- Department of Experimental Biomedicine and Clinical Neurosciences, School of Medicine, Postgraduate Residency Program in Neurological Surgery, Neurosurgical Clinic, “Paolo Giaccone”, Palermo, Italy
- Department of Biomedicine, Neurosciences and Advanced Diagnostics-BIND, Neuroradiology Unit, University of Palermo, Palermo, Italy.
Correspondence Address:
Roberta Costanzo, Department of Experimental Biomedicine and Clinical Neurosciences, School of Medicine, Postgraduate Residency Program in Neurological Surgery, Neurosurgical Clinic, AOUP “Paolo Giaccone”, Palermo, Italy.
DOI:10.25259/SNI_151_2024
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Vincenzo Gulino1, Roberta Costanzo1, Francesca Pedalino1, Giuseppe Salvaggio2, Cesare Gagliardo2, Domenico Gerardo Iacopino1, Rosario Maugeri1. Multiple Abdominal abscesses following ventriculoperitoneal shunt placement in an immunosuppressed patient: An illustrative case. 26-Apr-2024;15:142
How to cite this URL: Vincenzo Gulino1, Roberta Costanzo1, Francesca Pedalino1, Giuseppe Salvaggio2, Cesare Gagliardo2, Domenico Gerardo Iacopino1, Rosario Maugeri1. Multiple Abdominal abscesses following ventriculoperitoneal shunt placement in an immunosuppressed patient: An illustrative case. 26-Apr-2024;15:142. Available from: https://surgicalneurologyint.com/surgicalint-articles/12875/
Abstract
Background: Ventriculoperitoneal (VP) shunt placement is one of the most performed procedures in neurosurgery to treat various types of hydrocephalus (HC). Immediate or late postoperative complications may quite commonly occur, especially in immunosuppressed patients, who are predisposed to develop rare and difficult-to-treat conditions.
Case Description: Herein, we report the case of a 41-year-old female patient with a prior history of acute myeloid leukemia, followed by a tetra-ventricular acute HC due to a spontaneous non-aneurysmal subarachnoid hemorrhage. After an urgent external ventricular drainage placement, she underwent careful testing of “shunt dependency,” which ended with a VP shunt placement. After 2 months, she presented at the emergency department with worsening abdominal pain and fever. She underwent a computed tomography scan with contrast administration, which has shown abscesses in the abdominal cavity. An urgent surgical revision of the VP shunt and antibiotics administration followed this. After inflammatory markers normalization, due to the high risk of post-infective peritoneal adherence and consequent impairment of cerebrospinal fluid absorption, a ventriculoatrial shunt was considered the most appropriate solution.
Conclusion: Abdominal abscesses are a rare but subtle complication after VP shunt placement. Their management depends on etiology, patient clinical characteristics, and manifestations. Prompt interventions have been shown to improve clinical outcomes and optimize quality of life in such delicate patients.
Keywords: Abdomen, Abscess, Complication, Shunt, Ventriculoperitoneal
INTRODUCTION
Hydrocephalus (HC) is generally defined as a dynamic disorder between the production and absorption of cerebrospinal fluid (CSF), resulting in abnormal expansion of cerebral ventricles and frequently associated with increased intracranial pressure.[
The management of HC includes different approaches according to the obstructive/communicating dichotomy and characteristics of patients. Ventriculoperitoneal (VP) shunt placement is one of the most commonly performed procedures associated with various complications: infection, functional and/or mechanical impairment, or others.[
Herein, we present the case of an immunocompromised patient who has developed abdominal abscesses after VP shunt placement, followed by a revision of the literature.
CASE REPORT
We present the case of a 41-year-old woman affected by acute myeloid leukemia who, 1 month after chemotherapy, was admitted to the hematology unit to perform a routine medical check-up. There, she suddenly developed a tension-type headache, vomiting, and lethargy. An urgent brain computed tomography (CT) scan showed a non-aneurysmal subarachnoid hemorrhage [
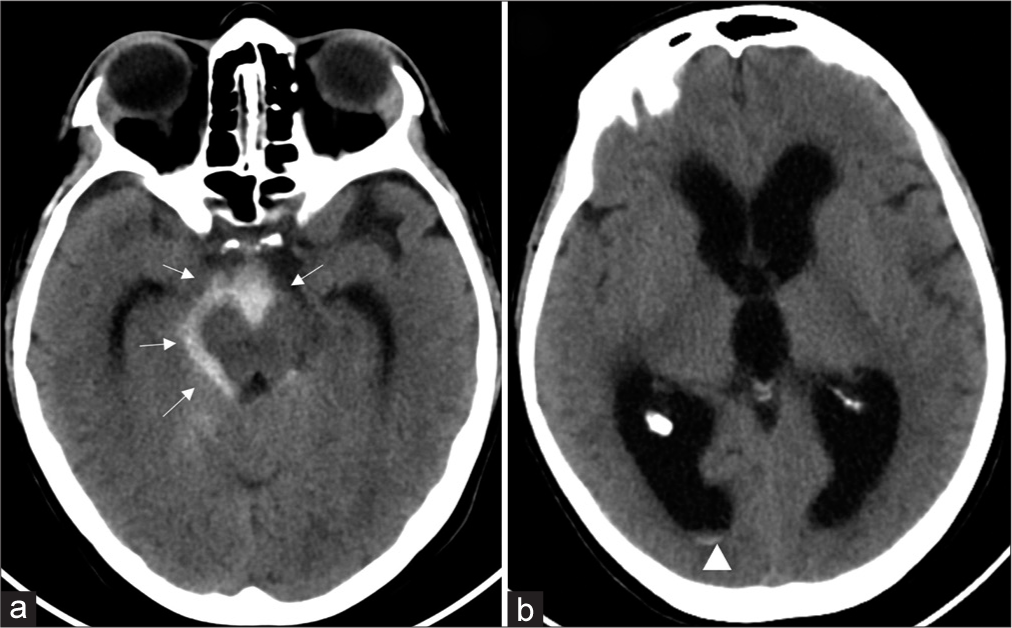
Figure 1:
(a) Axial computed tomography (CT) image shows subarachnoid hemorrhage in the peri-mesencephalic and interpeduncular cisterns (arrows). (b) Axial CT image of the same patient shows significant dilatation of the ventricular system consistent with hydrocephalus. Minimal intraventricular hemorrhage (arrowhead) is also noted layering in the occipital horns of the right lateral ventricles, which are significantly dilated.
Then, she urgently underwent an external ventricular drainage (EVD) placement. During the postoperative course, the patient improved with progressive neurological recovery, and a “shunt dependency” test was performed; after its failure, once CSF was clear and laboratory tests negative, the patient underwent VP shunt placement 1 month later and was transferred back to the Hematology Unit to continue proper therapy.
Two months later, she presented at the emergency department with a fever and severe abdominal pain. No increase in fluorosis indexes was detected at preliminary laboratory tests. A contrast-enhancement CT scan showed abscess collections and pseudocysts in the mesogastrium [
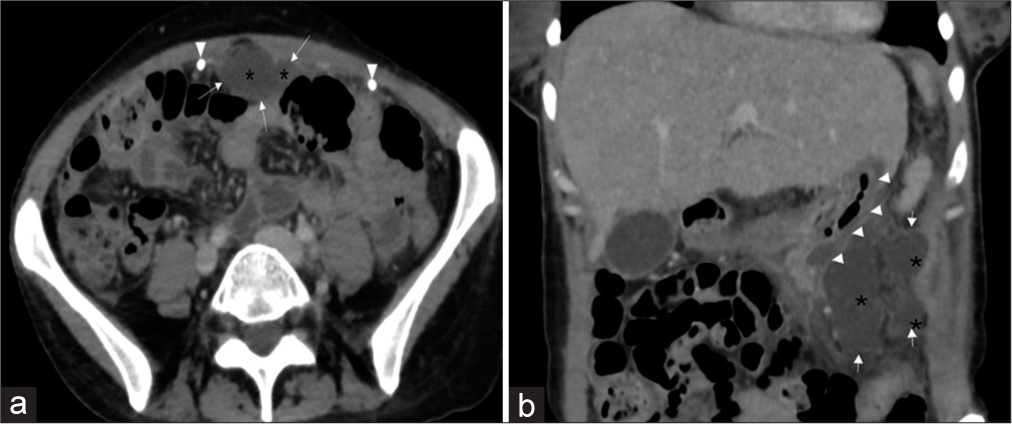
Figure 2:
(a) Axial contrast-enhanced computed tomography (CT) image shows homogeneous collections (asterisks) with thin contrast-enhancing walls (arrows) in the anterior peritoneal cavity. The ventriculoperitoneal shunt distal catheter is also visible (arrowheads). (b) Reformatted coronal contrast-enhanced CT image shows homogeneous collections (asterisks) with contrast-enhancing walls (arrows) adjacent to the gastric wall (arrowheads).
No further complications were detected, and a post-procedural clinical improvement was first noticed without any sign of fever or abdominal pain. Nevertheless, after a few days, the patient developed nausea, drowsiness, headache, and progressive neurological impairment (Glasgow Coma Scale 11- E 2, V4, M 5). She underwent an urgent CT scan, which showed a tetraventricular HC. Laboratory test parameters showed a sudden increase of inflammatory markers (white blood cells [WBC] 9 10^3/µL, C-reactive protein [CRP] 276 mg/L, and monocyte distribution width [MDW] 25.9). Then, she underwent urgent surgery for removal of the VP system, followed by Bactiseal© EVD placement.
Subsequently, blood culture investigations isolated Candida albicans and Enterococcus faecium. No bacterial growth was shown at the CSF investigation, despite the catheter showing a bacterial colonization (i.e., E. faecium) as well. An intravenous administration of linezolid 600 mg/2 times a day, rifampicin 600 mg/day, and itraconazole 100 mg/day was started and lasted for 4 weeks. Due to the failure to improve inflammatory markers, meropenem/vaborbactam 2 g/2 g 4 times a day was added, with a progressive clinical and neurological improvement. Antibiotic therapy lasted for 6 weeks, and EVD was replaced twice.
After symptoms recovery, a decrease of WBC, CRP, and procalcitonin due to “shunt dependency,” a new surgical procedure was mandatory. Due to the high risk of post-infective peritoneal adherence and consequent impairment of CSF absorption, a ventriculoatrial (VA) shunt was considered the best option. VA shunt was finally performed after a careful risk-benefit assessment.
The postoperative course was uneventful, without any complications or new onset of neurological deficit. Then, she was transferred to the hematologic unit to continue proper therapy. A 6-month follow-up has assessed a good recovery, with a significant improvement in the patient’s quality of life.
DISCUSSION
The use of the peritoneal cavity for CSF absorption in VP shunting is a procedure first introduced in 1908 by Kausch[
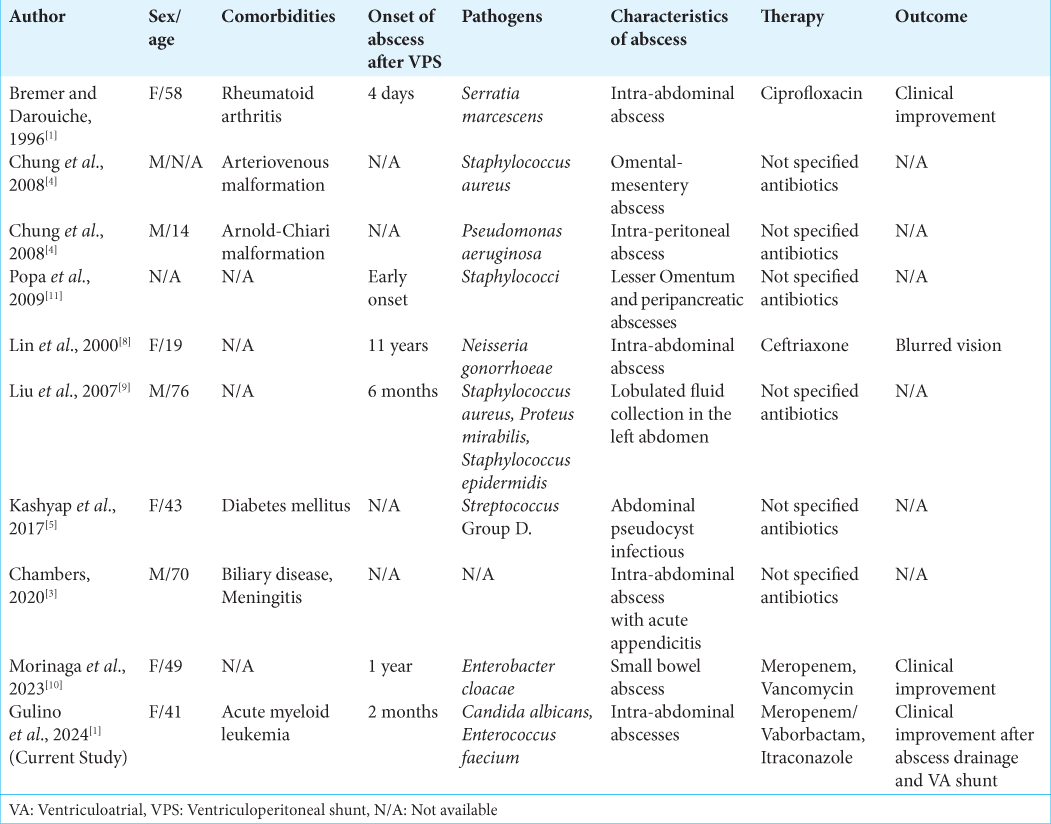
Indeed, according to our literature review, just nine cases of intra-abdominal abscess after VP-shunt placement are reported, as shown in
In three cases,[
Liu et al.[
Indeed, Lin et al.[
In another case described by Chambers,[
To the best of our knowledge, this is the first reported case describing abdominal complications caused by a yeast infection (i.e., C. albicans) in an immunosuppressed patient with a VP shunt.
Abdominal complications, such as abscesses or infections, can occur after VP shunt placement. VP shunt infection stands as a common cause of shunt failure, affecting approximately 8.1% of patients.[
Since the inherent difficulties related to their management, Kashyap et al.[
The accurate diagnosis of an abscess is typically achieved through an abdominal contrast CT scan,[
Treatment strategies heavily rely on the etiology, patient presentation, and clinical manifestations. At present, various treatment methods have been described, including CT-guided fluid aspiration (to reduce mass effect and identify pathogens), paracentesis, and laparotomy.[
CONCLUSION
Multiple abdominal abscesses with pseudocysts represent a rare but important complication of VP shunt. Clinicians other than neurosurgeons should be aware of this complication and keep it on the differential diagnosis for those who have a history of VP shunt placement presenting with acute abdomen symptoms. Neurological and clinical monitoring is crucial, especially for immunocompromised patients. Early and timely intervention has been shown to improve clinical and neurological outcomes and optimize the quality of life for such complicated patients.
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Bremer AA, Darouiche RO. Ventriculoperitoneal shunt infection due to Serratia marcescens. J Infect. 2005. 50: 138-41
2. Browd SR, Ragel BT, Gottfried ON, Kestle JR. Failure of cerebrospinal fluid shunts: Part I: Obstruction and mechanical failure. Pediatr Neurol. 2006. 34: 83-92
3. Chambers JG. Acute appendicitis with abscess in the setting of a ventriculoperitoneal shunt, requiring shunt explanation due to meningitis. Am Surg. 2020. 86: 1559-60
4. Chung JJ, Yu JS, Kim JH, Nam SJ, Kim MJ. Intraabdominal complications secondary to ventriculoperitoneal shunts: CT findings and review of the literature. AJR Am J Roentgenol. 2009. 193: 1311-7
5. Kashyap S, Ghanchi H, Minasian T, Dong F, Miulli D. Abdominal pseudocyst as a complication of ventriculoperitoneal shunt placement: Review of the literature and a proposed algorithm for treatment using 4 illustrative cases. Surg Neurol Int. 2017. 8: 78
6. Kausch W. Die Behandlung des hydrocephalus der kleinen kinder. Arch Kiln Chir. 1908. 87: 709-96
7. Knochel JQ, Koehler PR, Lee TG, Welch DM. Diagnosis of abdominal abscesses with computed tomography, ultrasound, and 111In leukocyte scans. Radiology. 1980. 137: 425-32
8. Lin MF, Lau YJ, Hu BS, Shi ZY, Lin YH. Gonococcal meningitis and intra-abdominal abscess in the presence of a ventriculoperitoneal shunt. Scand J Infect Dis. 2000. 32: 567-9
9. Liu KL, Lee TC, Lin MT, Chen SJ. Education and imaging. Gastrointestinal: Abdominal abscess associated with a ventriculoperitoneal shunt. J Gastroenterol Hepatol. 2007. 22: 757
10. Morinaga T, Nakahara O, Tsuji A, Kuramoto K, Iizaka M, Hayashida S. Laparoscopic intervention for late-onset perforating peritonitis due to a ventriculoperitoneal shunt: A case report and literature review. Surg Case Rep. 2023. 9: 154
11. Popa F, Grigorean VT, Onose G, Popescu M, Strambu V, Sandu AM. Laparoscopic treatment of abdominal complications following ventriculoperitoneal shunt. J Med Life. 2009. 2: 426-36
12. Sirinek KR. Diagnosis and treatment of intra-abdominal abscesses. Surg Infect (Larchmt). 2000. 1: 31-8
13. Taylor KJ, Wasson JF, De Graaff C, Rosenfield AT, Andriole VT. Accuracy of grey-scale ultrasound diagnosis of abdominal and pelvic abscesses in 220 patients. Lancet. 1978. 1: 83-4