- Department of Neurosurgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona,
- Department of Neurosurgery, Rutgers New Jersey Medical School, Newark, New Jersey, United States.
Correspondence Address:
Mark C. Preul, Department of Neurosurgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona, United States.
DOI:10.25259/SNI_1097_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Ali Tayebi Meybodi1, Allison S. Liang2, Pooneh Mokhtari2, Leandro Borba Moreira1, Xiaochun Zhao1, Michael T. Lawton1, Mark C. Preul1. Nervus intermedius: Microsurgical and anatomic relationships to the cerebellopontine angle neurovascular complex. 03-Feb-2023;14:37
How to cite this URL: Ali Tayebi Meybodi1, Allison S. Liang2, Pooneh Mokhtari2, Leandro Borba Moreira1, Xiaochun Zhao1, Michael T. Lawton1, Mark C. Preul1. Nervus intermedius: Microsurgical and anatomic relationships to the cerebellopontine angle neurovascular complex. 03-Feb-2023;14:37. Available from: https://surgicalneurologyint.com/surgicalint-articles/12143/
Abstract
Background: The nervus intermedius (NI) comprises fibers originating from the trigeminal, superior salivary, and solitary tract nuclei, which join the facial nerve (cranial nerve [CN] VII). Neighboring structures include the vestibulocochlear nerve (CN VIII), the anterior inferior cerebellar artery (AICA), and its branches. Microsurgical procedures at the cerebellopontine angle (CPA) benefit from understanding NI anatomy and relationships, especially for the microsurgical treatment of geniculate neuralgia, where the NI is transected. This study sought to characterize common relationships between the NI rootlets, CN VII, CN VIII, and the meatal loop of AICA at the internal auditory canal (IAC).
Methods: Seventeen cadaveric heads underwent retrosigmoid craniectomy. Following complete unroofing of the IAC, the NI rootlets were individually exposed to identify their origins and insertion points. The AICA and its meatal loop were traced to assess their relationship with the NI rootlets.
Results: Thirty-three NIs were identified. The median number of NI rootlets was 4 per NI (interquartile range, 3–5). The rootlets mainly originated from the proximal premeatal segment of CN VIII (81 of 141, 57%) and inserted onto CN VII at the IAC fundus (89 of 141, 63%). When crossing the acoustic-facial bundle, the AICA most frequently passed between the NI and CN VIII (14 of 33, 42%). Five composite patterns of neurovascular relationships were identified regarding NI.
Conclusion: Although certain anatomical trends can be identified, the NI has a variable relationship with the adjacent neurovascular complex at the IAC. Therefore, anatomical relationships should not be used as the sole method of NI identification during CPA surgery.
Keywords: Anterior inferior cerebellar artery, Geniculate neuralgia, Internal auditory canal, Nervus intermedius, Vestibular schwannoma
INTRODUCTION
The nervus intermedius (NI), first identified by Italian physician Bartolomeo Eustachi in 1563 and described in detail in 1777 by German anatomist Heinrich August Wrisberg,[
Microsurgical procedures at the cerebellopontine angle (CPA) may require identification of the NI for transection or microvascular decompression (MVD) for geniculate neuralgia (GN), and the NI also serves as an anatomical landmark for vestibular schwannoma resections.[
MATERIALS AND METHODS
This work did not involve patients; therefore, no institutional review board approval was required.
Cadaveric dissection
Thirty-three sides of 17 embalmed cadaveric heads were prepared for dissection. The arteries and veins were injected with red and blue silicone dye, respectively. The head was placed in a 3-pin head holder in the lateral position with 10° of head rotation to the contralateral side. A retrosigmoid craniectomy was performed. The dura was opened based on the lateral and sigmoid sinuses, and the arachnoid of the CPA was dissected open to explore the microanatomy of the acoustic-facial bundle (AFB) and associated vascular loop of AICA using a Pentero surgical microscope (Zeiss, Oberkochen, Germany). Next, the dura of the posterior aspect of the IAC was opened and incised in a semicircular fashion and reflected based on the posterior lip of the IAC [
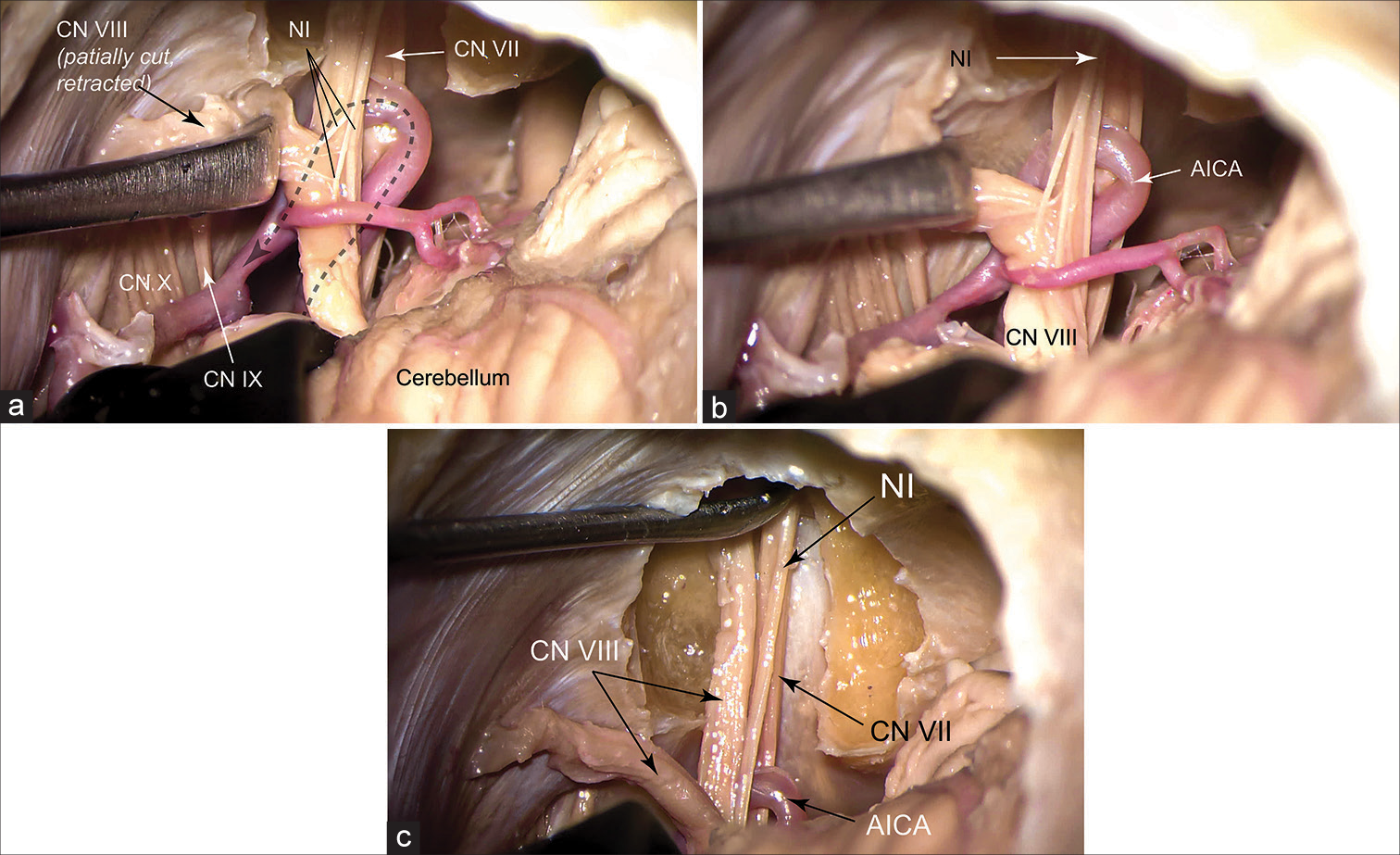
Figure 1:
Cadaveric exposure of the nervus intermedius (NI) after performing a left retrosigmoid craniectomy in lateral position and drilling the posterior wall of the internal auditory canal. (a) The vestibular nerves are cut and retracted to expose the NI. The dashed arrow shows the course of the anterior inferior cerebellar artery (AICA). The AICA passes between the NI and cranial nerve (CN) VII in this specimen. (b) Magnified view of (a) showing 4 rootlets composing the NI. (c) Intrameatal course of the NI is shown as it joins the facial nerve at the fundus. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Assessment of neurovascular relationships
The following anatomic variables were assessed and recorded: (1) origin and (2) insertion of the NI, (3) direction of the AICA approaching the AFB, (4) relationship of the meatal loop of the AICA to the IAC meatus, and (5) relationship between the AICA and NI. Origin of the NI was recorded as “brainstem,” “proximal premeatal,” “distal premeatal,” or “intrameatal.” Insertion of the NI onto CN VII was recorded as “proximal premeatal,” “distal premeatal,” “intrameatal,” or “fundal.” The AICA approached the AFB either “superiorly” or “inferiorly.” The meatal loop of the AICA was either “lateral” or “medial” to the IAC meatus. Finally, the composite neurovascular associations were studied to identify the common patterns.
Statistical analysis
Each variable was cross-tabulated based on the other five variables to generate six groups of distributions. The significance of the difference in distributions of variables in each group was assessed using the Chi-square test using SPSS (IBM, Armonk, New York). P < 0.05 was considered significant.
RESULTS
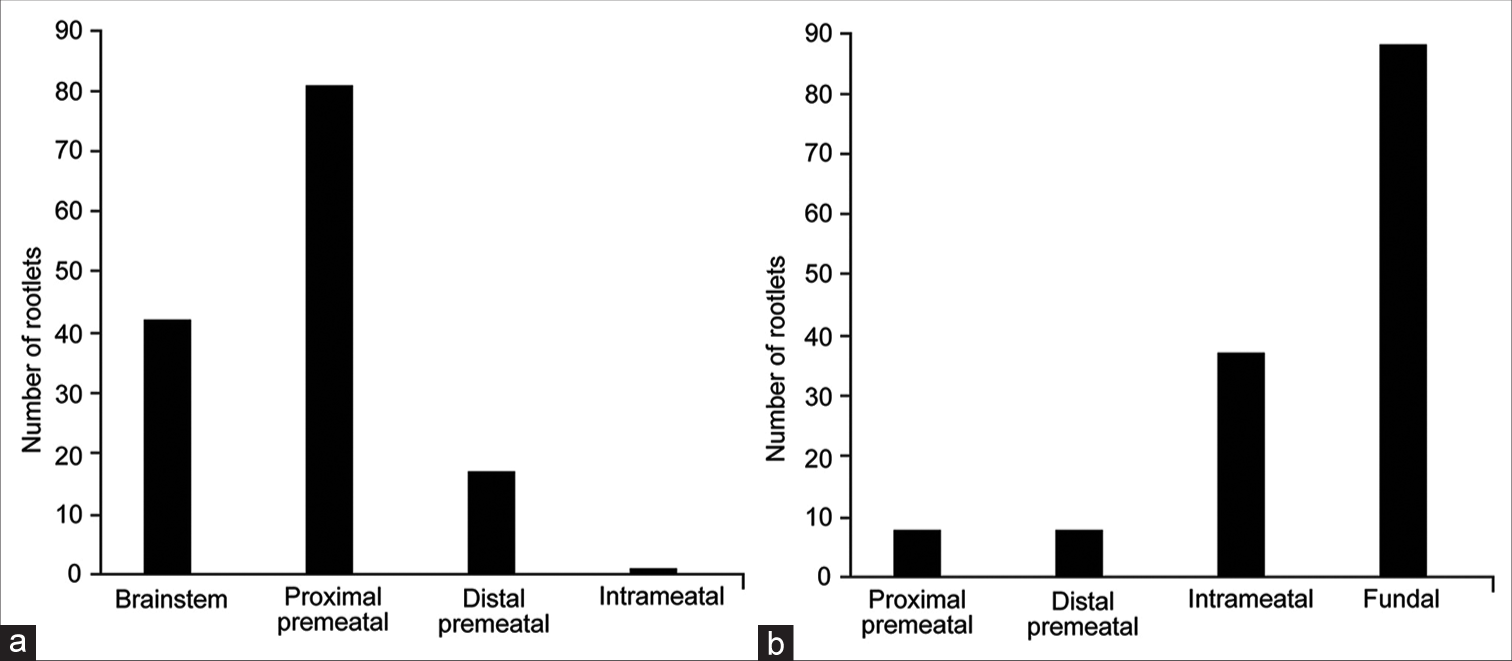
The nervous intermedius was identified in all 33 specimens. One specimen had a duplicate AICA from origin, making the total number of 34 AICAs. A total of 141 rootlets of the NI were identified in 33 CPAs. The median number of rootlets per specimen was 4 (range, 1–11). Most of the NI rootlets (57%, 81 of 141) originated from the proximal premeatal segment of CN VIII. Less commonly, NI rootlets also originated from the brainstem and the distal premeatal and intrameatal segments of CN VIII [
Analysis of NI relations to AFB and the meatal loop of AICA
AICA-AFB “passing-through” relationship
Origin of NI rootlets
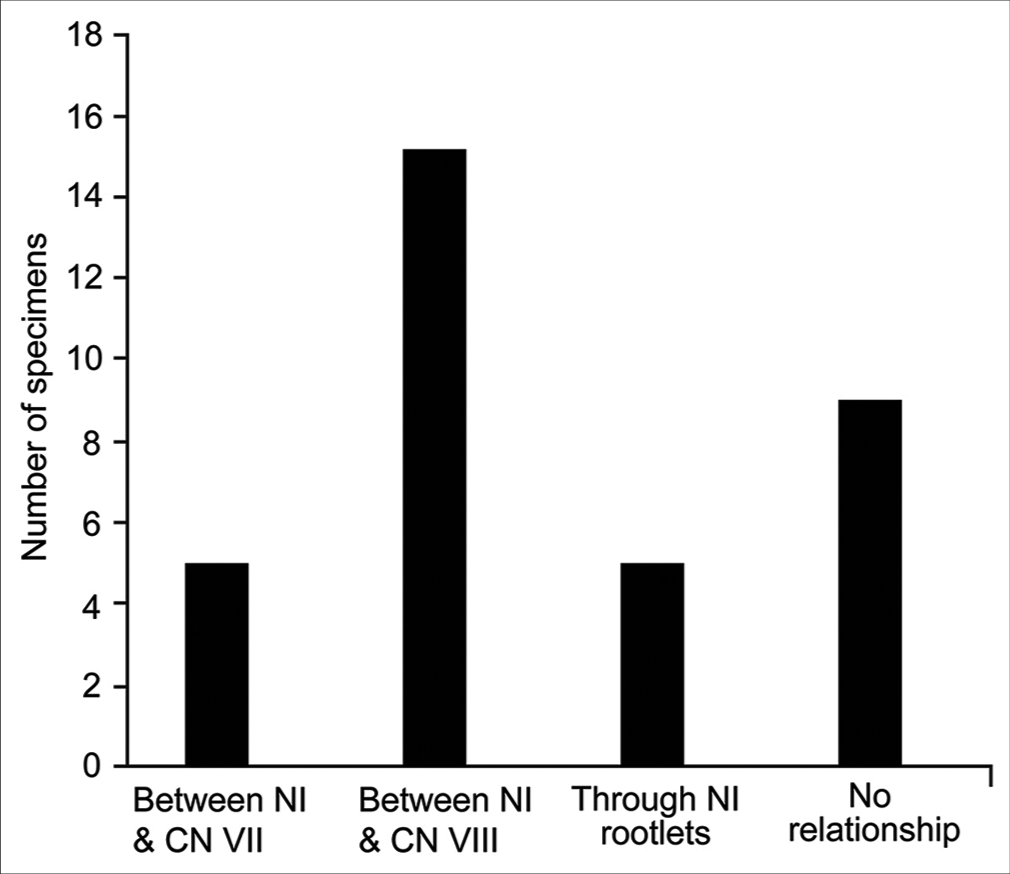
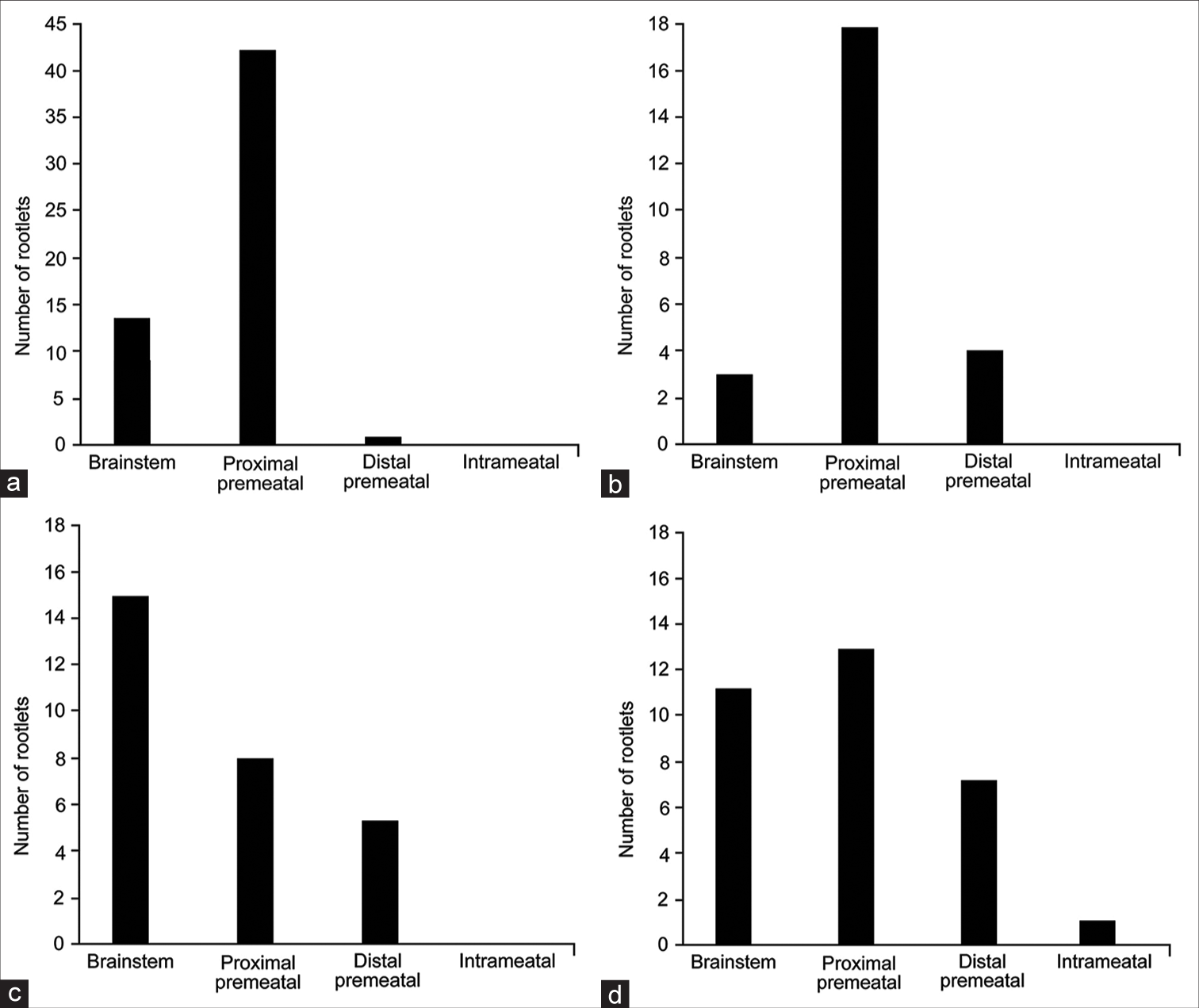
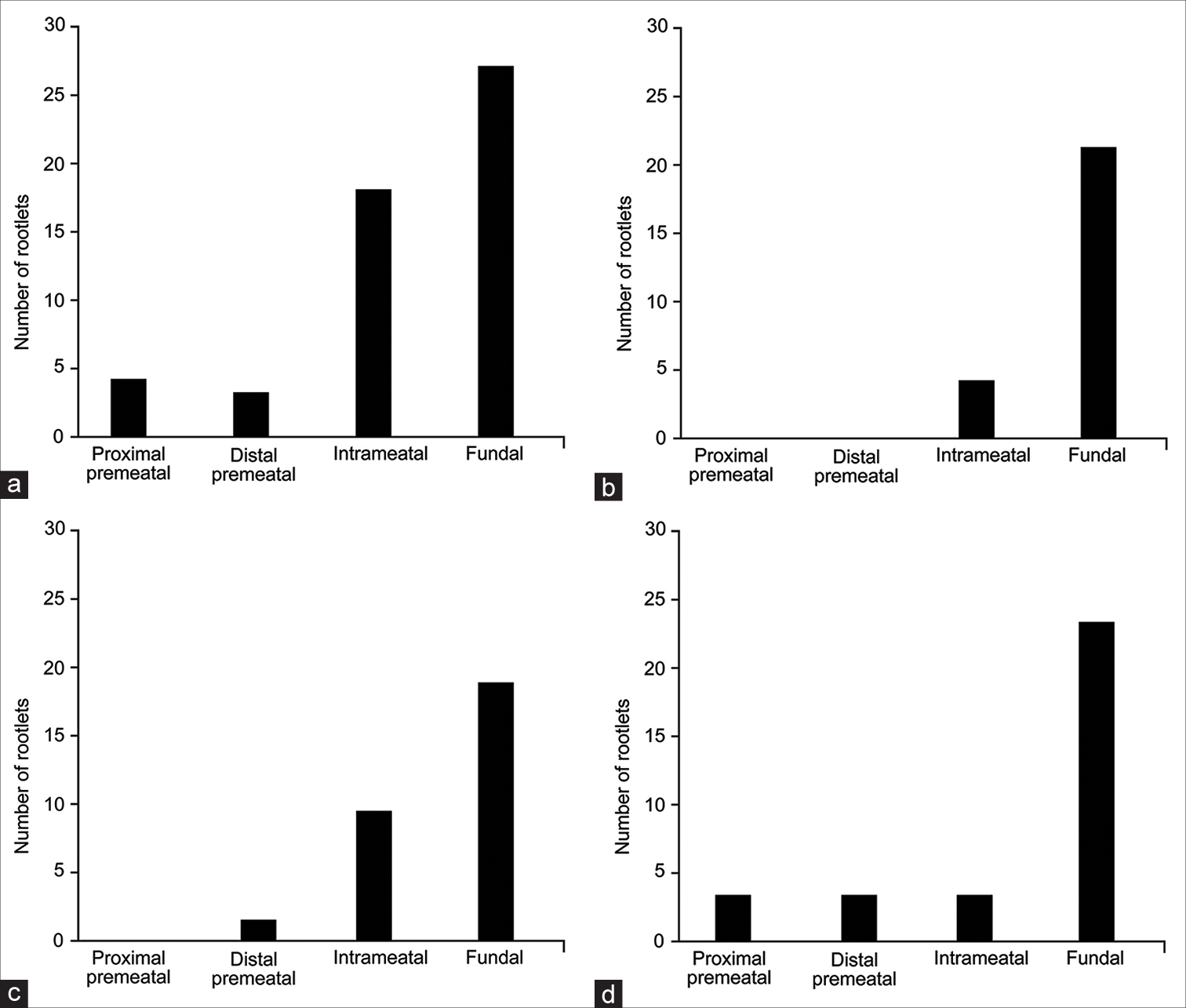
AICA passed through the AFB in 24 of 33 (73%) CPAs. As mentioned above, the most common configuration was AICA passing between NI and CN VIII (14 of 24, 58%). Chi-squared analysis revealed significant differences in the distribution of NI rootlet origins across different crossing patterns (χ2 (9, n = 141) = 30.5, P < 0.01). When the AICA passed between CN VIII and NI (n = 15) or between CN VII and NI (n = 5), the most common origin point of NI rootlets was the proximal premeatal segment of CN VIII (42 of 56 [75%] and 18 of 25 [72%], respectively) [
Figure 4:
Distributions of the nervus intermedius (NI) rootlet origins for each of the anterior inferior cerebellar artery (AICA)-NI relationships. (a) AICA coursing between the NI and cranial nerve (CN) VIII. (b) AICA coursing between the NI and CN VII. (c) AICA coursing through the rootlets of the NI. (d) AICA not coursing through the acoustic-facial bundle. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
However, most rootlets still originated from the proximal premeatal segment (41%, 13 of 32) [
Insertion of NI rootlets
In all “pass-through” patterns, the fundal CN VII was the most common insertion point of NI rootlets. This insertion point was most predominant in specimens in which the AICA passed between the NI and CN VII (accounting for 84% of rootlets [21 of 25]) and least predominant in specimens in which the AICA passed between the NI and CN VIII (accounting for 52% of rootlets [27 of 52]) [
Figure 5:
Distributions of the nervus intermedius (NI) rootlet insertions for each of the anterior inferior cerebellar artery (AICA)-NI relationships. (a) AICA coursing between the NI and cranial nerve (CN) VIII. (b) AICA coursing between the NI and CN VII. (c) AICA coursing through the rootlets of the NI. (d) AICA not coursing through the acoustic-facial bundle. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
AICA meatal loop and AFB approaching direction
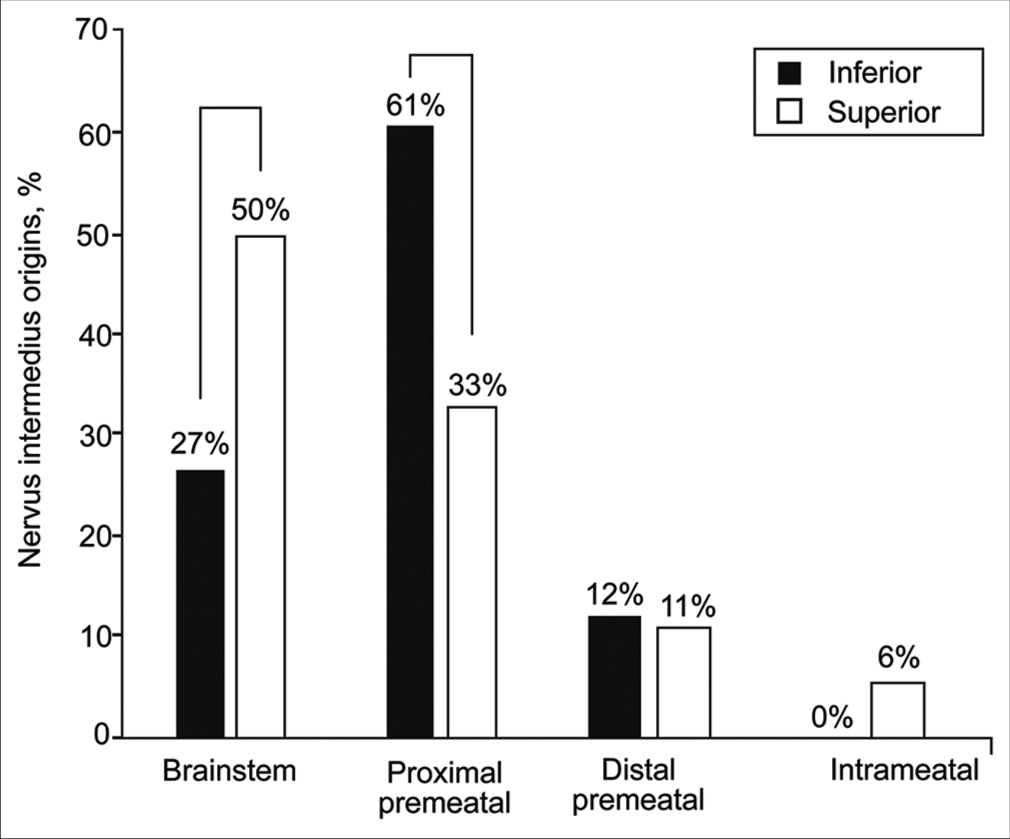
In all different AFB-AICA “passing through” relationships, the AICA approached the AFB more commonly from an inferior direction [
Direction of AICA approaching AFB
NI origin
Thirty of 34 AICAs approached the AFB from an inferior trajectory (88%). In these cases, 61% (75 of 123) of NI rootlets originated from the proximal premeatal segment, and 27% (33 of 123) originated from the brainstem. No NI rootlet originated from the intrameatal segment of CN VIII in these cases. When the AICA approached the AFB from a superior direction, more NI rootlets (9 of 18, 50%) originated from the brainstem than from the proximal premeatal segment of CN VIII (6 of 18, 33%), whereas a small number originated from the intrameatal segment of CN VIII (1 of 18, 6%). Significant variations in NI rootlet origin were observed between superior versus inferior AICA approaches to the AFB (χ2 (9, n = 141) = 19.4, P = 0.02).
Figure 6:
Relative distribution of nervus intermedius origins based on the anterior inferior cerebellar artery approach trajectory toward the acoustic-facial bundle (inferior vs. superior). Brackets show significant differences. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
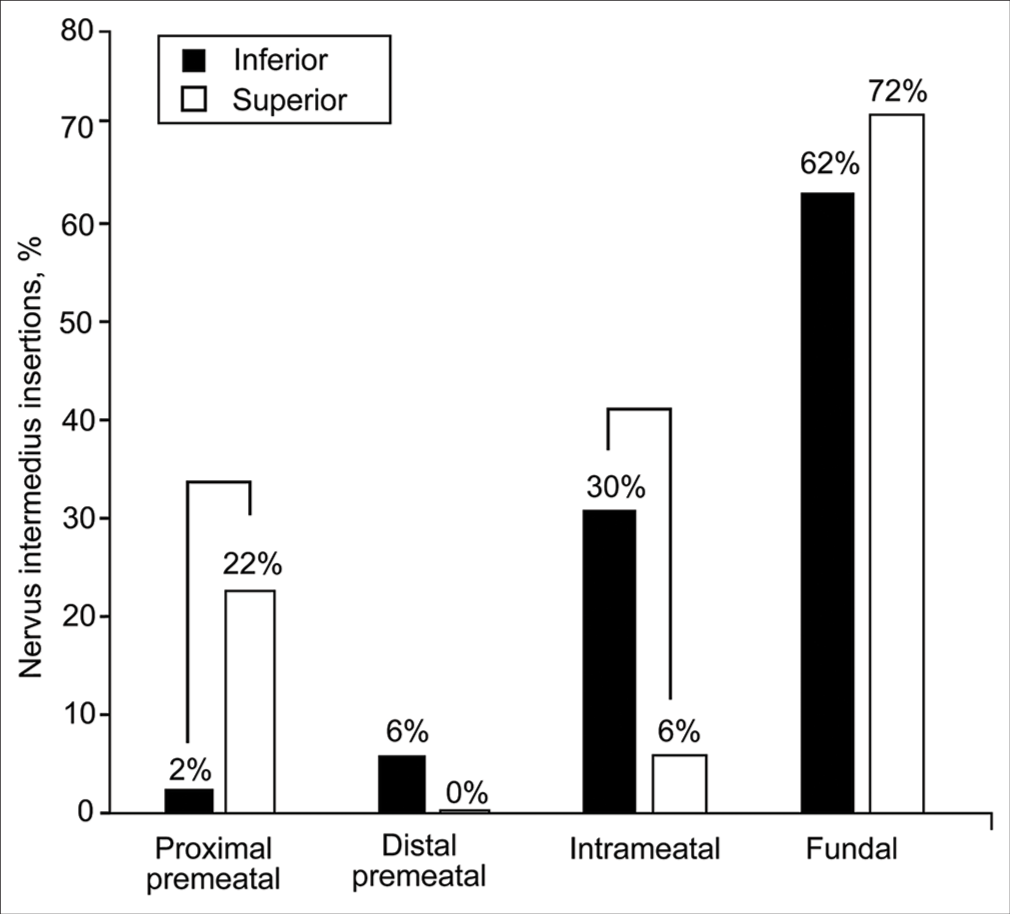
NI insertion
When the AICA approached the AFB from an inferior trajectory, the NI usually joined CN VII in its fundal (76 of 123, 62%) or intrameatal (37 of 123, 30%) segments. A significantly larger number of NI rootlets joined CN VII at its proximal premeatal segment when the AICA approached the AFB from a superior direction (4 of 18, 22%) compared with an inferior direction (3 of 123, 2%) (χ2 (3, n = 141) = 17.2, P = 0.002) [
Figure 7:
Relative distribution of nervus intermedius insertions based on the anterior inferior cerebellar artery approach trajectory toward the acoustic-facial bundle (inferior vs. superior). Brackets show significant differences. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Meatal loop to meatus relationship
Almost half (53%) of the meatal loops were located medial to the IAC.
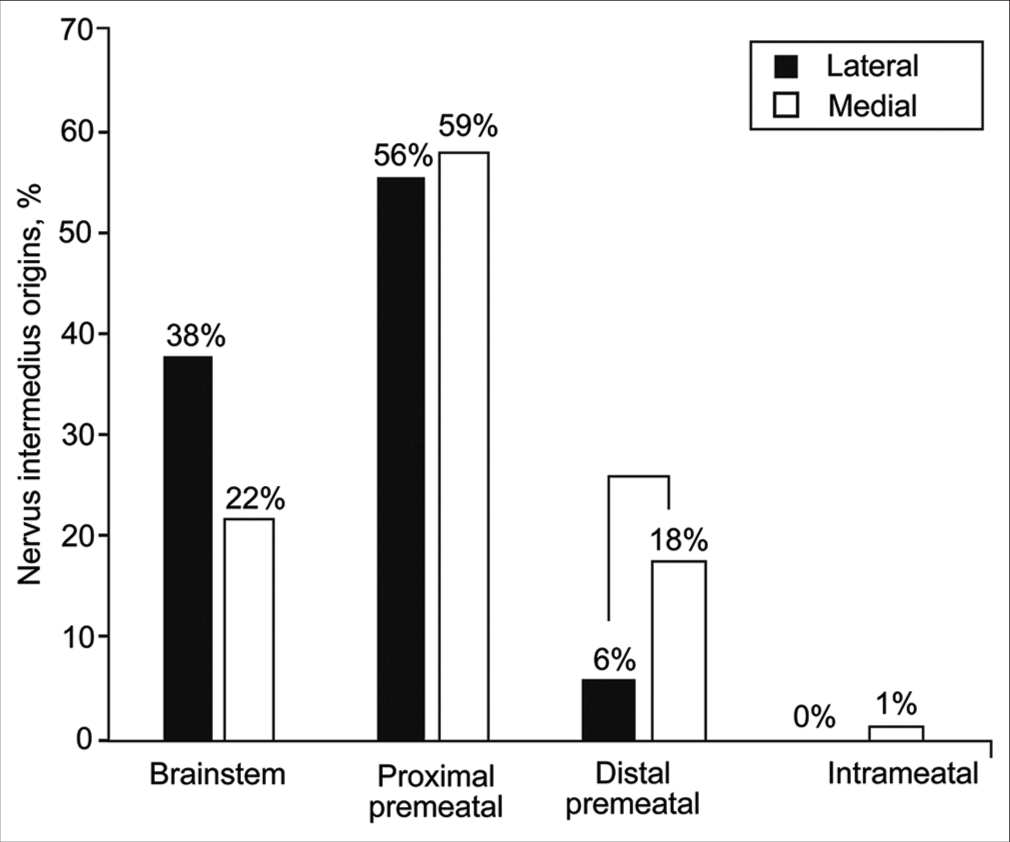
NI origin
NI rootlets most commonly originated from the proximal premeatal segment of CN VIII regardless of the relationship of the AICA meatal loop to the IAC (43 of 73 [59%] when medial and 38 of 68 [56%] when lateral). However, when the meatal loop was medial to the IAC, a significantly higher number of rootlets originated in the distal premeatal segment (18% [13 of 73] vs. 6% [4 of 68]) [
Figure 8:
Relative distribution of nervus intermedius origins based on the relationship between the anterior inferior cerebellar artery meatal loop and the internal auditory canal meatus (lateral vs. medial). Brackets show significant differences. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
NI insertion
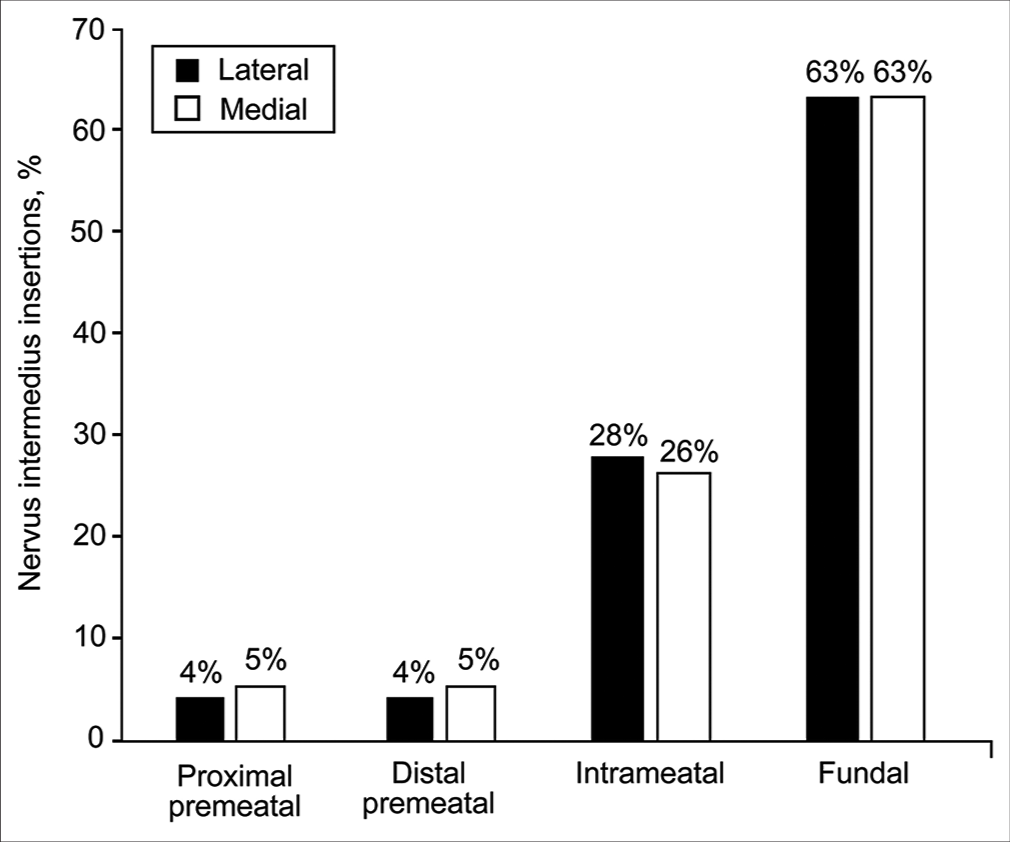
Patterns of NI rootlet insertion onto CN VII between the 2 meatal loop configurations did not differ in their relationship to the IAC. In addition, 63% of rootlets (89 of 141) inserted onto CN VII at its fundal portion regardless of whether the meatal loop was medial or lateral to the IAM (43 of 68 [63%] when lateral and 46 of 73 [63%] when medial) [
Figure 9:
Relative distribution of nervus intermedius insertions based on the relationship between the anterior inferior cerebellar artery meatal loop and the internal auditory canal meatus (lateral vs. medial). No significant differences were found. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Composite neurovascular patterns
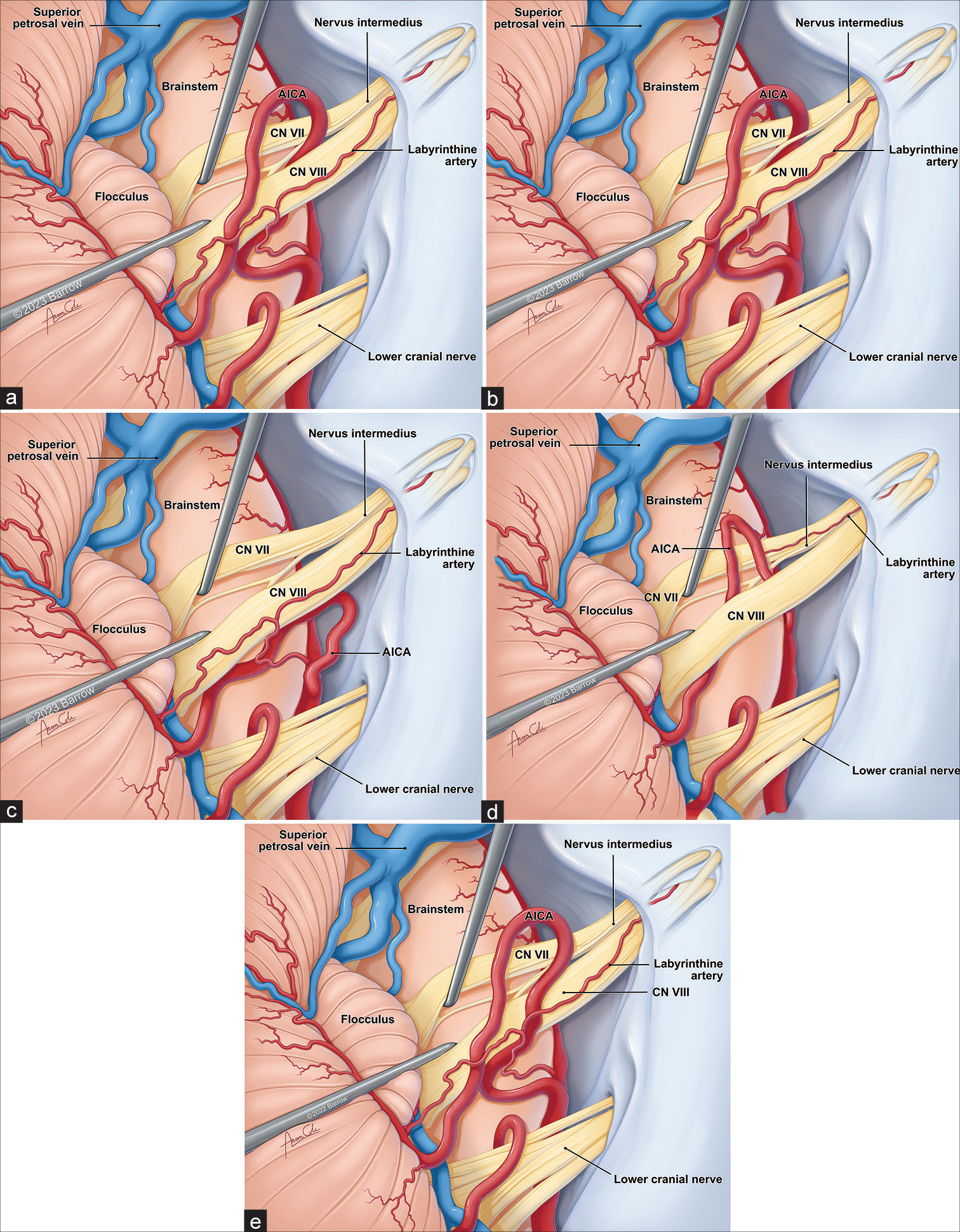
Besides the individual neurovascular associations, the following composite patterns were studied. Because the most common points of origin and insertion of the NI rootlets were brainstem/proximal premeatal and distal premeatal/ fundal, these parameters were not included in composite pattern recognition. Similarly, the meatal loop relationship to the IAC meatus was not significantly associated with any pattern. However, the following parameters were included for this analysis: (1) direction of the AICA approach to the AFB, (2) relationship of the AICA meatal loop and AFB, and (3) relationship between the AICA (or its branches) and the NI. Based on the above parameters, the following composite association patterns were found: (1) pattern 1, in which the AICA approaches the AFB from a superior or inferior direction, with the AICA meatal loop passing through the AFB between the NI and CN VIII (15 of 34, 44%); (2) pattern 2, in which the AICA approaches the AFB from a superior or inferior direction, with the AICA meatal loop passing through the AFB between the NI and CN VII (5 of 34, 15%); (3) pattern 3, in which the AICA approaches the AFB from an inferior direction, with the meatal loop being inferior to the AFB and not passing through the AFB or having a post bifurcation AICA branch passing through the NI rootlets (5 of 34, 15%); (4) pattern 4, in which the AICA approaches the AFB from an inferior direction, with the AICA meatal loop passing through the AFB between the NI rootlets (4 of 34, 12%); and (5) pattern 5 (miscellaneous), in which the AICA approaches the AFB from a superior or inferior direction with the AICA meatal loop being anterior or superior to the AFB and not passing through the AFB (5 of 34, 15%).
Figure 10:
Artist’s illustration of the most common composite patterns of neurovascular relationships in the cerebellopontine angle regarding the nervus intermedius (NI). (a) In pattern 1, the AICA approaches the acoustic-facial bundle (AFB) from a superior or inferior direction, with the AICA meatal loop passing through the AFB between the NI and CN VIII (44%). (b) In pattern 2, the AICA approaches the AFB from a superior or inferior direction, with the AICA meatal loop passing through the AFB between the NI and CN VII (15%). (c) In pattern 3, the AICA approaches the AFB from an inferior direction, with the meatal loop being inferior to the AFB and not passing through the AFB or having a post bifurcation AICA branch passing through the NI rootlets (5%). (d) In pattern 4, the AICA approaches the AFB from an inferior direction, with the AICA meatal loop passing through the AFB between the NI rootlets (12%). (e) In pattern 5 (miscellaneous), the AICA approaches the AFB from a superior or inferior direction with the AICA meatal loop being anterior or superior to the AFB and not passing through the AFB (15%). AICA: Anterior inferior cerebellar artery, CN: Cranial nerve. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
DISCUSSION
In this study, we present a detailed microanatomy of the NI and its relationship with the AICA-AFB neurovascular complex. We found that the most common configuration of NI consists of the nerve first originating from the proximal premeatal segment of CN VIII and joining CN VII at the IAC fundus, with the AICA passing between the NI and CN VIII. We also categorized the most common composite patterns of interrelations between AICA, NI, and AFB. These microanatomical details may be useful for the CPA surgeon, especially when the identification of the NI is critical, such as during the microsurgical treatment of the GN. GN, described by Hunt in 1937,[
Microanatomical highlights
NI rootlets
Earlier studies have shown that the NI usually comprises 2–5 rootlets, which is similar to our finding of a median of 4 rootlets (interquartile range, 3–5).[
AICA and NI
As the AICA leaves the basilar artery to course laterally toward the AFB, it may approach the AFB from an inferior (most common) or a superior trajectory. When the AICA takes its usual approach inferiorly toward the AFB, the origin and insertion of the NI rootlets were as one would usually expect — proximal premeatal and fundal, respectively. However, when the AICA approached the AFB from a superior trajectory (4 of 34, 12%), the most common point of origin of the NI was the brainstem. Interestingly, with this AICA-AFB relationship, a significant number (4 of 18, 22%) of insertion points onto CN VIII was at the premeatal segment.
Twenty five of 34 AICAs (73%) crossed through the AFB complex. This pattern is important because, in these cases, the AICA most commonly coursed between the NI and CN VIII. However, in a significant number of specimens, it crossed the AFB, passing between CN VII and the NI or through the NI rootlets. Therefore, the surgeon should understand these variable relationships and recognize their patterns [
A notable exception was found regarding the relationship between the meatal loop of the AICA and the IAC. When the AICA meatal loop was medial to the IAC, a higher proportion of the NI rootlets originated from the distal premeatal segment of CN VIII (18% vs. 6%) and fewer rootlets originated from the brainstem (22% vs. 38%), both of which differences were statistically significant (P < 0.05) [
Applied microsurgical anatomy
From an operative perspective, our study shows that the microanatomy of the NI is extremely variable. However, certain common patterns could be identified and used as a guide to help identify the NI during surgery [
Overall, the key relationships between the NI and AICA identified in this study further contribute to the recently growing body of literature elucidating CPA anatomy. The surgeon should be vigilant of the NI origin and insertion variants and look to the AICA to aid with the identification of the NI. Understanding specific AICA and meatal loop orientations associated with specific NI rootlet origins improves preoperative imaging assessment and intraoperative decision-making. Ultimately, the goal is to increase the precision and safety of CPA microsurgery and decrease NI-related complications.
CONCLUSION
Variations in NI rootlet origins appear with specific orientations of the meatal loop of the AICA at the IAC. This study illustrates expected NI-AICA relationships that surgeons can anticipate in preoperative image assessments and intraoperative exploration of the CPA. It is important to note that the origin and insertion points of the NI remain unpredictable. Consideration of the AICA path helps with such clarification, but anatomic data are insufficient for this purpose. Preoperative high-definition imaging and intraoperative physiology are invaluable adjuncts to the microanatomical patterns presented in the current study.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
This study was funded in part by funds from the Newsome Chair in Neurosurgery Research held by Dr. Preul and the Barrow Neurological Foundation.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
SUPPLIMENTAL FIGURES
Supplemental Figure 1:
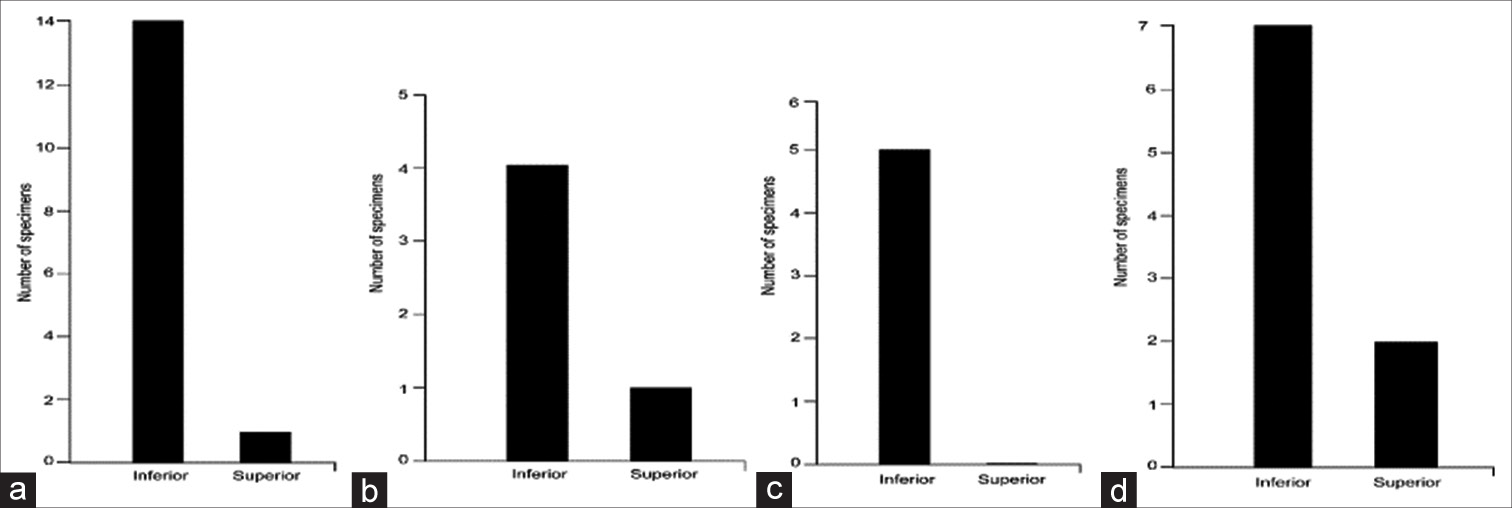
Distribution of the directions of approach of the anterior inferior cerebellar artery (AICA) toward the acoustic-facial bundle (AFB) showing the relationships of the AICA with the AFB roots. (a) Between the nervus intermedius (NI) and cranial nerve (CN) VIII (n = 15). (b) Between the NI and CN VII (n = 5). (c) Through the NI rootlets (n = 5). (d) No relationship (n = 9). Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
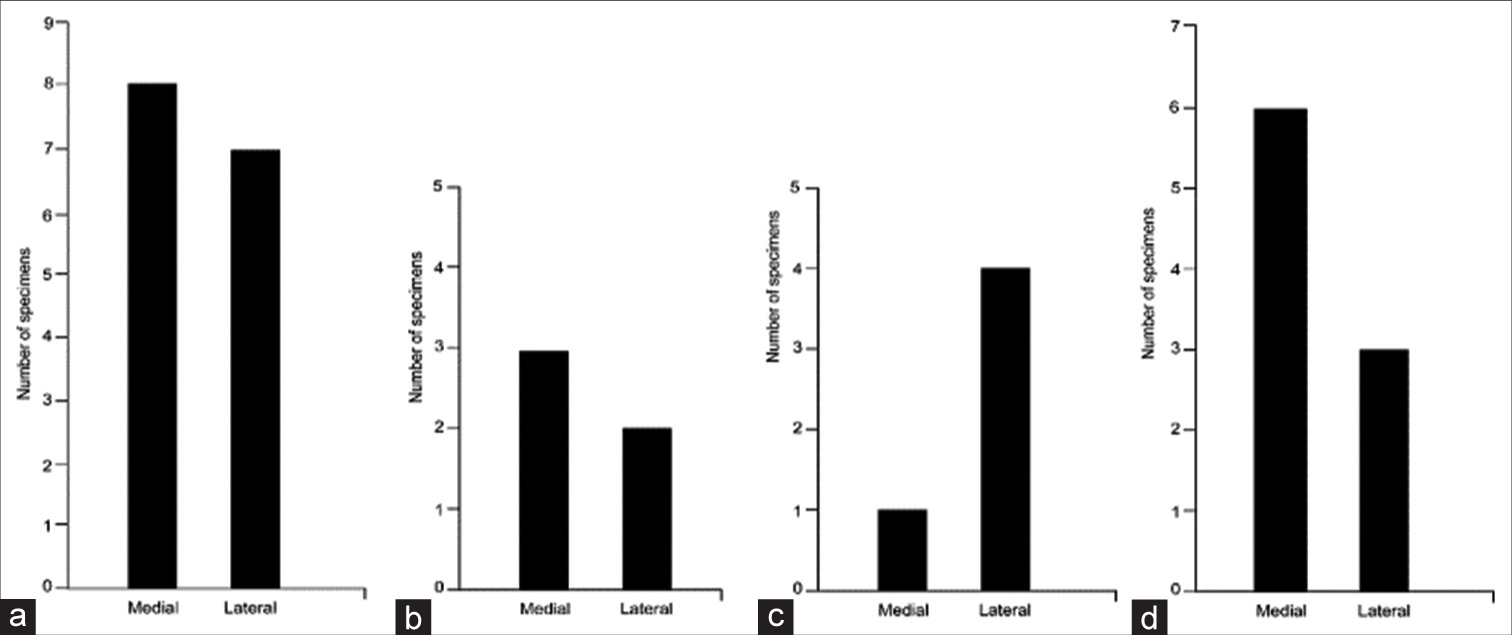
Supplemental Figure 2:
Distribution of the relationships between the anterior inferior cerebellar artery (AICA) meatal loop and the meatus of the internal auditory canal (IAC) showing the relationships of the AICA with the acoustic-facial bundle roots. (a) Between the nervus intermedius (NI) and cranial nerve (CN) VIII (n = 15). (b) Between the NI and CN VII (n = 5). (c) Through the NI rootlets (n = 5). (d) No relationship (n = 9). Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
References
1. Alfieri A, Fleischhammer J, Peschke E, Strauss C. The nervus intermedius as a variable landmark and critical structure in cerebellopontine angle surgery: An anatomical study and classification. Acta Neurochir (Wien). 2012. 154: 1263-8
2. Alfieri A, Rampp S, Strauss C, Fleischhammer J, Rachinger J, Scheller C. The relationship between nervus intermedius anatomy, ultrastructure, electrophysiology, and clinical function. Usefulness in cerebellopontine microsurgery. Acta Neurochir (Wien). 2014. 156: 403-8
3. Alfieri A, Strauss C, Prell J, Peschke E. History of the nervus intermedius of Wrisberg. Ann Anat. 2010. 192: 139-44
4. Alonso F, Kassem MW, Iwanaga J, Oskouian RJ, Loukas M, Demerdash A. Anterior inferior cerebellar arteries juxtaposed with the internal acoustic meatus and their relationship to the cranial nerve VII/VIII complex. Cureus. 2017. 9: e1570
5. Ashram YA, Jackler RK, Pitts LH, Yingling CD. Intraoperative electrophysiologic identification of the nervus intermedius. Otol Neurotol. 2005. 26: 274-9
6. Burmeister HP, Baltzer PA, Dietzel M, Krumbein I, Bitter T, Schrott-Fischer A. Identification of the nervus intermedius using 3T MR imaging. AJNR Am J Neuroradiol. 2011. 32: 460-4
7. Esposito G, Messina R, Carai A, Colafati GS, Savioli A, Randi F. Cochleovestibular nerve compression syndrome caused by intrameatal anterior inferior cerebellar artery loop: Synthesis of best evidence for clinical decisions. World Neurosurg. 2016. 96: 556-61
8. Fortuna A, La Torre E, Forni C. The cisternal segment of the nevus intermedius of Wrisberg: An anatomical study under the operating microscope. Acta Neurochir (Wien). 1972. 27: 53-62
9. Fernandes EG, van Doormaal T, de Ru S, Miller K, Han KS. Microvascular decompression of the VII/VIII cranial nerve complex for the treatment of intermediate nerve neuralgia: A retrospective case series. Oper Neurosurg (Hagerstown). 2018. 15: 378-85
10. Hunt JR. Geniculate neuralgia (neuralgia of the nervus facialis). Arch Neurol Psychiatry. 1937. 37: 253-85
11. Inoue T, Shima A, Hirai H, Suzuki F, Matsuda M. Nervus intermedius neuralgia treated with microvascular decompression: A case report and review of the literature. NMC Case Rep J. 2017. 4: 75-8
12. McDermott AL, Dutt SN, Irving RM, Pahor AL, Chavda SV. Anterior inferior cerebellar artery syndrome: Fact or fiction. Clin Otolaryngol Allied Sci. 2003. 28: 75-80
13. Oh CS, Chung IH, Lee KS, Tanaka S. Morphological study on the rootlets comprising the root of the intermediate nerve. Anat Sci Int. 2003. 78: 111-3
14. Peris-Celda M, Oushy S, Perry A, Graffeo CS, Carlstrom LP, Zimmerman RS. Nervus intermedius and the surgical management of geniculate neuralgia. J Neurosurg. 2018. 131: 343-51
15. Rhoton AL, Kobayashi S, Hollinshead WH. Nervus intermedius. J Neurosurg. 1968. 29: 609-18
16. Rizer FM, Guthikonda M, Lippy WH, Schuring AG. Simultaneous presentation of facial nerve neuroma and otosclerosis. Am J Otol. 1994. 15: 427-30
17. Rodgers B, Chamoun R, Newell K, Lin J. Nervus intermedius meningioma. J Laryngol Otol. 2014. 128: 543-6
18. Rupa V, Saunders RL, Weider DJ. Geniculate neuralgia: The surgical management of primary otalgia. J Neurosurg. 1991. 75: 505-11
19. Scheller C, Rachinger J, Prell J, Kornhuber M, Strauss C. Schwannoma of the intermediate nerve. J Neurosurg. 2008. 109: 144-8
20. Sherman JD, Dagnew E, Pensak ML, van Loveren HR, Tew JM. Facial nerve neuromas: Report of 10 cases and review of the literature. Neurosurgery. 2002. 50: 450-6
21. Tang IP, Freeman SR, Kontorinis G, Tang MY, Rutherford SA, King AT. Geniculate neuralgia: A systematic review. J Laryngol Otol. 2014. 128: 394-9
22. Tubbs RS, Steck DT, Mortazavi MM, Cohen-Gadol AA. The nervus intermedius: A review of its anatomy, function, pathology, and role in neurosurgery. World Neurosurg. 2013. 79: 763-7