- Department of Pathology, Beth Israel Deaconess Medical Center, Harvard Medical School Boston, Massachusetts, United States.
- Department of Surgery, Beth Israel Deaconess Medical Center, Harvard Medical School Boston, Massachusetts, United States.
Correspondence Address:
Hemant Varma
Department of Pathology, Beth Israel Deaconess Medical Center, Harvard Medical School Boston, Massachusetts, United States.
DOI:10.25259/SNI_904_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Jared T Ahrendsen1, Justin M. Moore2, Hemant Varma1. Neuroglial heterotopia of the middle ear: A case series and systematic literature review. 17-Feb-2021;12:60
How to cite this URL: Jared T Ahrendsen1, Justin M. Moore2, Hemant Varma1. Neuroglial heterotopia of the middle ear: A case series and systematic literature review. 17-Feb-2021;12:60. Available from: https://surgicalneurologyint.com/surgicalint-articles/neuroglial-heterotopia-of-the-middle-ear-a-case-series-and-systematic-literature-review/
Abstract
Background: The differential diagnosis for mass forming lesions of the middle ear is broad. While uncommon, neuroglial heterotopias can occur in the middle ear and can be a source of diagnostic confusion for clinician, radiologist, and pathologist alike.
Methods: We identified three cases of neuroglial heterotopia of the middle ear in our institutional archives from 2000 to 2020 and performed extensive histological and immunohistochemical characterization of the three lesions. We conducted a systematic literature review to identify 27 cases published in the English literature between the years 1980 and 2020. Only cases with histological verification of neuroglial heterotopia specifically involving the middle ear were included. We compiled the clinical, radiological, and histopathological findings for all 30 cases.
Results: Patients most frequently presented with chronic otitis media (40%), hearing loss (40%), or prior history of ear surgery or trauma (13%). The median age at surgery was 49 years with a male predominance (M:F 2:1); however, a bimodal age distribution was noted with an earlier onset (11 years or younger) in a subset of patients. Immunohistochemical characterization showed mature neuronal and reactive glial populations with low Ki67 proliferation index and chronic inflammatory infiltrates. There was no neuronal dysplasia or glial atypia, consistent with benign, nonneoplastic, mature glioneuronal tissue.
Conclusion: Immunohistochemical characterization of these lesions and clinical follow-up confirms their benign natural history. Potential etiologies include developmental misplacement, trauma, and chronic inflammation/ reactive changes resulting in sequestered encephalocele.
Keywords: Heterotopia, Middle ear, Neuro-otology, Skull base
INTRODUCTION
Extracranial neuroglial heterotopias are infrequent lesions that present as masses and can be a source of diagnostic confusion for the surgeon, radiologist, and pathologist, and need to be distinguished from neoplasms to avoid unnecessary overtreatment. These lesions most commonly occur in midline structures of the head and neck, particularly the nasal cavity.[
Neuroglial heterotopias in nonmidline structures, such as the middle ear, are rare with only cases reports and two limited case series published in the literature.[
The histopathological features of these lesions have been described in very few cases and are limited in overall scope and completeness.[
MATERIALS AND METHODS
This study was approved under Institutional Review Board (IRB) protocol number 2016-p-000316, which includes waived requirement for patient consent on archived pathology tissue.
We identified three cases of neuroglial heterotopia of the middle ear in our institutional archives from 2000 to 2020. Clinical records were abstracted for demographic information, presenting symptoms, prior surgical history, diagnostic imaging, and clinical follow-up. Surgical specimens were processed by formalin fixation and paraffin embedding and stained with hematoxylin and eosin (H and E). All antibodies used in this study (GFAP, Olig2, synaptophysin, NeuN, CD34, and Ki67) were obtained from Dako (Agilent Pathology Solutions, Santa Clara, CA). Immunohistochemistry staining was performed on a Dako Autostainer Link48 (Agilent Pathology Solutions, Santa Clara, CA), according to our institution’s standard operating procedure.
A literature review was performed by searching the PubMed from years 1980 to 2020 according to PRISMA guidelines. Globally, our study included human patients of any age reported in the English literature published after 1980 to cover all reports in the era of modern imaging. The MEDLINE database was searched with the PubMed search engine with the following Boolean phrase: (“neuroglial” OR “brain” OR “neural” OR “glial”) AND (“heterotopia” OR “heterotopic” OR “ectopia” OR “ectopic” OR “choristoma” OR “sequestered encephalocele”) AND “middle ear.” After applying an English language filter, the search returned 21 results. First, article titles and abstracts were screened for applicability and availability and suitable abstracts were reviewed in detail. Then, a detailed review of the full article was performed for applicability. This review resulted in selection of 14 articles outlining 27 separate cases of neuroglial heterotopia of the middle ear. A symptom or finding was deemed present if mentioned in the article at least once and deemed absent or not present if not mentioned. Data were tabulated.
RESULTS
Clinical history
Case 1
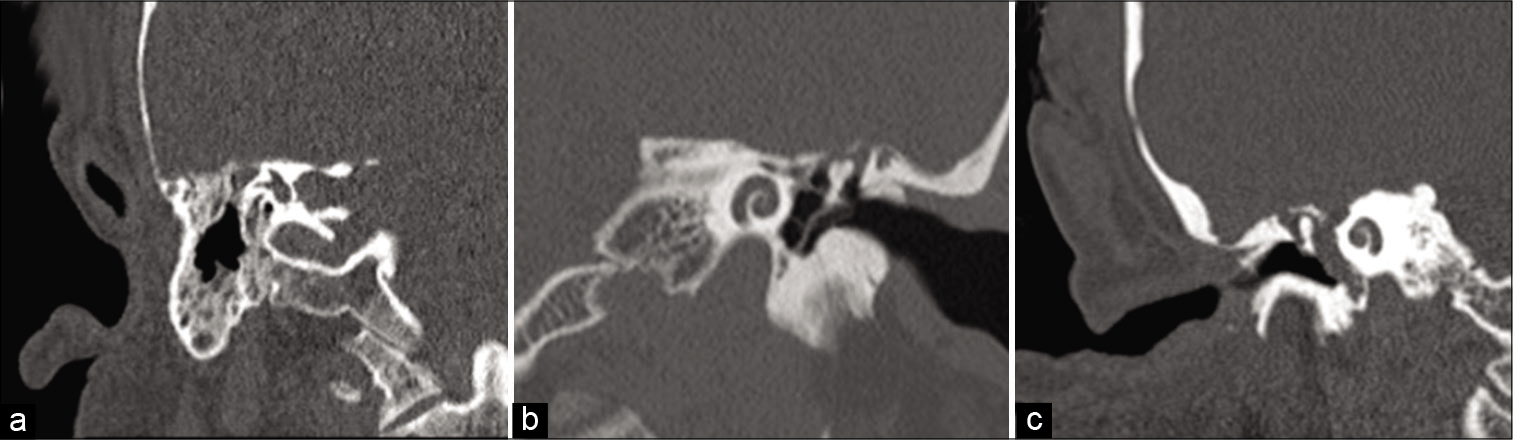
A 49-year-old male who presented with recurrent chronic otitis media and bilateral conductive hearing loss. He previously had Eustachian tubes placed many years prior. The patient had no history of congenital anomalies. Computed topography (CT) scans demonstrated opacification of the mastoids suggesting bilateral mastoiditis, as well as a soft-tissue mass within the right epitympanum [
Case 2
A 49-year-old male who presented with chronic left-sided otitis media and the sensation of left ear fullness. Eustachian tube placement relieved the pressure temporarily; however, symptoms recurred after 2 years. The patient had no history of congenital anomalies, prior trauma, or other surgical procedures in the area. Head CT was significant for a soft-tissue mass in the middle ear with a small equivocal defect in the tegmen tympani [
Case 3
A 48-year-old woman who presented with progressive bilateral decrease in hearing over 2 years with occasional tinnitus and right ear fullness. An audiogram revealed mild-to-moderate conductive hearing loss in the right ear. Prior history was notable for chronic otitis media as a child but no history of congenital anomalies, prior surgery, or local trauma. Examination revealed a bulging but intact right tympanic membrane. Head CT revealed complete opacification of the right middle ear cavity with possible though not definitive dehiscence of the tegmen tympani [
Clinical follow-up data were limited in Cases 1 and 2, with no recurrence or complications at 1.5 and 1 month following surgery, respectively. These patients were subsequently lost to follow up. The patient in case 3 experienced no recurrence of symptoms or relapse 4 years after surgery.
Histopathology
Gross examination of the resected masses revealed variably colored fragments of soft tissue that were tan-pink in Case 1 (up to 0.6 cm), yellow-pink in Case 2 (0.4 cm), and pink-red in Case 3 (0.5 cm). All three cases had similar histological findings and revealed mildly hypercellular mature neuroglial tissue with reactive astrogliosis [
Figure 2:
Histological features of neuroglial heterotopia (H and E). Mature neuroglial tissue with reactive gliosis in background neuropil was apparent in Case 1 (a), Case 2 (b), and Case 3 (c). Rosenthal fibers were apparent in Case 1 (d) and prominent macrophage infiltration was seen in Case 1 (d) and Case 3 (f). Glandular middle ear mucosa was present along the edge of Case 2 (e). Images a-c, e and f at ×200 . Image d at ×400 .
The lesions were further characterized by immunohistochemistry to assess the neuroglial origin of the tissue and to exclude neoplasm. All three cases were diffusely positive for GFAP, which also highlighted reactive gemistocytic astrocytes [
Figure 3:
Immunohistochemical analysis of neuroglial tissue from the middle ear. (a) GFAP and (b) Olig2 highlight astrocytes and oligodendrocytes, respectively, without atypia. (c) NeuN demonstrates the presence of mature-appearing differentiated neurons. (d) Synaptophysin highlights the background neuropil. (e) CD34 highlights background blood vessels without aberrant neuronal expression. (f) Numerous CD68-positive macrophages/microglia are present. All images at ×200.
Literature review
We exhaustively reviewed the literature from 1980 to 2020 according to the PRISMA guidelines[
DISCUSSION
Here, we provide a comprehensive clinical, radiological, and histopathological characterization of neuroglial heterotopias of the middle ear. Our results extend findings of prior reports, including age at diagnosis, presenting symptoms, and radiological findings [
The origin of neuroglial heterotopia in the middle ear is uncertain with multiple potential explanations, including aberrant development, trauma, and/or chronic infection/ inflammation of the middle ear cavity.[
An alternative explanation is that a temporary bony defect in the skull base leads to encephalocele formation with sequestration of the herniated brain tissue after subsequent closure of the healed defect. The tegmen tympani is a thin bone that separates the middle ear cavity from the cranial cavity. Repeated bouts of inflammation, infection, and/or trauma to this area of bone could create a temporary breach, resulting in encephalocele formation. Once the inflammation resolves, healing and fibrosis may sequester and isolate neuroglial tissue within the middle ear without radiologic or clinical evidence of connection to the CNS. Given the thin separation between the intracranial space and the middle ear by the tegmen tympani, combined with a propensity toward middle ear infection/inflammation in the patients presented here, this pathological mechanism seems plausible, especially in patients with a prior history of infections or trauma.
The correct diagnosis of middle ear neuroglial heterotopias has important clinical implications. Presenting symptoms are generally vague and nonspecific. However, once a mass is identified (typically by imaging studies), the differential diagnosis of isolated mass lesions of the middle ear is broad and includes cholesteatoma, adenoma, paraganglioma, teratoma, meningioma, retrotympanic vascular tumor, endolymphatic sac tumor, neuroma, schwannoma, and carcinoma. When heterotopic tissue is encountered in the middle ear, it is most commonly composed of salivary gland tissue.[
CONCLUSION
We present the clinical, radiological, and detailed histopathological characteristics of neuroglial heterotopia in the middle ear. Immunohistochemical characterization and a low Ki67 proliferation index of these lesions confirm their benign natural history. The reactive inflammatory changes and history of prior surgery/trauma or chronic infection/ inflammation suggest potential etiologies for these lesions. Heterotopic neuroglial tissue should be on the differential for mass lesions of the middle ear and correctly recognized as a benign, nonneoplastic lesion with favorable long-term outcome after surgery.
Declaration of patient consent
Institutional Review Board permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Aanaes K, Hasselby JP, Bilde A, Therkildsen MH, von Buchwald C. Heterotopic neuroglial tissue: Two cases involving the tongue and the buccal region. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008. 105: e22-9
2. Al-Ammar AY, Noumas HS, Alqahtani M. A midline nasopharyngeal heterotopic neuroglial tissue. J Laryngol Otol. 2006. 120: e25
3. Anjaneyulu C, Deka RC. Heterotopic neuroglial tissue in hard palate. Indian J Pediatr. 2004. 71: 451-2
4. Anthwal N, Thompson H. The development of the mammalian outer and middle ear. J Anat. 2016. 228: 217-32
5. Buckmiller LM, Brodie HA, Doyle KJ, Nemzek W. Choristoma of the middle ear: A component of a new syndrome?. Otol Neurotol. 2001. 22: 363-8
6. Chen D, Dedhia K, Ozolek J, Mehta D. Case series of congenital heterotopic neuroglial tissue in the parapharyngeal space. Int J Pediatr Otorhinolaryngol. 2016. 86: 77-81
7. Corsi A, Veccia N, Riminucci M. Glial choristoma of the tongue, Clinicopathological analysis of a case and pathogenetic insights. Pol J Pathol. 2017. 68: 162-7
8. Drapkin AJ. Rudimentary cephalocele or neural crest remnant?. Neurosurgery. 1990. 26: 667-73
9. Farneti P, Balbi M, Foschini MP. Neuroglial choristoma of the middle ear. Acta Otorhinolaryngol Ital. 2007. 27: 94-7
10. Francis HW, Nager GT, Holliday MJ, Long DM. Association of heterotopic neuroglial tissue with an arachnoid cyst in the internal auditory canal. Skull Base Surg. 1995. 5: 37-49
11. Glasscock ME, Dickins JR, Jackson CG, Wiet RJ, Feenstra L. Surgical management of brain tissue herniation into the middle ear and mastoid. Laryngoscope. 1979. 89: 1743-54
12. Gyure KA, Thompson LD, Morrison AL. A clinicopathological study of 15 patients with neuroglial heterotopias and encephaloceles of the middle ear and mastoid region. Laryngoscope. 2000. 110: 1731-5
13. Heffner DK. Brain in the middle ear or nasal cavity: Heterotopia or encephalocele?. Ann Diagn Pathol. 2004. 8: 252-7
14. Kamerer DB, Caparosa RJ. Temporal bone encephalocele-Diagnosis and treatment. Laryngoscope. 1982. 92: 878-82
15. Klein MV, Schwaighofer BW, Sobel DF, Fantozzi RD, Hesselink JR. Heterotopic brain in the middle ear: CT findings. J Comput Assist Tomo. 1989. 13: 1058-60
16. Lee JI, Kim KK, Park YK, Eah KY, Kim JR. Glial choristoma in the middle ear and mastoid bone: A case report. J Korean Med Sci. 2004. 19: 155-8
17. Levine PA, Gulya AJ, Glasscock ME, Pensak ML. Neural choristoma of the middle ear. Otolaryngol Head Neck Surg. 1987. 97: 52-6
18. Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK. The 2016 World Health Organization classification of tumors of the central nervous system: A summary. Acta Neuropathol. 2016. 131: 803-20
19. Martinez-Peñuela A, Quer S, Beloqui R, Bulnes MD, Martinez-Peñuela JM. Glial choristoma of the middle ear: Report of 2 cases. Otol Neurotol. 2011. 32: e26-7
20. McGregor DH, Cherian R, Kepes JJ, Kepes M. Case reports: Heterotopic brain tissue of middle ear associated with cholesteatoma. Am J Med Sci. 1994. 308: 180-3
21. Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009. 6: e1000097
22. Penner CR, Thompson LD. Nasal glial heterotopia: A clinicopathologic and immunophenotypic analysis of 10 cases with a review of the literature. Ann Diagn Pathol. 2003. 7: 354-9
23. Plontke SK, Preyer S, Pressler H, Mundinger PM, Plinkert PK. Glial lesion of the infratemporal fossa presenting as a soft tissue middle ear mass-rudimentary encephalocele or neural Wcrest remnant?. Int J Pediatr Otorhinolaryngol. 2000. 56: 141-7
24. Quatre R, Baguant A, Gil H, Schmerber S. Glial heterotopia of the middle ear. Eur Ann Otorhinolaryngol Head Neck Dis. 2020. 137: 207-9
25. Shemanski KA, Voth SE, Patitucci LB, Ma Y, Popnikolov N, Katsetos CD. Glial choristoma of the middle ear. Ear Nose Throat J. 2013. 92: 555-7
26. Shim HJ, Kang YK, An YH, Hong YO. Neuroglial choristoma of the middle ear with massive tympanosclerosis: A case report and literature review. J Audiol Otol. 2016. 20: 179-82
27. Slater DN, Timperley WR, Smith CM. Heterotopic middle ear gliomatosis. Histopathology. 1988. 12: 230-1
28. Uğuz MZ, Arslanoğlu S, Terzi S, Etit D. Glial heterotopia of the middle ear. J Laryngol Otol. 2007. 121: e4
29. Waldman EH, DeSantis E, Lustig LR, Tunkel DE. Pathology quiz case. Arch Otolaryngol Head Neck Surg. 2005. 131: 363