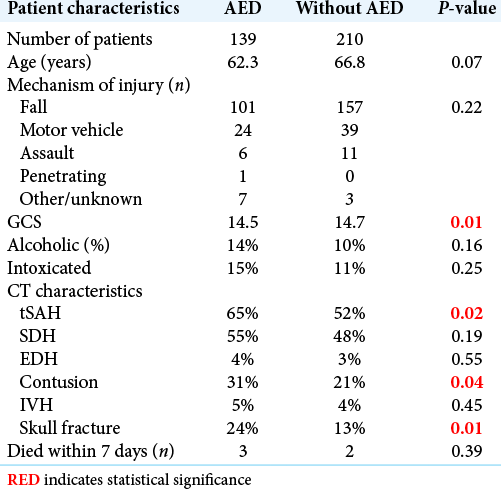
Among those with 7-day follow-up, demographic, clinical, and radiographic information is listed in [
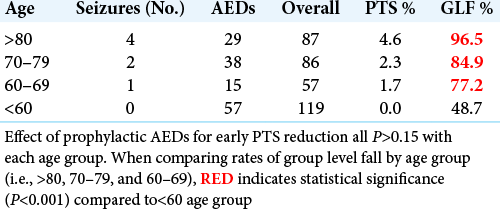
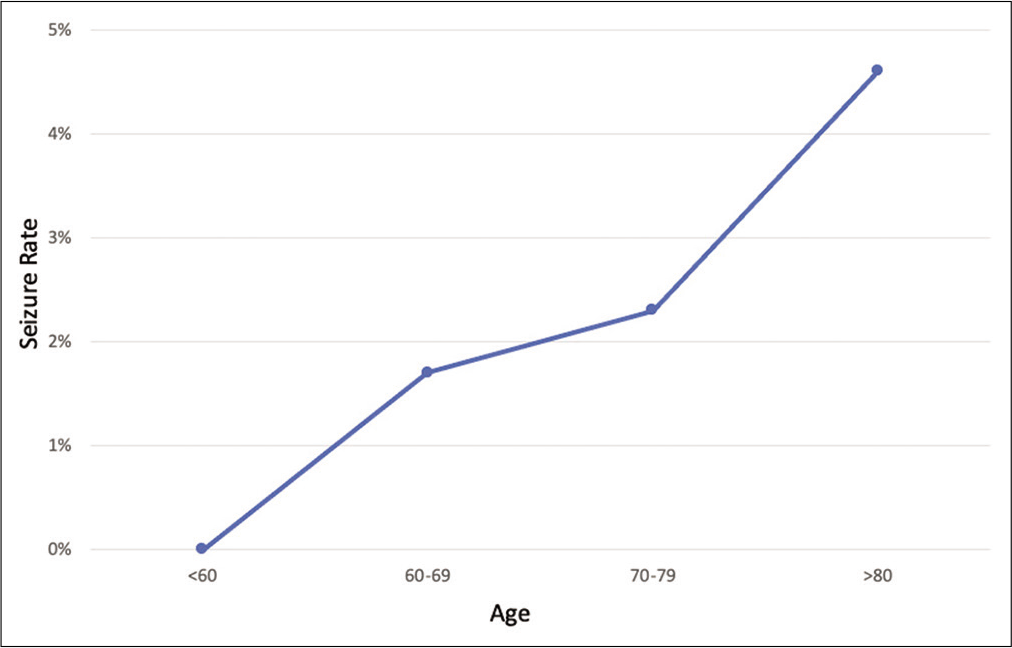
Patients who had a PTS were on average older (81.4 years) than patients without a seizure (64.7 years) regardless of AED use (P = 0.02). The seizure rate linearly increased by age groups: <60 years old (0%); 60–70 years old (1.7%); 70–80 years old (2.3%); and >80 years old [4.6%; Figure
Among patients >80 years old, the rate of seizures without prophylactic AEDs was 6.9% (0% for those with prophylactic AEDs). The effect of prophylactic AEDs was not significant within any age subgroup.
Although patients were assigned prophylactic AED use on a week-by-week basis, several cofounders were noted when stratifying by prophylactic AED groups. The rates of cerebral contusions (p=0.03) and skull fractures [P = 0.01;
DISCUSSION
A paucity of data exists in the literature describing the early PTS rates or effectiveness of prophylactic AEDs in mild TBI despite the large prevalence, societal impact, and cost. Neurosurgeons at many centers prescribe prophylactic AEDs in mild TBI[
The quality of data for seizure prophylaxis in mild TBI is low.[
We completed a multicenter and prospective trial evaluating the effects of prophylactic AEDs in mild TBI to address these shortcomings. Our study failed to find a benefit for prophylactic AEDs in reducing early PTS. We prospectively reported the incidence of early PTS at 2.0% overall, 2.9% without prophylactic AEDs, and 0.7% with prophylactic AEDs. With a NNT of 46.7, providers have a better understanding of the risk-benefit ratio for prophylactic AEDs in mild TBI.
Among our secondary outcomes, we found a significant difference in age between patients who had a seizure and those who did not have a seizure (P = 0.02). The seizure rate linearly increased when stratifying patients by age [
Among patients who had a seizure, all had a ground level fall as the mechanism of injury, a low impact mechanism associated with the elderly. Falls accounted for 96.5% of injuries in the >80 age group and decreased in younger patients. With an increasingly ageing population, our study highlights how minor injuries can have increased risks for negative consequences such as TBI and PTS. Effective methods to screen elderly patients for falls may reduce the risks of mild TBI in this patient population.
Our study has several weaknesses. Mainly, on a post hoc power analysis, we were underpowered to detect a benefit for prophylactic AEDs based on our effect size. Another weakness is our failure to identify radiographic features correlated with seizures such as brain contusions and skull fractures.[
CONCLUSION
In a multicenter and prospective study evaluating the effectiveness of prophylactic AEDs in early PTS for mild TBI patients, we were underpowered to find a benefit for prophylactic AEDs for early PTS rates. Patients who had seizures tended to be older, which can help guide personalized treatment decision.
Data availability
Data will be made available to qualified researchers on request.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
SUPPLEMENTARY TABLE
References
1. Armitage P, Berry G, Matthews JF.editors. Statistical Methods in Medical Research. New York: John Wiley and Sons; 2008. p.
2. Binder LM, Rohling ML, Larrabee GJ. A review of mild head trauma. Part I: Meta-analytic review of neuropsychological studies. J Clin Exp Neuropsychol. 1997. 19: 421-31
3. Cancelliere C, Kristman VL, Cassidy JD, Hincapié CA, Côté P, Boyle E. Systematic review of return to work after mild traumatic brain injury: Results of the international collaboration on mild traumatic brain injury prognosis. Arch Phys Med Rehabil. 2014. 95: S201-9
4. Candy N, Tsimiklis C, Poonnoose S, Trivedi R. The use of antiepileptic medication in early post traumatic seizure prophylaxis at a single institution. J Clin Neurosci. 2019. 69: 198-205
5. Carney N, Totten AM, O’Reilly C, Ullman JS, Hawryluk GW, Bell MJ.editors. Guidelines for the management of severe traumatic brain injury, fourth edition. Neurosurgery. 2017. 80: 6-15
6. Cifu DX, Eapen BC, Janak JC, Pugh MJ, Orman JA. Epidemiology of traumatic brain injury. Trauma Brain Inj Rehabil Med. 2015. 66: 6-35
7. Debenham S, Sabit B, Saluja R, Lamoureux J, Bajsarowicz P, Maleki M. A critical look at phenytoin use for early post-traumatic seizure prophylaxis. Can J Neurol Sci. 2011. 38: 896-901
8. Defrin R. Chronic post-traumatic headache: Clinical findings and possible mechanisms. J Man Manip Ther. 2014. 22: 36-43
9. Dimopoullos GT, Schrader GT, Fletcher BH. Electrophoretic studies of bovine serum. IV Differences in serum glycoproteins due to age and sex. Proc Soc Exp Biol Med. 1959. 102: 704-7
10. Fordington S, Manford M. A review of seizures and epilepsy following traumatic brain injury. J Neurol. 2020. 267: 3105-11
11. Gardner RC, Yaffe K. Epidemiology of mild traumatic brain injury and neurodegenerative disease. Mol Cell Neurosci. 2015. 66: 75-80
12. Guyatt GH.editors. Guyatt 2008 what is “Quality of Evidence” and why is it Important to Clinicians. 2008. p. 995-8
13. Hazama A, Ziechmann R, Arul M, Krishnamurthy S, Galgano M, Chin LS. The effect of keppra prophylaxis on the incidence of early onset, post-traumatic brain injury seizures. Cureus. 2018. 10: 2674
14. Inglet S, Baldwin M, Quinones AH, Majercik S, Collingridge DS, MacDonald J. Seizure prophylaxis in patients with traumatic brain injury: A single-center study. Cureus. 2016. 8: 6-8
15. Khor D, Wu J, Hong Q, Benjamin E, Xiao S, Inaba K. Early seizure prophylaxis in traumatic brain injuries revisited: A prospective observational study. World J Surg. 2018. 42: 1727-32
16. Looti AL, Kwon M, Bishu K, Ovbiagele B. Are seizures associated with increased odds of 30-day readmission after traumatic brain injury? Evidence from the national readmission database. Neurology. 2020. 94: 5143
17. Lowenstein DH. Epilepsy after head injury: An overview. Epilepsia. 2009. 50: 4-9
18. Ma CY, Xue YJ, Li M, Zhang Y, Li GZ. Sodium valproate for prevention of early posttraumatic seizures. Chin J Traumatol Engl Ed. 2010. 13: 293-6
19. Malison N. Antiepileptic for seizure prophylaxis in traumatic brain injury patients. BKK Med J. 2017. 13: 87
20. Ottman R, Barker-Cummings C, Leibson CL, Vasoli VM, Hauser WA, Buchhalter JR. Validation of a brief screening instrument for the ascertainment of epilepsy. Epilepsia. 2010. 51: 191-7
21. Temkin N, Dikmen S, Wilensky A, Keihm J, Chabal S, Winn HR. A randomized double-blind study of phenytoin for the prevention of post-traumatic seizures. N Engl J Med. 1990. 323: 497-502
22. Thompson K, Pohlmann-Eden B, Campbell LA, Abel H. Pharmacological treatments for preventing epilepsy following traumatic head injury. Cochrane Database Syst Rev. 2015. 2015: CD009900
23. Wat R, Mammi M, Paredes J, Haines J, Alasmari M, Liew A. The effectiveness of antiepileptic medications as prophylaxis of early seizure in patients with traumatic brain injury compared with placebo or no treatment: A systematic review and meta-analysis. World Neurosurg. 2019. 122: 433-40
24. Xu JC, Shen J, Shao WZ, Tang LJ, Sun YZ, Zhai XF. The safety and efficacy of levetiracetam versus phenytoin for seizure prophylaxis after traumatic brain injury: A systematic review and meta-analysis. Brain Inj. 2016. 30: 1054-61
25. Zangbar B, Khalil M, Gruessner A, Joseph B, Friese R, Kulvatunyou N. Levetiracetam prophylaxis for post-traumatic brain injury seizures is ineffective: A propensity score analysis. World J Surg. 2016. 40: 2667-72